About the Report
Base Year 2024Global Large Molecule Drug Substance CDMO Market Overview
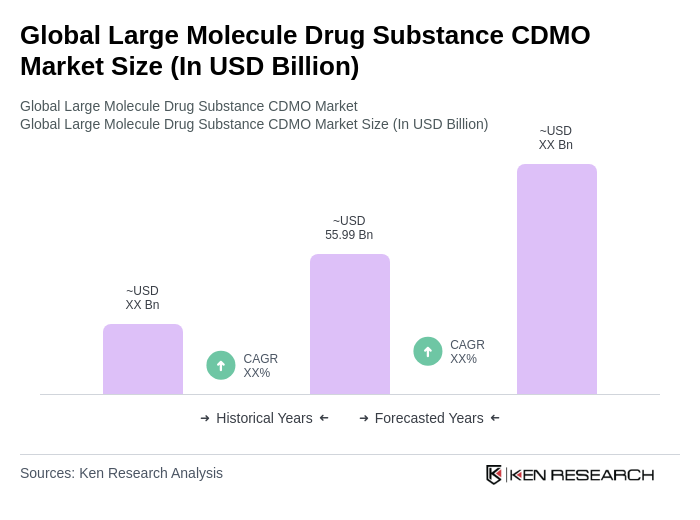
- The Global Large Molecule Drug Substance CDMO Market is valued at USD 55.99 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for biologics, advancements in biopharmaceutical manufacturing technologies, and the rising prevalence of chronic diseases. The market is also supported by the growing trend of outsourcing manufacturing processes by pharmaceutical companies to focus on core competencies. Recent trends include accelerated adoption of advanced bioprocessing technologies, expansion of CDMO service portfolios to include cell and gene therapies, and increased strategic partnerships between CDMOs and pharmaceutical companies to streamline drug development and commercialization .
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany being the most significant contributors. The dominance of these regions can be attributed to their robust healthcare infrastructure, high investment in research and development, and the presence of leading biopharmaceutical companies that drive innovation and production capabilities. North America holds the largest market share, while Asia Pacific is emerging as the fastest-growing region due to increased investments and expansion of manufacturing capacities .
- In 2023, the U.S. FDA implemented the "Modernization of Biologics Regulation Final Rule" (21 CFR Parts 600, 610, 680, and 1271), issued by the U.S. Food and Drug Administration. This regulation updates and strengthens requirements for the approval and manufacturing of biologics, including large molecule drugs, with a focus on enhanced safety, efficacy, and quality control. CDMOs must comply with expanded Good Manufacturing Practice (GMP) standards, rigorous lot release procedures, and updated documentation requirements, directly impacting operational strategies and regulatory compliance for biologics manufacturing .
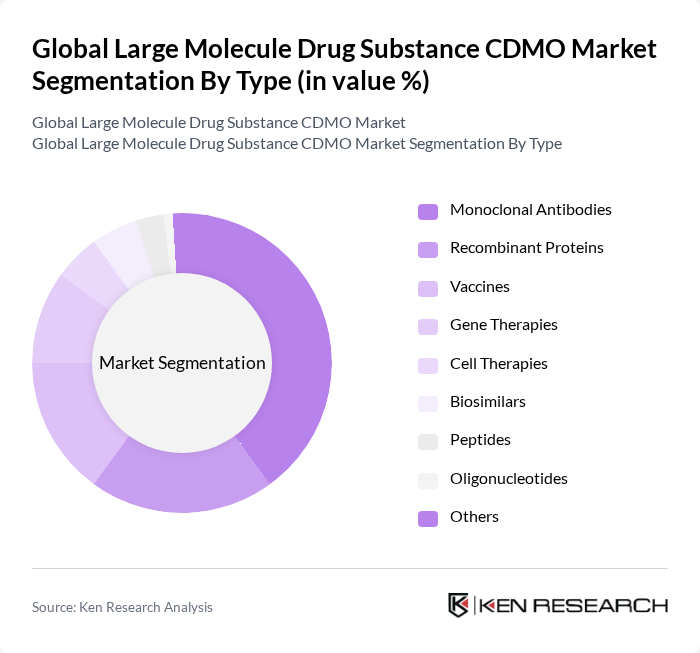
Global Large Molecule Drug Substance CDMO Market Segmentation
By Type:The market is segmented into Monoclonal Antibodies, Recombinant Proteins, Vaccines, Gene Therapies, Cell Therapies, Biosimilars, Peptides, Oligonucleotides, and Others. Monoclonal Antibodies are leading the market due to their widespread application in oncology and autoimmune diseases, supported by significant investments in research and development and the growing demand for targeted therapies. Recombinant proteins and vaccines also represent substantial segments, with cell and gene therapies showing rapid growth as CDMOs expand capabilities in advanced modalities .
