Global Liver Cancer Diagnostics Market Overview
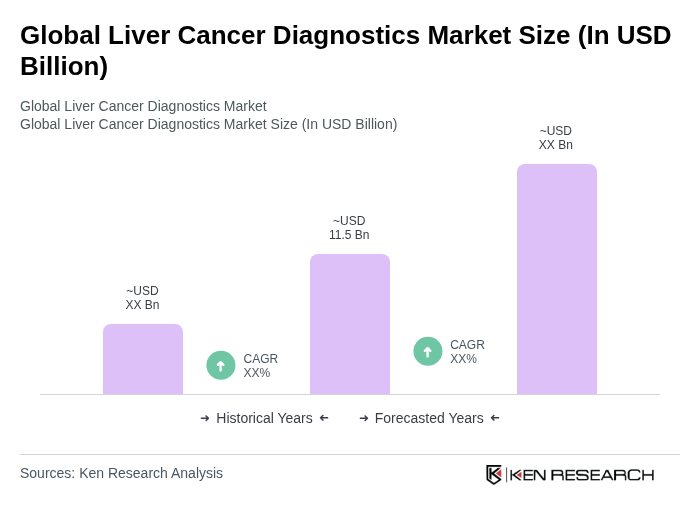
- The Global Liver Cancer Diagnostics Market is valued at USD 11.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of liver cancer, advancements in diagnostic technologies such as AI-assisted imaging, liquid biopsy, and biomarker assays, and rising awareness about early detection among healthcare professionals and patients. The market is also supported by the growing geriatric population, which is more susceptible to liver diseases, and by the expansion of healthcare infrastructure in emerging economies .
- Key players in this market include the United States, Germany, and Japan, which dominate due to their advanced healthcare infrastructure, significant investments in research and development, and high adoption rates of innovative diagnostic technologies. The presence of leading pharmaceutical and biotechnology companies in these regions further enhances their market position .
- In 2023, the U.S. government issued updated screening recommendations for liver cancer through the United States Preventive Services Task Force (USPSTF), which provides evidence-based guidelines for early detection and screening of liver cancer in high-risk populations. These guidelines require healthcare providers to implement best practices for early detection, including the use of imaging and biomarker-based diagnostics, and are supported by funding from the National Institutes of Health (NIH) for research and development in liver cancer diagnostics. (Source: United States Preventive Services Task Force, “Screening for Hepatocellular Carcinoma: Recommendation Statement,” USPSTF, 2023)
Global Liver Cancer Diagnostics Market Segmentation
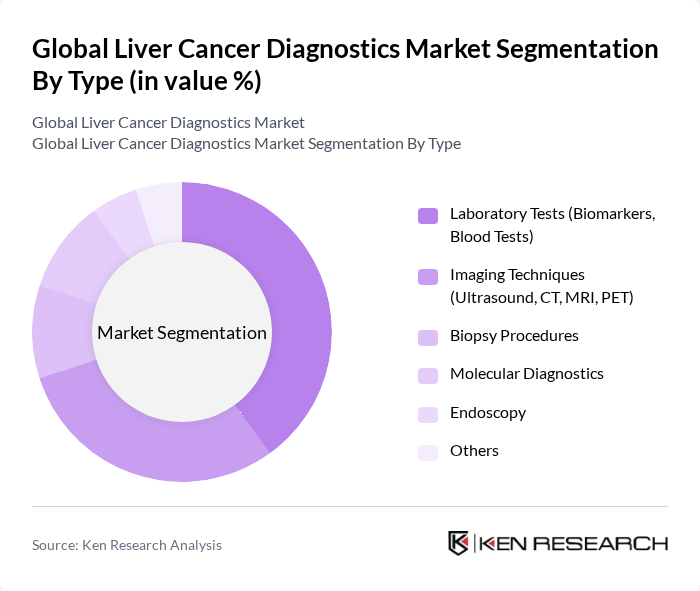
By Type:The market is segmented into various types of diagnostic methods, including laboratory tests, imaging techniques, biopsy procedures, molecular diagnostics, endoscopy, and others. Among these, laboratory tests—particularly blood tests and biomarkers—lead the market due to their non-invasive nature, cost-effectiveness, and high accuracy in early detection and monitoring of liver cancer. Imaging techniques, such as ultrasound, CT, MRI, and PET, are also widely adopted for their ability to provide detailed anatomical and functional information. Molecular diagnostics and liquid biopsy technologies are gaining traction due to their role in personalized medicine and early-stage detection .
