Region:Asia
Author(s):Dev
Product Code:KRAD1822
Pages:82
Published On:November 2025
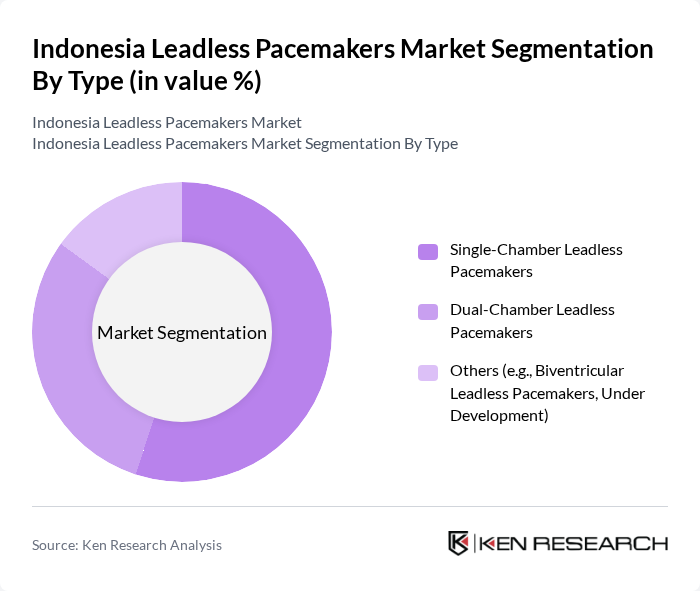
By Type:The market is segmented into three main types of leadless pacemakers: Single-Chamber Leadless Pacemakers, Dual-Chamber Leadless Pacemakers, and Others (including Biventricular Leadless Pacemakers and those under development). Among these, Single-Chamber Leadless Pacemakers are currently leading the market due to their procedural simplicity, proven effectiveness in bradyarrhythmia management, and lower cost compared to dual-chamber options. The growing preference for minimally invasive procedures and the increasing awareness of the benefits of leadless technology are driving the adoption of this sub-segment .
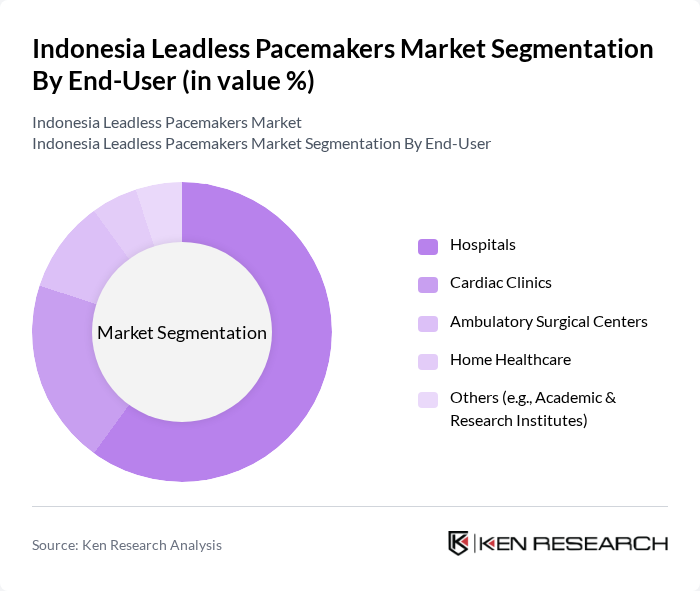
By End-User:The leadless pacemakers market is segmented by end-user into Hospitals, Cardiac Clinics, Ambulatory Surgical Centers, Home Healthcare, and Others (including Academic & Research Institutes). Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is primarily due to the availability of advanced medical facilities, skilled healthcare professionals, and the capacity to perform complex cardiac procedures. The trend towards outpatient care and the increasing number of cardiac surgeries are also contributing to the growth of this segment .
The Indonesia Leadless Pacemakers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Abbott Laboratories, Boston Scientific, Biotronik, MicroPort Scientific Corporation, Lepu Medical Technology, EBR Systems, Osypka Medical, LivaNova, ZOLL Medical, Shree Pacetronix, Vitatron (Medtronic Subsidiary), Sorin Group (now part of LivaNova), Cardiac Science (now part of ZOLL Medical), St. Jude Medical (now part of Abbott Laboratories) contribute to innovation, geographic expansion, and service delivery in this space .
The future of the leadless pacemaker market in Indonesia appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the government prioritizes healthcare infrastructure improvements, the accessibility of innovative cardiac solutions is expected to rise. Additionally, the integration of telemedicine and remote monitoring technologies will likely enhance patient management and follow-up care. These trends indicate a shift towards more patient-centric approaches, fostering a conducive environment for the growth of leadless pacemakers in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Single-Chamber Leadless Pacemakers Dual-Chamber Leadless Pacemakers Others (e.g., Biventricular Leadless Pacemakers, Under Development) |
| By End-User | Hospitals Cardiac Clinics Ambulatory Surgical Centers Home Healthcare Others (e.g., Academic & Research Institutes) |
| By Age Group | Pediatric Patients Adult Patients Geriatric Patients |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Others (e.g., Papua, Nusa Tenggara) |
| By Clinical Application | Arrhythmia Management Bradycardia Treatment Heart Failure Treatment Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists in Major Hospitals | 60 | Interventional Cardiologists, Electrophysiologists |
| Healthcare Procurement Managers | 50 | Hospital Procurement Officers, Supply Chain Managers |
| Medical Device Distributors | 40 | Sales Representatives, Distribution Managers |
| Patients with Pacemakers | 45 | Cardiac Patients, Patient Advocacy Group Members |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Regulatory Agency Representatives |
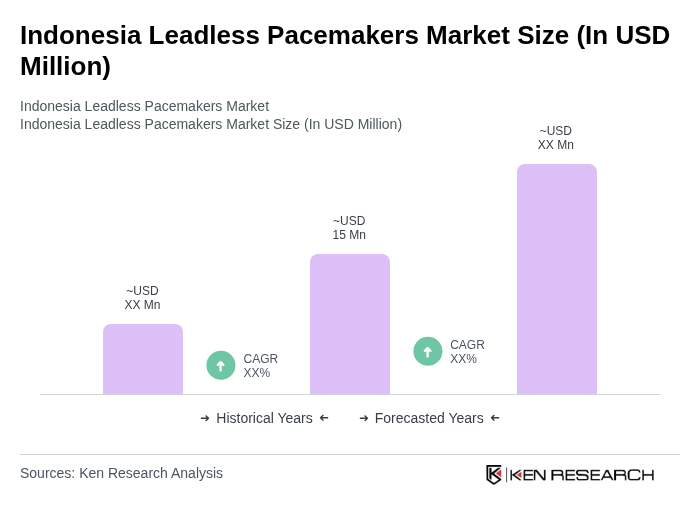
The Indonesia Leadless Pacemakers Market is valued at approximately USD 15 million, driven by the rising prevalence of cardiovascular diseases and advancements in minimally invasive cardiac device technology.