Region:Global
Author(s):Dev
Product Code:KRAC4174
Pages:94
Published On:October 2025
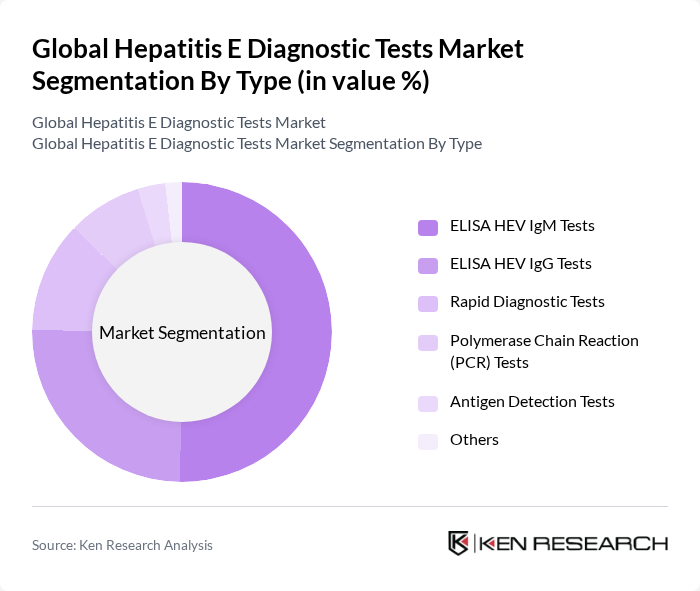
By Type:The market is segmented into various types of diagnostic tests, including ELISA HEV IgM Tests, ELISA HEV IgG Tests, Rapid Diagnostic Tests, Polymerase Chain Reaction (PCR) Tests, Antigen Detection Tests, and Others. Among these, ELISA HEV IgM Tests dominate the market due to their high sensitivity, ease of use, and cost-effectiveness in detecting acute infections. The demand for PCR Tests is also increasing, as they provide rapid and accurate results, which are crucial for timely treatment.
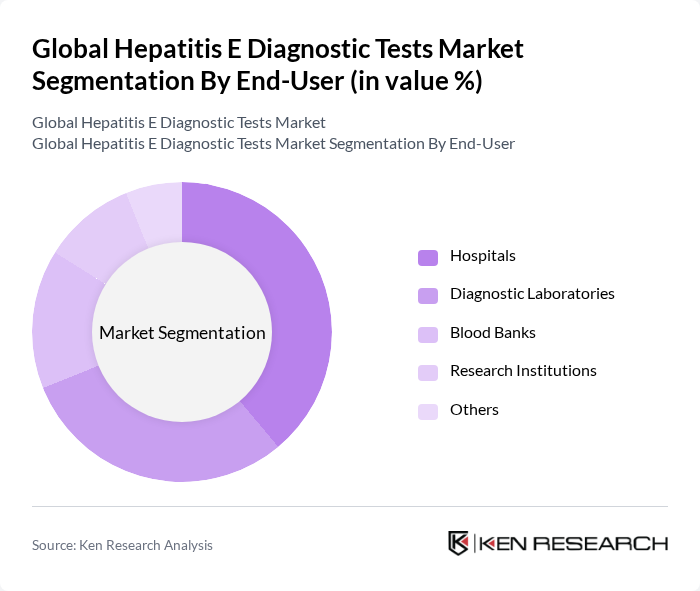
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Blood Banks, Research Institutions, and Others. Hospitals are the leading end-users of hepatitis E diagnostic tests, primarily due to their need for accurate and timely diagnosis in clinical settings. Diagnostic Laboratories also play a significant role, as they are equipped with advanced testing technologies and cater to a wide range of patients.
The Global Hepatitis E Diagnostic Tests Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Bio-Rad Laboratories, Cepheid, Hologic, Inc., Thermo Fisher Scientific, Ortho Clinical Diagnostics, GenMark Diagnostics, Quidel Corporation, MedMira Inc., DiaSorin S.p.A., Mylab Discovery Solutions, Eiken Chemical Co., Ltd., Fujirebio, Fortress Diagnostics, MP Biomedicals, Mikrogen Diagnostik GmbH, Guangzhou Wondfo Biotech Co., Ltd., GenScript Biotech Corporation, Medsource Ozone Biomedicals Pvt. Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Hepatitis E diagnostic tests market appears promising, driven by technological advancements and increased public health awareness. The integration of AI in diagnostics is expected to enhance test accuracy and efficiency, while the expansion of point-of-care testing will facilitate easier access to diagnostics. Additionally, ongoing collaborations with public health organizations will likely lead to improved testing protocols and increased funding for research initiatives, ultimately contributing to better disease management and control.
| Segment | Sub-Segments |
|---|---|
| By Type | ELISA HEV IgM Tests ELISA HEV IgG Tests Rapid Diagnostic Tests Polymerase Chain Reaction (PCR) Tests Antigen Detection Tests Others |
| By End-User | Hospitals Diagnostic Laboratories Blood Banks Research Institutions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa Others |
| By Application | Clinical Diagnosis Epidemiological Studies Blood Screening Others |
| By Sample Type | Blood Samples Serum Samples Plasma Samples Stool Samples Others |
| By Pricing Range | Low Price Range Mid Price Range High Price Range Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Directors, Diagnostic Test Managers |
| Healthcare Providers | 80 | Infectious Disease Specialists, General Practitioners |
| Public Health Officials | 60 | Health Policy Advisors, Epidemiologists |
| Diagnostic Test Manufacturers | 50 | Product Development Managers, Sales Executives |
| Patient Advocacy Groups | 40 | Patient Representatives, Community Health Workers |
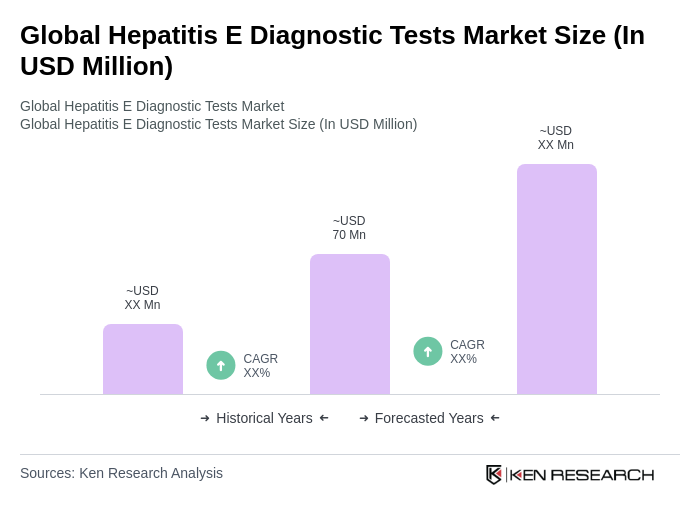
The Global Hepatitis E Diagnostic Tests Market is valued at approximately USD 70 million, reflecting a significant demand for effective diagnostic solutions due to the rising prevalence of hepatitis E infections worldwide.