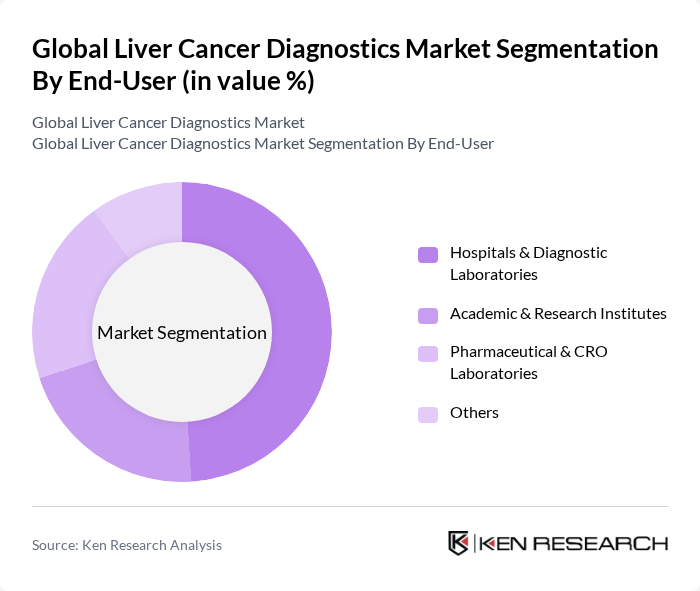
By End-User:The end-user segment includes hospitals and diagnostic laboratories, academic and research institutes, pharmaceutical and CRO laboratories, and others. Hospitals and diagnostic laboratories dominate this segment due to their critical role in providing comprehensive diagnostic services, multidisciplinary expertise, and integrated care for liver cancer patients. The increasing number of liver cancer cases and the need for advanced diagnostic technologies further support the dominance of this segment. Academic and research institutes focus on biomarker discovery and clinical research, while pharmaceutical and CRO laboratories are driven by clinical trials and drug development needs .
Global Liver Cancer Diagnostics Market Competitive Landscape
The Global Liver Cancer Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, bioMérieux, Thermo Fisher Scientific, QIAGEN, Illumina, Hologic, Agilent Technologies, GE Healthcare, Philips Healthcare, PerkinElmer, Becton, Dickinson and Company, Fujifilm Holdings Corporation, Canon Medical Systems Corporation, Guardant Health, Sysmex Corporation, Mindray Medical International, Epigenomics AG, Exact Sciences Corporation contribute to innovation, geographic expansion, and service delivery in this space .
Global Liver Cancer Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Liver Cancer:The incidence of liver cancer is rising significantly, with approximately 905,000 new cases reported globally in 2020, according to the World Health Organization. This alarming trend is driven by factors such as hepatitis infections and alcohol consumption, particularly in regions like Asia and Africa. The World Bank projects that by the future, the number of liver cancer cases could increase by 15%, necessitating enhanced diagnostic solutions to manage this growing health crisis effectively.
- Advancements in Diagnostic Technologies:The liver cancer diagnostics sector is witnessing rapid technological advancements, including the development of imaging techniques and biomarker tests. For instance, the introduction of non-invasive tests has reduced the need for biopsies, improving patient comfort and diagnostic accuracy. The global market for diagnostic imaging is expected to reach $45 billion by the future, driven by innovations in MRI and CT technologies, which are crucial for early liver cancer detection and monitoring.
- Rising Awareness and Screening Programs:Increased public awareness about liver cancer and its risk factors has led to more proactive screening initiatives. In the future, the American Cancer Society reported a 20% increase in liver cancer screenings compared to previous years. Government and non-profit organizations are investing heavily in educational campaigns, with funding exceeding $200 million in the future, aimed at promoting early detection and improving survival rates through timely diagnostics.
Market Challenges
- High Cost of Diagnostic Procedures:The financial burden associated with liver cancer diagnostics remains a significant challenge. Diagnostic tests, including advanced imaging and biomarker assays, can cost upwards of $3,000 per patient. This high cost limits accessibility, particularly in low-income regions where healthcare budgets are constrained. The World Bank estimates that healthcare spending in developing countries averages only $100 per capita, exacerbating disparities in diagnostic availability and leading to late-stage cancer diagnoses.
- Lack of Skilled Professionals:The liver cancer diagnostics market faces a critical shortage of trained healthcare professionals. According to the World Health Organization, there is an estimated deficit of 18 million healthcare workers globally, with diagnostic specialists being particularly scarce. This shortage hampers the effective implementation of screening programs and the timely diagnosis of liver cancer, especially in rural and underserved areas, where access to skilled professionals is limited.
Global Liver Cancer Diagnostics Market Future Outlook
The future of liver cancer diagnostics is poised for transformative growth, driven by technological innovations and an increasing focus on personalized medicine. As healthcare systems globally adopt artificial intelligence and machine learning for diagnostic purposes, the accuracy and efficiency of liver cancer detection will improve significantly. Furthermore, the integration of telemedicine is expected to enhance access to diagnostic services, particularly in remote areas, thereby facilitating early detection and treatment, ultimately improving patient outcomes.
Market Opportunities
- Emerging Markets for Expansion:Emerging economies, particularly in Asia and Africa, present significant opportunities for growth in liver cancer diagnostics. With rising disposable incomes and increasing healthcare investments, these regions are expected to see a surge in demand for advanced diagnostic technologies. The World Bank projects that healthcare expenditure in these regions will grow by 10% annually, creating a favorable environment for market players to expand their operations.
- Development of Non-Invasive Diagnostic Methods:There is a growing opportunity in the development of non-invasive diagnostic methods, which are gaining traction due to their patient-friendly nature. Innovations such as liquid biopsies and advanced imaging techniques are expected to revolutionize liver cancer diagnostics. The global market for non-invasive diagnostic tests is projected to reach $12 billion by the future, driven by increasing demand for safer and more efficient diagnostic options.