Region:Global
Author(s):Shubham
Product Code:KRAB0684
Pages:90
Published On:August 2025
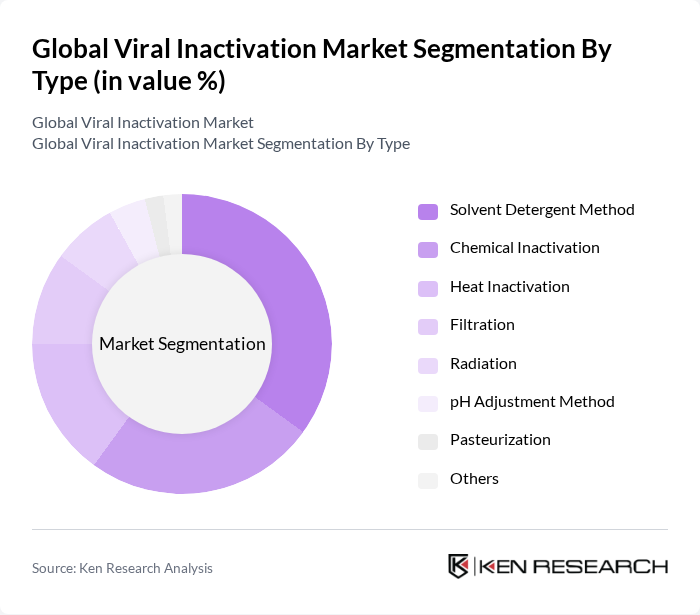
By Type:The viral inactivation market is segmented by method, including Chemical Inactivation, Heat Inactivation, Filtration, Radiation, Solvent Detergent Method, pH Adjustment Method, Pasteurization, and Others. Among these, the Solvent/Detergent Method is the leading sub-segment in terms of revenue due to its established efficacy in plasma-derived products and blood safety applications. Chemical Inactivation and Heat Inactivation are also widely used, with chemical agents favored for their reliability in vaccine and biologics manufacturing, and heat inactivation commonly applied in blood product processing to maintain product integrity. Filtration and advanced irradiation methods are gaining traction as manufacturers seek scalable and efficient viral clearance solutions .
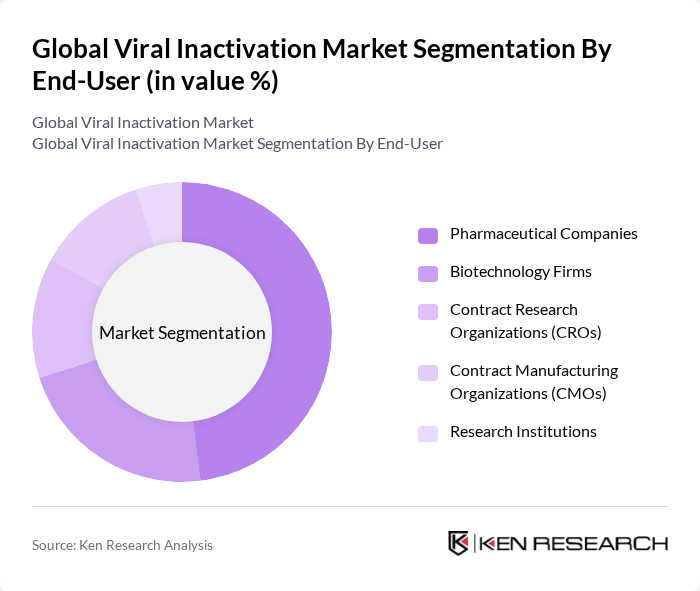
By End-User:The market is segmented by end-users, including Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), and Research Institutions. Pharmaceutical Companies represent the largest end-user segment, driven by their extensive use of viral inactivation in drug development and manufacturing. Biotechnology Firms and CROs are also significant contributors, reflecting the growing trend of outsourcing and the increasing complexity of biologics pipelines. CMOs and Research Institutions play crucial roles in supporting innovation and compliance with evolving regulatory requirements .
The Global Viral Inactivation Market is characterized by a dynamic mix of regional and international players. Leading participants such as Merck KGaA, Pall Corporation, Sartorius AG, Cytiva (formerly GE Healthcare Life Sciences), Thermo Fisher Scientific Inc., Bio-Rad Laboratories, Inc., FUJIFILM Diosynth Biotechnologies, Charles River Laboratories International, Inc., Lonza Group AG, Becton, Dickinson and Company, Octapharma AG, Grifols S.A., Takeda Pharmaceutical Company Limited, Amgen Inc., Regeneron Pharmaceuticals, Inc., Parker Hannifin Corporation, Rad Source Technologies, Clean Cells, WuXi AppTec (formerly WuXi PharmaTech), and Danaher Corporation contribute to innovation, geographic expansion, and service delivery in this space .
The future of the viral inactivation market in None appears promising, driven by ongoing technological advancements and increasing regulatory scrutiny. As biopharmaceutical companies prioritize safety and efficacy, the demand for innovative viral inactivation solutions is expected to rise. Additionally, the growing focus on personalized medicine will likely create new avenues for viral inactivation technologies, ensuring that products meet stringent safety standards while catering to diverse patient needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Chemical Inactivation Heat Inactivation Filtration Radiation Solvent Detergent Method pH Adjustment Method Pasteurization Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Research Institutions |
| By Application | Blood and Blood Products Vaccines and Therapeutics Cell Therapy Gene Therapy Tissue and Tissue Products Others |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology | Ultrafiltration Nanofiltration Chromatography UV Irradiation Others |
| By Regulatory Compliance | ISO Standards cGMP Compliance FDA Approval EMA Approval |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical Manufacturers | 120 | Production Managers, Quality Assurance Officers |
| Healthcare Providers | 90 | Infection Control Specialists, Hospital Administrators |
| Regulatory Bodies | 60 | Regulatory Affairs Managers, Compliance Officers |
| Research Institutions | 50 | Virologists, Research Scientists |
| Vaccine Manufacturers | 70 | Product Development Managers, Safety Officers |
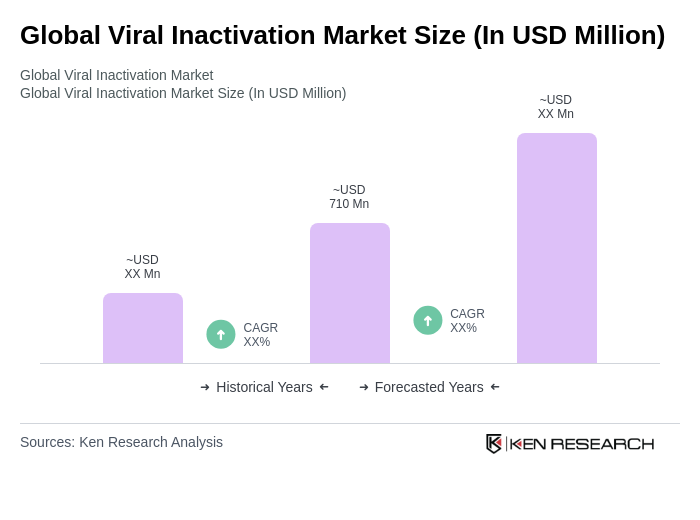
The Global Viral Inactivation Market is valued at approximately USD 710 million, driven by the increasing demand for viral safety in biopharmaceuticals, the rising prevalence of infectious diseases, and the expansion of the biologics sector.