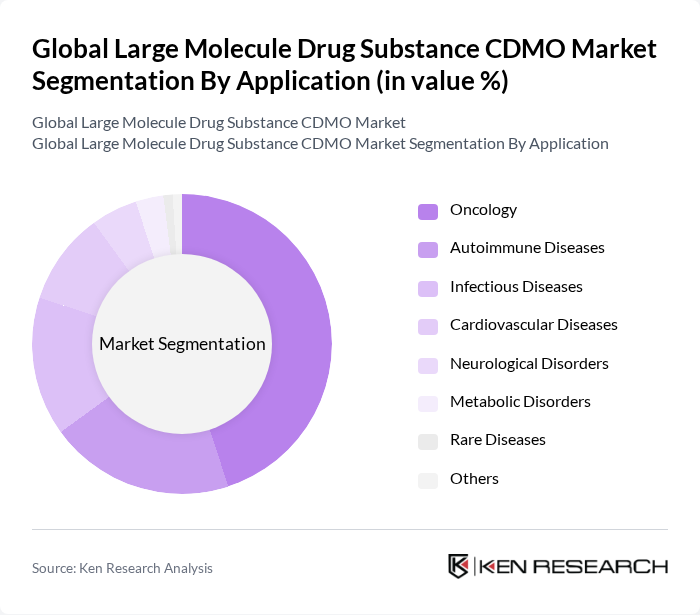
By Application:The applications of large molecule drug substances include Oncology, Autoimmune Diseases, Infectious Diseases, Cardiovascular Diseases, Neurological Disorders, Metabolic Disorders, Rare Diseases, and Others. Oncology remains the leading application area, driven by the increasing incidence of cancer and the demand for innovative therapies that improve patient outcomes. Autoimmune and infectious diseases also account for significant shares, while rare and metabolic disorders are gaining attention due to advancements in precision medicine .
Global Large Molecule Drug Substance CDMO Market Competitive Landscape
The Global Large Molecule Drug Substance CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Samsung Biologics, WuXi AppTec, Catalent, Inc., Boehringer Ingelheim, FUJIFILM Diosynth Biotechnologies, AbbVie Contract Manufacturing, Recipharm AB, AGC Biologics, Rentschler Biopharma SE, KBI Biopharma, Eurofins Scientific, Avid Bioservices, Inc., Siegfried Holding AG, Patheon (Thermo Fisher Scientific) contribute to innovation, geographic expansion, and service delivery in this space.
Global Large Molecule Drug Substance CDMO Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The global biologics market is projected to reach $500 billion in future, driven by the rising prevalence of chronic diseases. In future, biologics accounted for approximately 30% of total pharmaceutical sales, highlighting a significant shift towards these therapies. The World Health Organization reported that biologics are increasingly preferred due to their targeted action and efficacy, further propelling demand for large molecule drug substances in the CDMO sector.
- Advancements in Biomanufacturing Technologies:The biomanufacturing sector is experiencing rapid technological advancements, with investments exceeding $20 billion in future. Innovations such as single-use technologies and continuous manufacturing processes are enhancing production efficiency and reducing time-to-market. According to the International Society for Pharmaceutical Engineering, these advancements are expected to lower operational costs by up to 15%, making large molecule production more attractive for CDMOs and their clients.
- Rising Investment in R&D:Global investment in pharmaceutical R&D reached $200 billion in future, with a significant portion allocated to biologics. The National Institutes of Health reported that funding for biologic research has increased by 10% annually, reflecting a strong commitment to developing new therapies. This surge in R&D investment is expected to drive demand for CDMO services, particularly in the large molecule segment, as companies seek to expedite drug development processes.
Market Challenges
- High Production Costs:The production costs for large molecule drugs can exceed $1,000 per gram, significantly impacting profitability for CDMOs. According to a future report by Deloitte, the high costs associated with raw materials and complex manufacturing processes pose a challenge for many companies. This financial burden can deter smaller firms from entering the market, limiting competition and innovation within the sector.
- Regulatory Compliance Complexity:Navigating the regulatory landscape for large molecule drugs is increasingly complex, with over 50% of CDMOs reporting challenges in compliance. The U.S. Food and Drug Administration and European Medicines Agency have stringent guidelines that require extensive documentation and validation processes. A future survey by BioPharma Dive indicated that 70% of CDMOs face delays due to regulatory hurdles, impacting their ability to meet market demands efficiently.
