Region:North America
Author(s):Shubham
Product Code:KRAC8957
Pages:93
Published On:November 2025
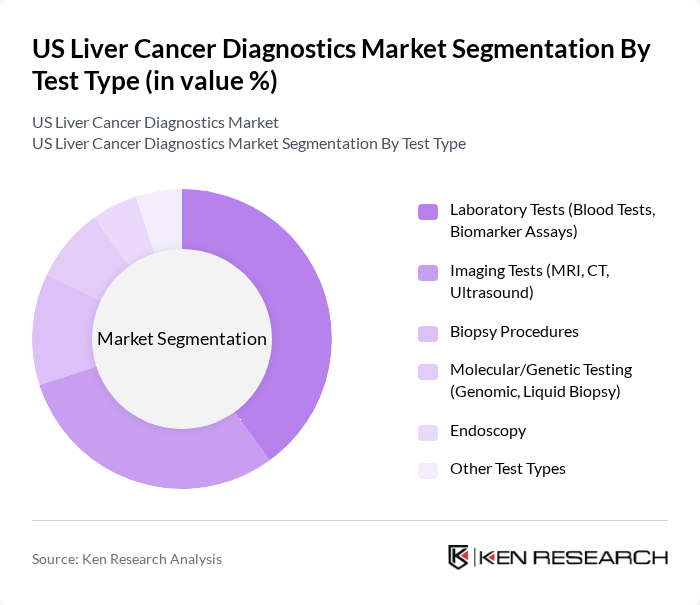
By Test Type:The market is segmented into various test types, including Laboratory Tests (Blood Tests, Biomarker Assays), Imaging Tests (MRI, CT, Ultrasound), Biopsy Procedures, Molecular/Genetic Testing (Genomic, Liquid Biopsy), Endoscopy, and Other Test Types. Among these, Laboratory Tests currently hold the largest market share due to their accuracy, cost-effectiveness, and critical role in disease staging and treatment monitoring. Imaging Tests remain essential for non-invasive diagnosis and treatment planning, while molecular/genetic testing and liquid biopsy are rapidly gaining traction due to technological advancements and FDA-backed innovations.
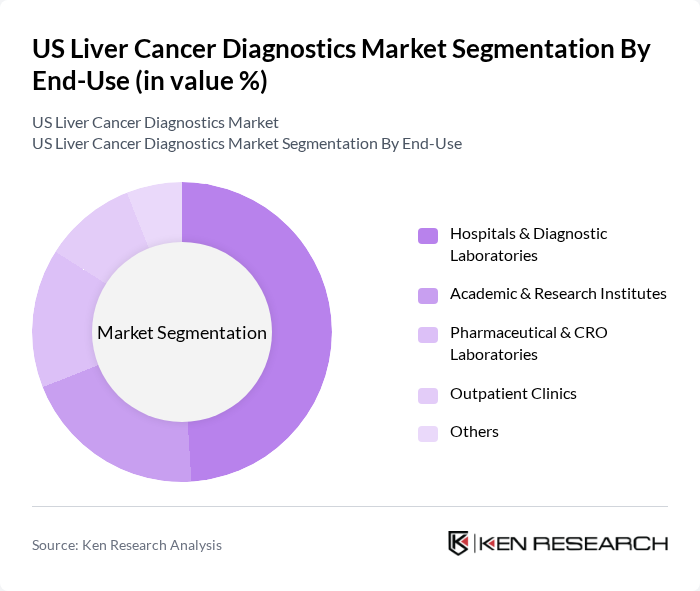
By End-Use:The market is categorized by end-use into Hospitals & Diagnostic Laboratories, Academic & Research Institutes, Pharmaceutical & CRO Laboratories, Outpatient Clinics, and Others. Hospitals & Diagnostic Laboratories dominate this segment, accounting for nearly half of the market share, due to their comprehensive diagnostic capabilities, access to advanced imaging and laboratory infrastructure, and central role in liver cancer management. Academic and research institutes are significant contributors, particularly in biomarker discovery and clinical research, while pharmaceutical and CRO laboratories drive demand through clinical trials and drug development.
The US Liver Cancer Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Abbott Laboratories, Roche Diagnostics, Thermo Fisher Scientific, Bio-Rad Laboratories, Hologic, Inc., Illumina, Inc., Quest Diagnostics, Laboratory Corporation of America Holdings (LabCorp), Guardant Health, Inc., Exact Sciences Corporation, Myriad Genetics, Inc., Agilent Technologies, Inc., PerkinElmer, Inc., Grifols S.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the US liver cancer diagnostics market appears promising, driven by ongoing innovations and a focus on patient-centered care. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the expansion of telemedicine services will improve access to specialists. As healthcare systems increasingly prioritize preventive measures, the demand for early detection and personalized treatment options will likely grow, shaping the market landscape in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Laboratory Tests (Blood Tests, Biomarker Assays) Imaging Tests (MRI, CT, Ultrasound) Biopsy Procedures Molecular/Genetic Testing (Genomic, Liquid Biopsy) Endoscopy Other Test Types |
| By End-Use | Hospitals & Diagnostic Laboratories Academic & Research Institutes Pharmaceutical & CRO Laboratories Outpatient Clinics Others |
| By Diagnostic Method | Non-invasive Methods (Imaging, Blood-based) Invasive Methods (Biopsy, Endoscopy) Biomarker-based Tests Imaging Techniques Molecular Diagnostics Others |
| By Stage of Cancer | Early Stage Intermediate Stage Advanced Stage Others |
| By Region | Northeast Midwest South West |
| By Patient Demographics | Age Group Gender Ethnicity Others |
| By Technology Adoption | Traditional Methods Advanced Technologies (AI, Liquid Biopsy, Genomics) Emerging Technologies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists Specializing in Liver Cancer | 60 | Medical Oncologists, Surgical Oncologists |
| Laboratory Managers in Diagnostic Facilities | 50 | Laboratory Directors, Quality Control Managers |
| Healthcare Payers and Insurers | 40 | Claims Analysts, Medical Directors |
| Patients Diagnosed with Liver Cancer | 45 | Patients, Caregivers |
| Healthcare Policy Experts | 40 | Health Economists, Policy Advisors |
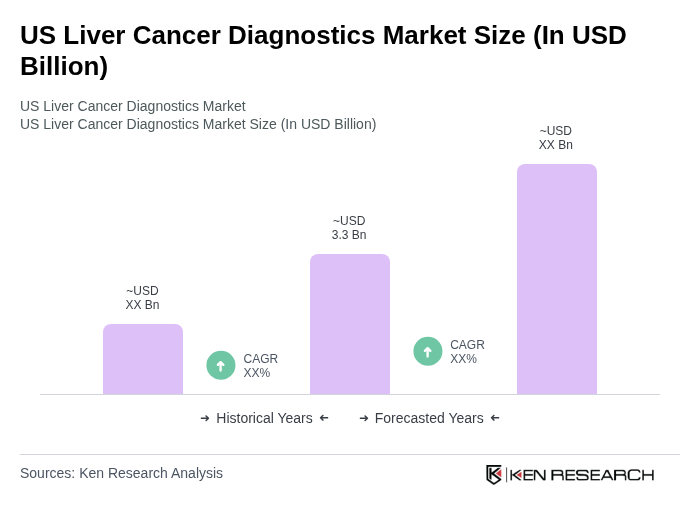
The US Liver Cancer Diagnostics Market is valued at approximately USD 3.3 billion, reflecting a significant growth driven by the increasing prevalence of liver cancer and advancements in diagnostic technologies.