Region:Global
Author(s):Geetanshi
Product Code:KRAC4421
Pages:83
Published On:October 2025
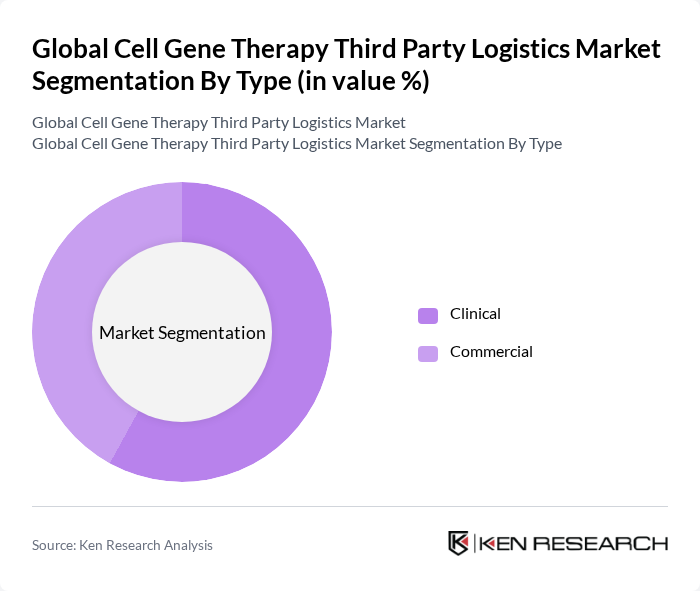
By Type:The market is segmented into Clinical and Commercial logistics. Clinical logistics focuses on the transportation and storage of investigational therapies, while Commercial logistics deals with the distribution of approved therapies to healthcare providers and patients. The Commercial segment is experiencing the fastest growth due to the increasing number of therapy approvals and the need for robust cold chain infrastructure to support widespread distribution. However, the Clinical segment continues to hold a significant share due to the high volume of ongoing clinical trials and the specialized handling required for investigational products .
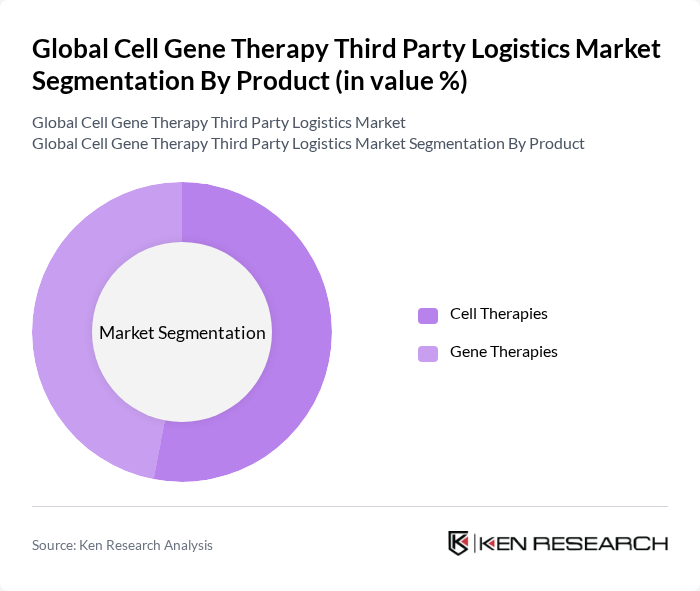
By Product:The market is divided into Cell Therapies and Gene Therapies. Cell Therapies involve the use of living cells to treat diseases, while Gene Therapies focus on modifying or manipulating genes to treat or prevent diseases. The Cell Therapies segment currently leads the market, reflecting the broader adoption and availability of cell-based treatments. Gene Therapies, while growing rapidly, are still emerging and require even more stringent logistics due to their high value and sensitivity .
The Global Cell Gene Therapy Third Party Logistics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Cencora Corporation (formerly AmerisourceBergen), McKesson Corporation, Cardinal Health, DHL Supply Chain, FedEx Corporation, United Parcel Service of America, Inc. (UPS), Kuehne + Nagel International AG, Marken (a UPS Healthcare company), Cryoport, Inc., EVERSANA, Knipper Health, Arvato SE (Bertelsmann), World Courier (AmerisourceBergen), BioLife Solutions, Inc., Thermo Fisher Scientific Inc. contribute to innovation, geographic expansion, and service delivery in this space .
The future of the cell gene therapy logistics market is poised for significant transformation, driven by technological advancements and evolving patient needs. As the integration of artificial intelligence and digital supply chain solutions becomes more prevalent, logistics providers will enhance operational efficiency and responsiveness. Additionally, the emphasis on patient-centric logistics will reshape service offerings, ensuring that therapies are delivered in a timely manner, ultimately improving patient outcomes and satisfaction across the healthcare landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Commercial |
| By Product | Cell Therapies Gene Therapies |
| By Temperature Range | Cryogenic Storage Frozen Storage Refrigerated Storage Ambient Storage |
| By Therapeutic Area | Oncology Cardiovascular Diseases Neurology Infectious Diseases Others |
| By End-Use | Biopharmaceutical Companies Research Institutions Hospitals and Treatment Centers Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Manufacturing Logistics | 100 | Logistics Managers, Supply Chain Directors |
| Clinical Trial Supply Chain Management | 60 | Clinical Operations Managers, Trial Coordinators |
| Cold Chain Logistics for Biologics | 50 | Cold Chain Specialists, Warehouse Managers |
| Regulatory Compliance in Gene Therapy Logistics | 40 | Regulatory Affairs Managers, Quality Assurance Officers |
| Patient-Centric Logistics Solutions | 70 | Patient Engagement Managers, Healthcare Logistics Coordinators |
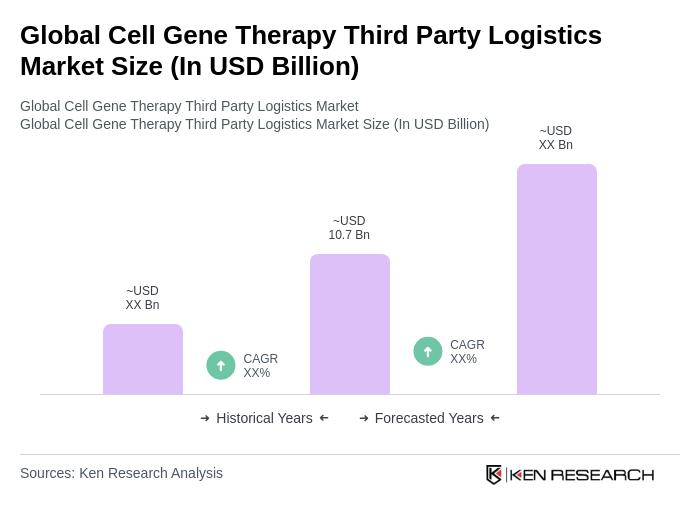
The Global Cell Gene Therapy Third Party Logistics Market is valued at approximately USD 10.7 billion, reflecting significant growth driven by the demand for advanced therapies and specialized logistics solutions for sensitive biological materials.