Region:Middle East
Author(s):Rebecca
Product Code:KRAD2418
Pages:85
Published On:January 2026
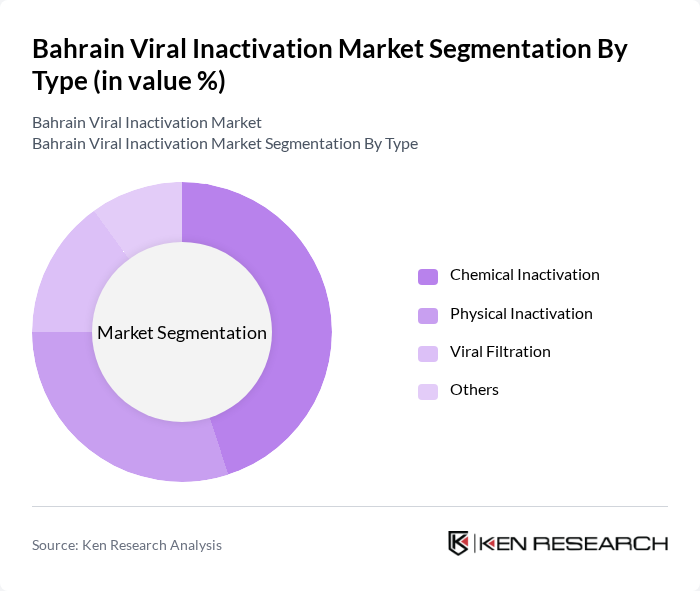
By Type:The market is segmented into various types of viral inactivation methods, including Chemical Inactivation, Physical Inactivation, Biological Inactivation, and Others. Each method has its unique applications and effectiveness in ensuring the safety of biological products. Chemical inactivation is widely used due to its efficiency in solvent-detergent processes, while physical methods are gaining traction for their non-toxic nature through techniques like nanofiltration and heat treatment. Biological inactivation is also emerging as a preferred choice in specific applications for cell and gene therapies.
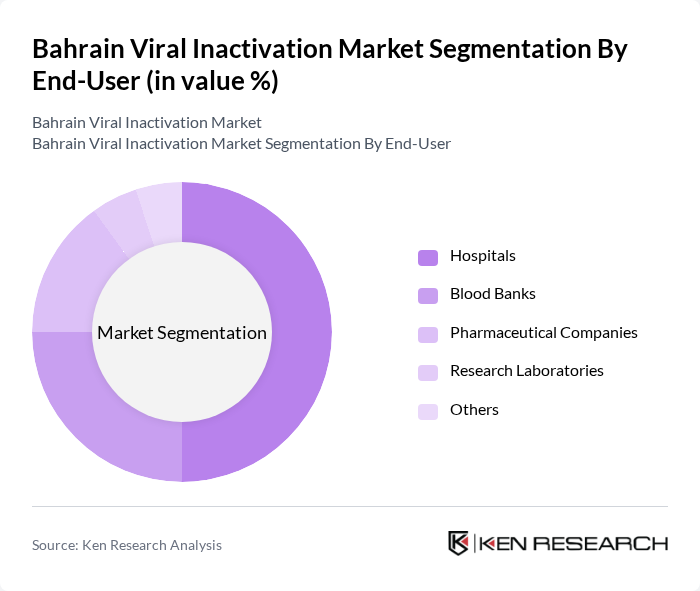
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, Hospitals and Clinics, and Others. Pharmaceutical companies are the dominant end-users due to their extensive need for viral inactivation in drug production. Biotechnology firms are also significant players, focusing on innovative therapies that require stringent safety measures. Research institutions contribute to the market by developing new technologies and methods.
The Bahrain Viral Inactivation Market is characterized by a dynamic mix of regional and international players. Leading participants such as BioReliance, Merck KGaA, Pall Corporation, Sartorius AG, GE Healthcare, Thermo Fisher Scientific, Fujifilm Diosynth Biotechnologies, Charles River Laboratories, Lonza Group, WuXi AppTec, Catalent, Inc., BBI Solutions, Viropro, Inc., Inovio Pharmaceuticals, Avid Bioservices contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain viral inactivation market appears promising, driven by increasing investments in healthcare infrastructure and a growing emphasis on biopharmaceutical safety. As the government continues to support innovation and research in healthcare technologies, the market is likely to see a surge in the adoption of automated and cost-effective viral inactivation solutions. Additionally, the expansion of biopharmaceutical manufacturing capabilities will further enhance the demand for advanced viral inactivation methods, ensuring a safer healthcare environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Chemical Inactivation Physical Inactivation Biological Inactivation Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Hospitals and Clinics Others |
| By Application | Blood and Blood Products Vaccines Cell and Gene Therapies Others |
| By Technology | Heat Treatment UV Light Treatment Filtration Others |
| By Industry | Healthcare Food and Beverage Environmental Others |
| By Region | Central Bahrain Northern Governorate Southern Governorate Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing | 45 | Production Managers, Quality Control Analysts |
| Biotechnology Research Facilities | 40 | Research Scientists, Lab Directors |
| Blood Bank Operations | 40 | Blood Bank Managers, Medical Technologists |
| Vaccine Development Programs | 45 | Clinical Researchers, Regulatory Affairs Specialists |
| Healthcare Institutions | 50 | Infection Control Officers, Hospital Administrators |
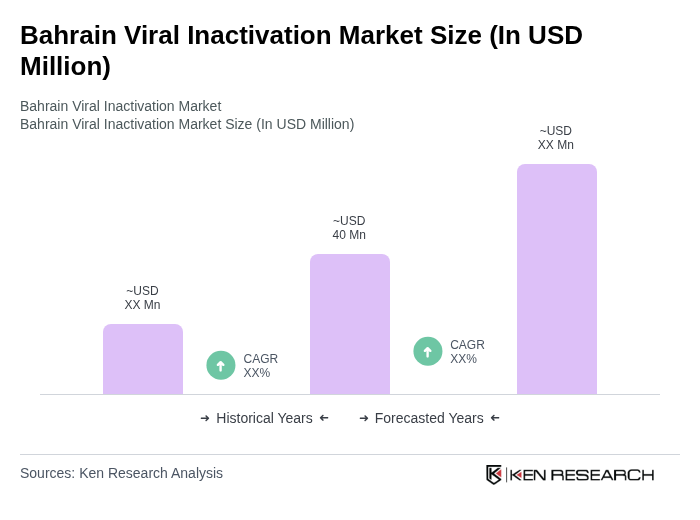
The Bahrain Viral Inactivation Market is valued at approximately USD 42 million, reflecting a five-year historical analysis. This growth is driven by the increasing demand for safe blood products and the rising prevalence of viral infections.