Global Large Molecule Drug Substance CDMO Market Future Outlook
The future of the large molecule drug substance CDMO market appears promising, driven by the increasing focus on biologics and personalized medicine. As companies continue to invest in advanced manufacturing technologies, the efficiency and scalability of production are expected to improve significantly. Additionally, the growing trend of outsourcing will likely lead to more strategic partnerships, enabling CDMOs to expand their capabilities and enhance service offerings, ultimately meeting the evolving needs of the pharmaceutical industry.
Market Opportunities
- Growth in Biosimilars:The biosimilars market is projected to reach $100 billion in future, presenting significant opportunities for CDMOs. As patents for original biologics expire, the demand for cost-effective alternatives is increasing. This trend is expected to drive collaboration between CDMOs and pharmaceutical companies, enhancing production capabilities and market reach.
- Emerging Markets Expansion:Emerging markets, particularly in Asia-Pacific, are witnessing rapid growth in healthcare spending, projected to reach $1 trillion in future. This expansion presents CDMOs with opportunities to establish operations in these regions, catering to local demand for biologics and large molecule drugs, while benefiting from lower operational costs and favorable regulatory environments.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Recombinant Proteins Vaccines Gene Therapies Cell Therapies Biosimilars Peptides Oligonucleotides Others |
| By Application | Oncology Autoimmune Diseases Infectious Diseases Cardiovascular Diseases Neurological Disorders Metabolic Disorders Rare Diseases Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Contract Research Organizations (CROs) Academic Institutes Others |
| By Production Scale | Clinical Scale Commercial Scale Pilot Scale Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa Others |
| By Service Type | Process Development Analytical Services Manufacturing Services Packaging Services Fill-Finish Services Regulatory Support Others |
| By Contract Type | Full-Service Contracts Project-Based Contracts Dedicated Capacity Agreements Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical and Biotechnology Companies
Contract Development and Manufacturing Organizations (CDMOs)
Supply Chain and Logistics Providers
Quality Assurance and Control Agencies
Pharmaceutical Industry Associations
Insurance Companies and Payers
Players Mentioned in the Report:
Lonza Group AG
Samsung Biologics
WuXi AppTec
Catalent, Inc.
Boehringer Ingelheim
FUJIFILM Diosynth Biotechnologies
AbbVie Contract Manufacturing
Recipharm AB
AGC Biologics
Rentschler Biopharma SE
KBI Biopharma
Eurofins Scientific
Avid Bioservices, Inc.
Siegfried Holding AG
Patheon (Thermo Fisher Scientific)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Large Molecule Drug Substance CDMO Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Large Molecule Drug Substance CDMO Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Large Molecule Drug Substance CDMO Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Biologics
3.1.2 Advancements in Biomanufacturing Technologies
3.1.3 Rising Investment in R&D
3.1.4 Expanding Applications in Therapeutics
3.2 Market Challenges
3.2.1 High Production Costs
3.2.2 Regulatory Compliance Complexity
3.2.3 Limited Skilled Workforce
3.2.4 Supply Chain Disruptions
3.3 Market Opportunities
3.3.1 Growth in Biosimilars
3.3.2 Emerging Markets Expansion
3.3.3 Strategic Partnerships and Collaborations
3.3.4 Technological Innovations in Drug Development
3.4 Market Trends
3.4.1 Shift Towards Outsourcing
3.4.2 Focus on Sustainability
3.4.3 Integration of AI and Automation
3.4.4 Personalized Medicine Development
3.5 Government Regulation
3.5.1 Stringent Quality Control Standards
3.5.2 Intellectual Property Protections
3.5.3 Environmental Regulations
3.5.4 Pricing and Reimbursement Policies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Large Molecule Drug Substance CDMO Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Large Molecule Drug Substance CDMO Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Recombinant Proteins
8.1.3 Vaccines
8.1.4 Gene Therapies
8.1.5 Cell Therapies
8.1.6 Biosimilars
8.1.7 Peptides
8.1.8 Oligonucleotides
8.1.9 Others
8.2 By Application
8.2.1 Oncology
8.2.2 Autoimmune Diseases
8.2.3 Infectious Diseases
8.2.4 Cardiovascular Diseases
8.2.5 Neurological Disorders
8.2.6 Metabolic Disorders
8.2.7 Rare Diseases
8.2.8 Others
8.3 By End-User
8.3.1 Pharmaceutical Companies
8.3.2 Biotechnology Firms
8.3.3 Research Institutions
8.3.4 Contract Research Organizations (CROs)
8.3.5 Academic Institutes
8.3.6 Others
8.4 By Production Scale
8.4.1 Clinical Scale
