Region:Global
Author(s):Shubham
Product Code:KRAD3672
Pages:80
Published On:November 2025
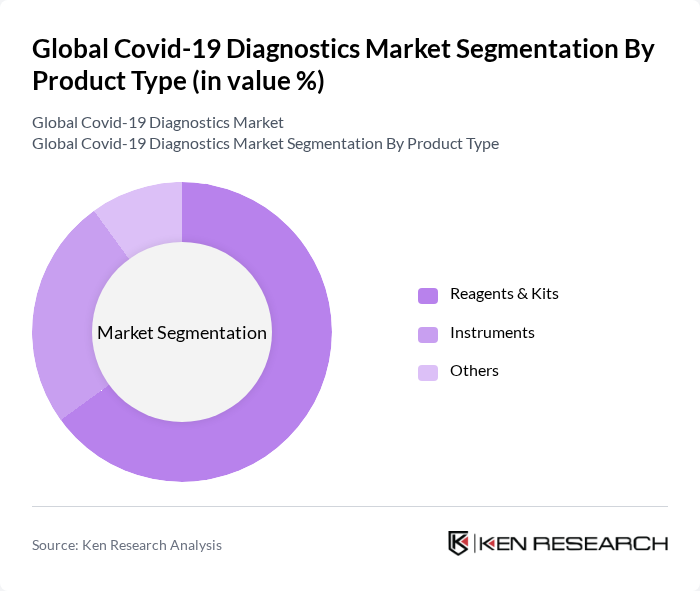
By Product Type:The product type segmentation includes Reagents & Kits, Instruments, and Others. Reagents & Kits are the most significant contributors to the market due to their essential role in testing processes, rapid scalability, and compatibility with both centralized and decentralized testing environments. The demand for these products surged as healthcare facilities required reliable and efficient solutions to manage the pandemic, with molecular and antigen-based kits leading adoption. Instruments, while also crucial, have seen a slower growth rate compared to reagents and kits, as they often require significant capital investment, specialized infrastructure, and trained personnel .
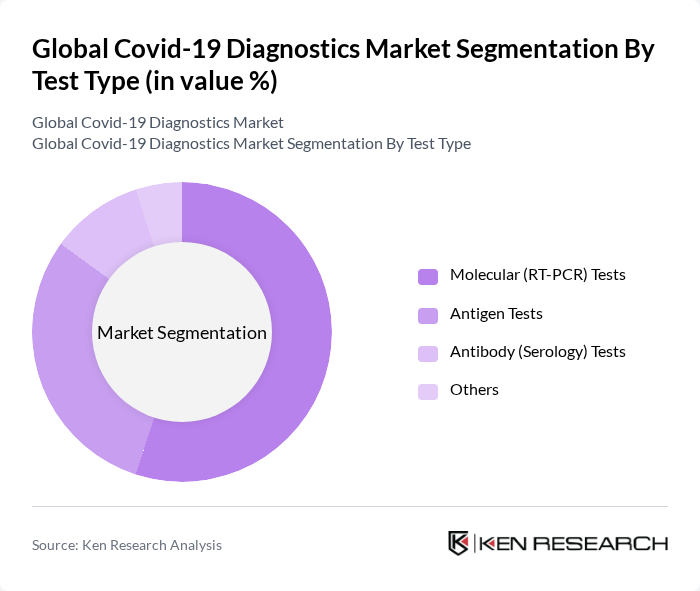
By Test Type:This segmentation includes Molecular (RT-PCR) Tests, Antigen Tests, Antibody (Serology) Tests, and Others. Molecular tests, particularly RT-PCR, dominate the market due to their high accuracy and reliability in detecting active infections, and their continued use as the gold standard for Covid-19 diagnosis. Antigen tests have gained popularity for their rapid turnaround, ease of use, and suitability for mass screening, while antibody tests are essential for epidemiological studies and understanding population-level immunity. The increasing need for timely, scalable, and accurate testing solutions has driven the growth of these test types significantly .
The Global Covid-19 Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Thermo Fisher Scientific, Becton, Dickinson and Company, Cepheid, Hologic, Inc., Bio-Rad Laboratories, QuidelOrtho Corporation, PerkinElmer, Inc., GenMark Diagnostics, Mesa Biotech, Luminex Corporation, Agilent Technologies, Mylab Discovery Solutions Pvt. Ltd., SD Biosensor, Inc., bioMérieux SA, QIAGEN N.V., Danaher Corporation, Seegene Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Covid-19 diagnostics market is poised for continued evolution, driven by technological advancements and changing consumer preferences. The shift towards home testing kits is expected to gain momentum, with an estimated 50 million kits projected to be sold in the future. Additionally, the integration of artificial intelligence in diagnostics will enhance accuracy and efficiency, streamlining testing processes. As telehealth services expand, the demand for remote diagnostics will further shape the market landscape, fostering innovation and accessibility in healthcare solutions.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Reagents & Kits Instruments Others |
| By Test Type | Molecular (RT-PCR) Tests Antigen Tests Antibody (Serology) Tests Others |
| By Technology | RT-PCR Technology CRISPR Technology Next-Generation Sequencing Immunoassay Others |
| By Sample Type | Nasopharyngeal Swabs Saliva Samples Blood Samples Others |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Home Care Settings Government Agencies Others |
| By Distribution Channel | Direct Sales Online Sales Retail Pharmacies Others |
| By Region | North America (U.S., Canada, Mexico) Europe (Germany, France, U.K., Italy, Spain, Rest of Europe) Asia-Pacific (China, Japan, India, South Korea, Australia, Rest of Asia-Pacific) Latin America (Brazil, Argentina, Rest of Latin America) Middle East & Africa (Saudi Arabia, UAE, South Africa, Rest of Middle East & Africa) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Quality Control Officers |
| Healthcare Providers | 80 | Physicians, Nurse Practitioners |
| Diagnostic Test Manufacturers | 60 | Product Managers, R&D Directors |
| Public Health Agencies | 50 | Epidemiologists, Public Health Officials |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
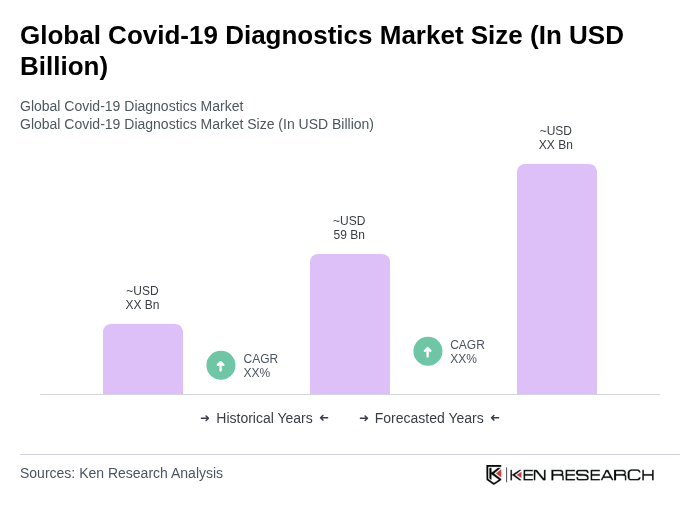
The Global Covid-19 Diagnostics Market is valued at approximately USD 59 billion, reflecting significant growth driven by the urgent need for rapid and accurate testing solutions during the pandemic and increased investments in healthcare infrastructure.