Region:Global
Author(s):Rebecca
Product Code:KRAD2764
Pages:89
Published On:November 2025
By Type:The market is segmented into various types of guidewires, including Hydrophilic Guidewires, Non-Hydrophilic Guidewires, Coated Guidewires, PTFE-Coated Guidewires, Nitinol Guidewires, Stainless Steel Guidewires, and Others. Among these, Hydrophilic Guidewires are gaining significant traction due to their enhanced flexibility and lubricity, which facilitate smoother navigation through the urinary tract. The increasing preference for minimally invasive procedures is driving the demand for these guidewires, making them a leading choice among healthcare professionals.

By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Academic & Research Institutes, and Others. Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is attributed to the high volume of urological procedures performed in hospitals, coupled with the availability of advanced medical technologies and skilled healthcare professionals. The increasing number of surgical interventions and the growing focus on patient care in hospital settings further bolster the demand for guidewires.

The Global Urology Guidewires Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Cook Medical LLC, Terumo Corporation, B. Braun Melsungen AG, Olympus Corporation, Teleflex Incorporated, Medtronic plc, Stryker Corporation, ConMed Corporation, Merit Medical Systems, Inc., Hologic, Inc., UroGen Pharma Ltd., Integer Holdings Corporation, BD (Becton, Dickinson and Company), Advin Health Care, Med pro Medical B.V., ACE Medical Devices contribute to innovation, geographic expansion, and service delivery in this space.
The future of the urology guidewires market appears promising, driven by technological advancements and an increasing focus on patient-centered care. As healthcare systems evolve, the integration of digital technologies and telemedicine is expected to enhance patient access to urological services. Furthermore, the ongoing research and development in innovative guidewire materials will likely lead to improved performance and safety, addressing current market challenges and expanding treatment options for patients worldwide.
| Segment | Sub-Segments |
|---|---|
| By Type | Hydrophilic Guidewires Non-Hydrophilic Guidewires Coated Guidewires PTFE-Coated Guidewires Nitinol Guidewires Stainless Steel Guidewires Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Academic & Research Institutes Others |
| By Application | Ureteral Procedures Cystoscopy Nephrostomy Urethral Stricture Management Stone Management Others |
| By Material | Stainless Steel Nitinol Polymer PTFE Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Urology Clinics | 60 | Urologists, Clinic Managers |
| Hospitals with Urology Departments | 50 | Surgeons, Procurement Officers |
| Medical Device Distributors | 40 | Sales Representatives, Distribution Managers |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
| Research Institutions | 40 | Clinical Researchers, Academic Professors |
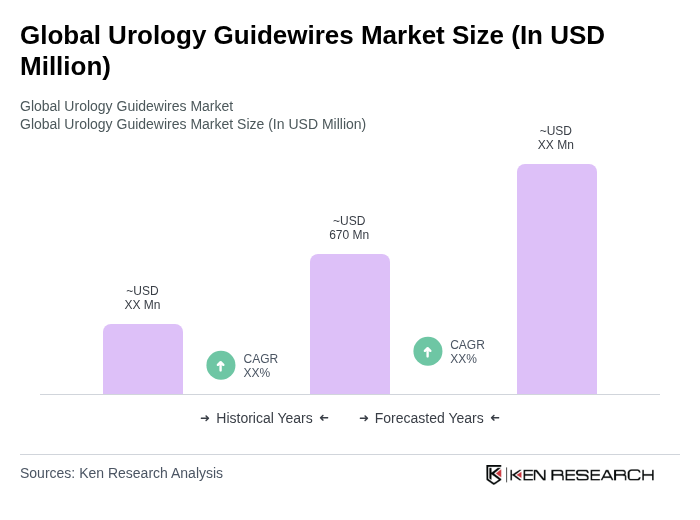
The Global Urology Guidewires Market is valued at approximately USD 670 million, driven by the increasing prevalence of urological disorders, advancements in medical technology, and a rise in minimally invasive surgeries.