8.4.2 Commercial Scale
8.4.3 Pilot Scale
8.4.4 Others
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.5.6 Others
8.6 By Service Type
8.6.1 Process Development
8.6.2 Analytical Services
8.6.3 Manufacturing Services
8.6.4 Packaging Services
8.6.5 Fill-Finish Services
8.6.6 Regulatory Support
8.6.7 Others
8.7 By Contract Type
8.7.1 Full-Service Contracts
8.7.2 Project-Based Contracts
8.7.3 Dedicated Capacity Agreements
8.7.4 Others
9. Global Large Molecule Drug Substance CDMO Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (YoY %)
9.2.4 Market Share (%)
9.2.5 Number of Commercial Manufacturing Sites
9.2.6 Capacity Utilization Rate (%)
9.2.7 Customer Retention Rate (%)
9.2.8 Regulatory Inspection Success Rate (%)
9.2.9 R&D Investment as % of Revenue
9.2.10 Number of New Large Molecule Projects Initiated (Annual)
9.2.11 Time-to-Market (Months)
9.2.12 Quality Compliance Rate (%)
9.2.13 Supply Chain Efficiency (On-Time Delivery %)
9.2.14 Innovation Rate (e.g., Patents Filed Annually)
9.2.15 Pricing Strategy (Premium, Value, Cost-Plus, etc.)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 List of Major Companies
9.5.1 Lonza Group AG
9.5.2 Samsung Biologics
9.5.3 WuXi AppTec
9.5.4 Catalent, Inc.
9.5.5 Boehringer Ingelheim
9.5.6 FUJIFILM Diosynth Biotechnologies
9.5.7 AbbVie Contract Manufacturing
9.5.8 Recipharm AB
9.5.9 AGC Biologics
9.5.10 Rentschler Biopharma SE
9.5.11 KBI Biopharma
9.5.12 Eurofins Scientific
9.5.13 Avid Bioservices, Inc.
9.5.14 Siegfried Holding AG
9.5.15 Patheon (Thermo Fisher Scientific)
10. Global Large Molecule Drug Substance CDMO Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Funding Trends
10.1.2 Policy Influence on Procurement
10.1.3 Strategic Partnerships with CDMOs
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Biomanufacturing Facilities
10.2.2 Budget Allocation for R&D
10.2.3 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Quality Assurance Challenges
10.3.2 Supply Chain Reliability Issues
10.3.3 Cost Management Concerns
10.4 User Readiness for Adoption
10.4.1 Training and Development Needs
10.4.2 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics Evaluation
10.5.2 Scalability of Solutions
10.5.3 Long-term Value Assessment
11. Global Large Molecule Drug Substance CDMO Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Direct Sales Approaches
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service Enhancements
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantages
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Selection
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Assessment
14. Potential Partner List
14.1 Distributors Identification
14.2 Joint Ventures Opportunities
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical associations and regulatory bodies
- Review of market trends and forecasts from scientific journals and publications
- Examination of financial reports and press releases from leading CDMO firms
Primary Research
- Interviews with executives from large molecule drug manufacturers
- Surveys targeting R&D heads and project managers in biopharmaceutical companies
- Field interviews with quality assurance and regulatory compliance professionals
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert opinions
- Triangulation of findings from primary interviews and secondary data sources
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global pharmaceutical spending trends
- Segmentation by therapeutic areas and geographical regions
- Incorporation of growth rates from emerging markets and biopharmaceutical sectors
Bottom-up Modeling
- Volume estimates derived from production capacities of key CDMO players
- Cost analysis based on service offerings and pricing models in the industry
- Calculation of market size using volume x average service cost methodology
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating market drivers such as innovation and regulatory changes
- Scenario modeling based on potential shifts in healthcare policies and funding
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical Development | 60 | R&D Directors, Project Managers |
| Manufacturing Operations | 50 | Operations Managers, Production Supervisors |
| Quality Control & Assurance | 40 | Quality Managers, Compliance Officers |
| Regulatory Affairs | 40 | Regulatory Affairs Specialists, Legal Advisors |
| Market Access & Commercialization | 45 | Market Access Managers, Business Development Executives |
Frequently Asked Questions
What is the current value of the Global Large Molecule Drug Substance CDMO Market?
The Global Large Molecule Drug Substance CDMO Market is valued at approximately USD 55.99 billion, reflecting significant growth driven by the increasing demand for biologics and advancements in biopharmaceutical manufacturing technologies.