Region:Global
Author(s):Geetanshi
Product Code:KRAE0633
Pages:99
Published On:December 2025
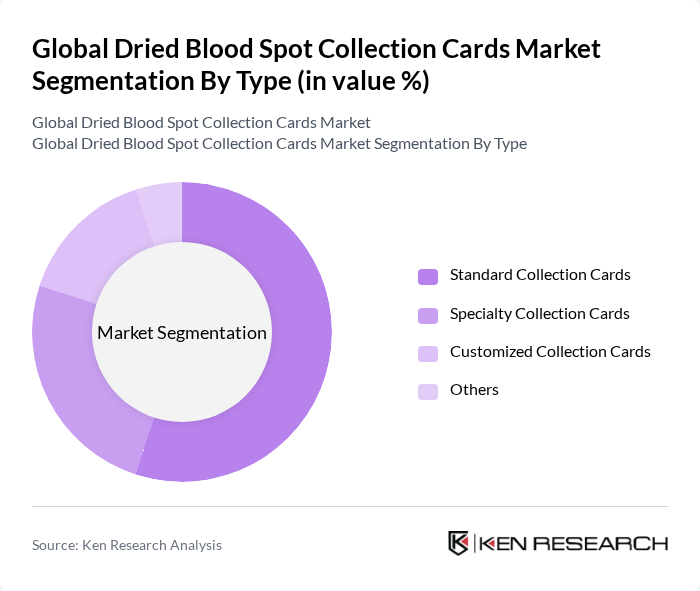
By Type:The market is segmented into various types of dried blood spot collection cards, including Standard Collection Cards, Specialty Collection Cards, Customized Collection Cards, and Others. Among these, Standard Collection Cards dominate the market due to their widespread use in routine testing and established protocols in healthcare settings. Their reliability and cost-effectiveness make them the preferred choice for many healthcare providers, leading to a significant market share.
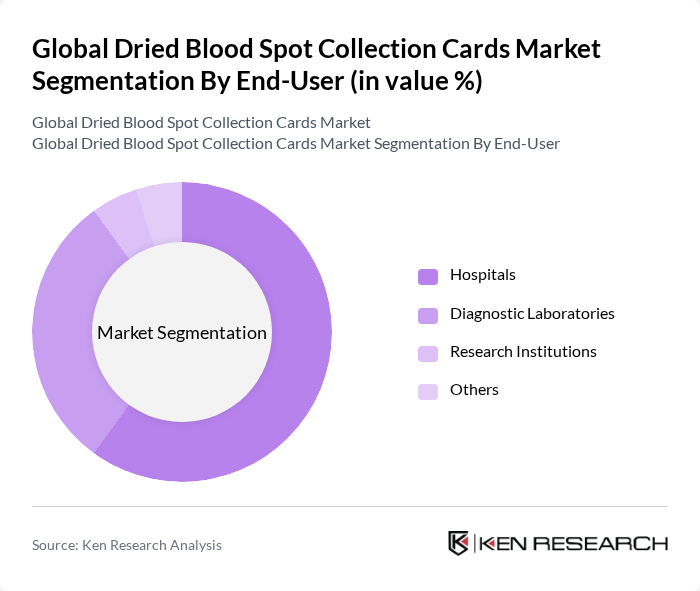
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, and Others. Hospitals are the leading end-users of dried blood spot collection cards, primarily due to their extensive use in newborn screening and routine diagnostic tests. The increasing number of hospitals adopting advanced diagnostic technologies further solidifies their position as the dominant end-user segment.
The Global Dried Blood Spot Collection Cards Market is characterized by a dynamic mix of regional and international players. Leading participants such as Neoteryx, PerkinElmer, Thermo Fisher Scientific, Becton, Dickinson and Company, F. Hoffmann-La Roche AG, Merck KGaA, Bio-Rad Laboratories, QIAGEN, Abcam plc, SeraCare Life Sciences, Greiner Bio-One, Ahlstrom-Munksjö, HemaCare Corporation, LGC Limited, Genomatix Software GmbH contribute to innovation, geographic expansion, and service delivery in this space.
The future of the dried blood spot collection cards market appears promising, driven by the increasing integration of digital health technologies and a growing emphasis on home healthcare solutions. As patients seek more convenient testing options, the demand for at-home diagnostic tools is expected to rise. Furthermore, the focus on sustainability will likely lead to the development of eco-friendly collection products, aligning with global health initiatives aimed at reducing environmental impact while improving patient care.
| Segment | Sub-Segments |
|---|---|
| By Type | Standard Collection Cards Specialty Collection Cards Customized Collection Cards Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Others |
| By Application | Infectious Disease Testing Genetic Testing Therapeutic Drug Monitoring Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology | Microfluidics Capillary Action Others |
| By Packaging Type | Individual Packs Bulk Packs Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Quality Control Officers |
| Healthcare Providers | 80 | Physicians, Nurse Practitioners |
| Manufacturers of Collection Cards | 60 | Product Development Managers, Sales Directors |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Research Institutions | 70 | Research Scientists, Lab Technicians |
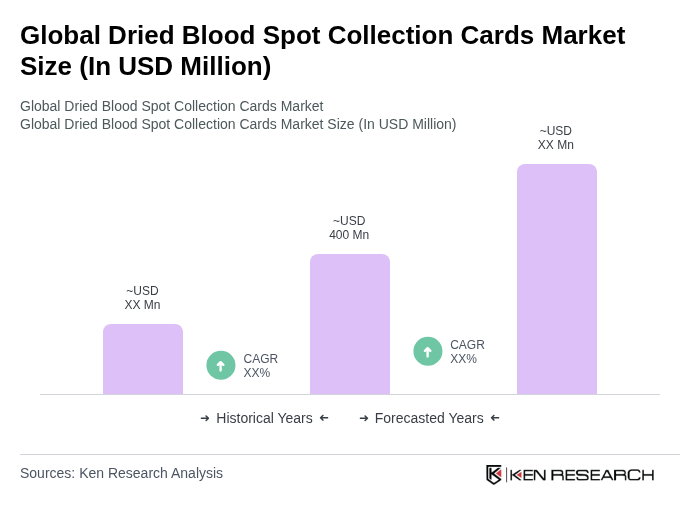
The Global Dried Blood Spot Collection Cards Market is valued at approximately USD 400 million, reflecting a significant growth driven by factors such as expanding newborn screening programs and the rising prevalence of genetic and chronic diseases.