Region:Asia
Author(s):Rebecca
Product Code:KRAC9655
Pages:95
Published On:November 2025
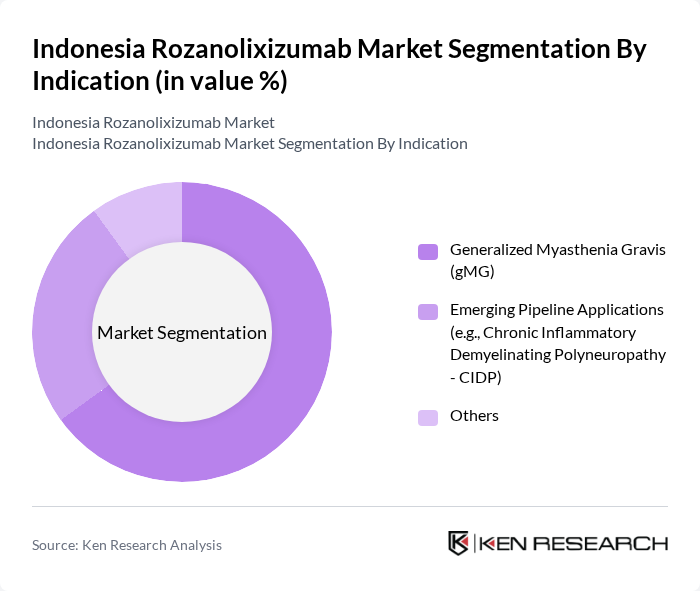
By Indication:The market is segmented into three main indications: Generalized Myasthenia Gravis (gMG), Emerging Pipeline Applications (such as Chronic Inflammatory Demyelinating Polyneuropathy - CIDP), and Others. Generalized Myasthenia Gravis remains the leading indication, reflecting its relatively higher prevalence and significant unmet need for targeted immunotherapies. Increased disease awareness, improved diagnostic rates, and the introduction of advanced biologics have driven a surge in demand for therapies like Rozanolixizumab among gMG patients .
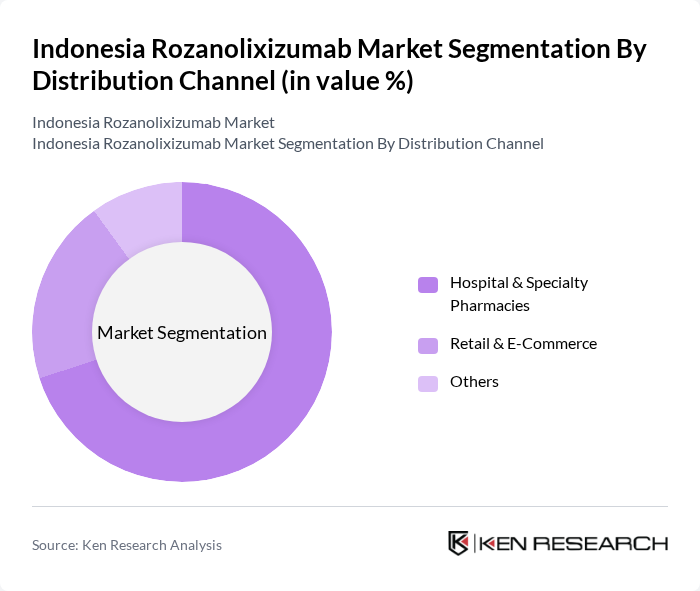
By Distribution Channel:The distribution channels for Rozanolixizumab include Hospital & Specialty Pharmacies, Retail & E-Commerce, and Others. Hospital & Specialty Pharmacies dominate the market, owing to their essential role in dispensing complex biologics and specialty medicines for chronic and rare diseases. The presence of over 32,000 pharmacies in Indonesia, with a concentration of advanced hospital and specialty outlets in major cities, underpins this trend. Distribution efficiency is further enhanced by digital logistics platforms and specialized wholesale partnerships, which ensure timely access to high-value therapies .
The Indonesia Rozanolixizumab Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Indofarma Tbk, PT Merck Tbk, PT Sanofi Indonesia, PT Pfizer Indonesia, PT Novartis Indonesia, PT Roche Indonesia, PT AstraZeneca Indonesia, PT GlaxoSmithKline Indonesia, PT Johnson & Johnson Indonesia, PT AbbVie Indonesia, PT Amgen Indonesia, PT Takeda Indonesia, and PT UCB Pharma Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Rozanolixizumab market in Indonesia appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the government continues to enhance healthcare infrastructure, more patients will gain access to advanced therapies. Additionally, the integration of digital health solutions is expected to facilitate better patient management and adherence to treatment protocols, further supporting the market's growth trajectory in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Indication | Generalized Myasthenia Gravis (gMG) Emerging Pipeline Applications (e.g., Chronic Inflammatory Demyelinating Polyneuropathy - CIDP) Others |
| By Distribution Channel | Hospital & Specialty Pharmacies Retail & E-Commerce Others |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender Socioeconomic Status Others |
| By Therapeutic Area | Neurology Rheumatology Others |
| By Region | Java Sumatra Kalimantan Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals | 100 | Rheumatologists, Immunologists |
| Pharmaceutical Distributors | 60 | Sales Managers, Product Managers |
| Patient Advocacy Groups | 40 | Group Leaders, Patient Representatives |
| Hospital Procurement Departments | 50 | Procurement Officers, Supply Chain Managers |
| Clinical Trial Coordinators | 40 | Research Nurses, Clinical Research Associates |
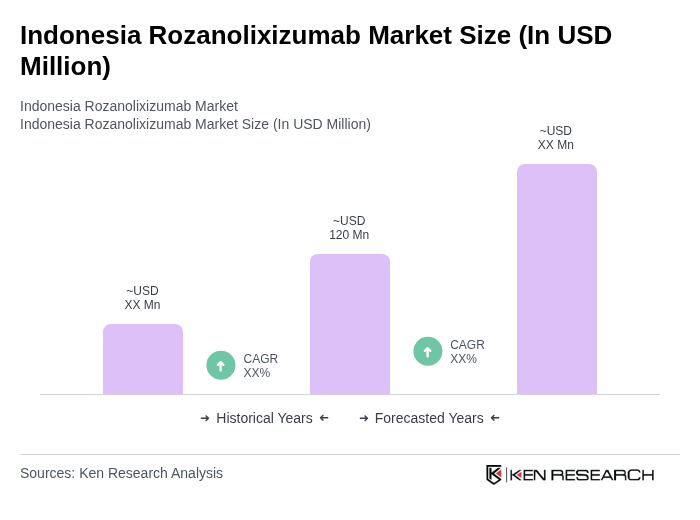
The Indonesia Rozanolixizumab market is valued at approximately USD 120 million, reflecting significant growth driven by the increasing prevalence of autoimmune diseases and the demand for advanced biologic therapies in the country.