Region:Global
Author(s):Rebecca
Product Code:KRAD4998
Pages:92
Published On:December 2025
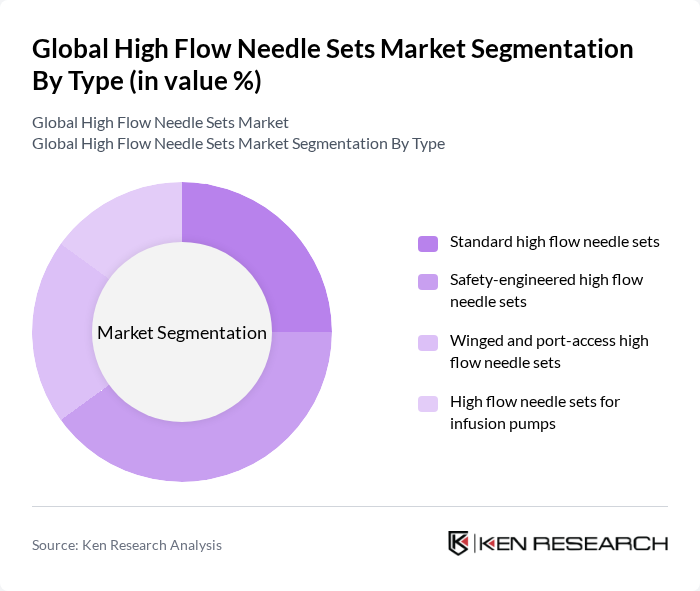
By Type:The market is segmented into four main types of high flow needle sets, which include standard high flow needle sets, safety-engineered high flow needle sets, winged and port-access high flow needle sets, and high flow needle sets for infusion pumps. This structure is aligned with common industry categorizations that separate conventional sets from safety devices, winged or port-access products, and lines optimized for use with infusion and contrast pumps. Among these, safety-engineered high flow needle sets are gaining traction due to their ability to reduce the risk of needlestick injuries, which is a significant concern in healthcare settings, and due to employer obligations under sharps injury prevention standards. The increasing emphasis on patient and healthcare worker safety, infection prevention, and compliance with occupational health regulations is driving the demand for these innovative products.
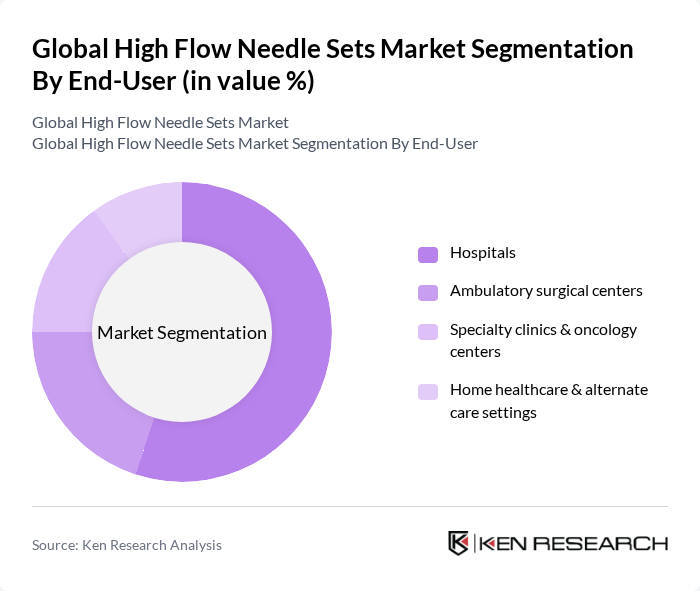
By End-User:The end-user segment includes hospitals, ambulatory surgical centers, specialty clinics & oncology centers, and home healthcare & alternate care settings. Hospitals are the leading end-users of high flow needle sets, driven by the high volume of inpatient and outpatient surgical procedures, emergency care, intensive care, and chemotherapy infusions requiring reliable high-flow access. The increasing number of outpatient and day-care procedures, the expansion of ambulatory surgical centers, and the shift towards home healthcare and home infusion therapies for oncology, parenteral nutrition, and chronic disease management are also contributing to the growth of this segment.
The Global High Flow Needle Sets Market is characterized by a dynamic mix of regional and international players. Leading participants such as Becton, Dickinson and Company (BD), Terumo Corporation, Smiths Group plc (Smiths Medical / ICU Medical), Medtronic plc, Nipro Corporation, Fresenius Medical Care AG & Co. KGaA, Cardinal Health, Inc., Baxter International Inc., B. Braun Melsungen AG, Vygon SA, Teleflex Incorporated, KORU Medical Systems, Inc., Medline Industries, LP, Amsino International, Inc., Thermo Fisher Scientific Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the high flow needle sets market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centered care. As healthcare systems evolve, the integration of smart needle technologies and the expansion of home healthcare services are expected to reshape the landscape in future. Additionally, the growing emphasis on patient safety and comfort will likely lead to further innovations, enhancing the overall effectiveness of needle sets in various medical applications.
| Segment | Sub-Segments |
|---|---|
| By Type | Standard high flow needle sets Safety-engineered high flow needle sets Winged and port-access high flow needle sets High flow needle sets for infusion pumps |
| By End-User | Hospitals Ambulatory surgical centers Specialty clinics & oncology centers Home healthcare & alternate care settings |
| By Application | Oncology and chemotherapy infusion Parenteral nutrition and fluid resuscitation Blood collection and apheresis Pain management & other therapeutic infusions |
| By Material | Stainless steel Polymer-based (e.g., PTFE, polyurethane) Silicone-coated and specialty alloys Others |
| By Distribution Channel | Direct sales to healthcare providers Medical device distributors & wholesalers Group purchasing organizations (GPOs) Online and e-procurement platforms |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Regulatory Compliance | FDA 510(k) and PMA approvals CE marking (MDR compliant) ISO 13485 and related quality certifications Other regional regulatory approvals |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Departments | 120 | Oncologists, Nursing Staff |
| Pediatric Care Units | 90 | Pediatricians, Child Nurses |
| Emergency Medical Services | 80 | Emergency Room Physicians, Paramedics |
| Outpatient Clinics | 100 | General Practitioners, Clinic Managers |
| Healthcare Procurement Departments | 70 | Procurement Officers, Supply Chain Managers |
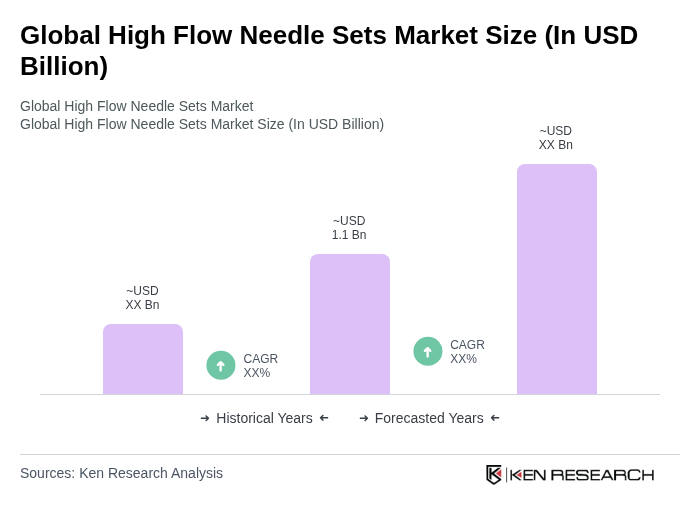
The Global High Flow Needle Sets Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by the increasing prevalence of chronic diseases and advancements in medical technology that enhance patient care and drug delivery efficiency.