About the Report
Base Year 2024Indonesia Unit Dose Manufacturing Market Overview
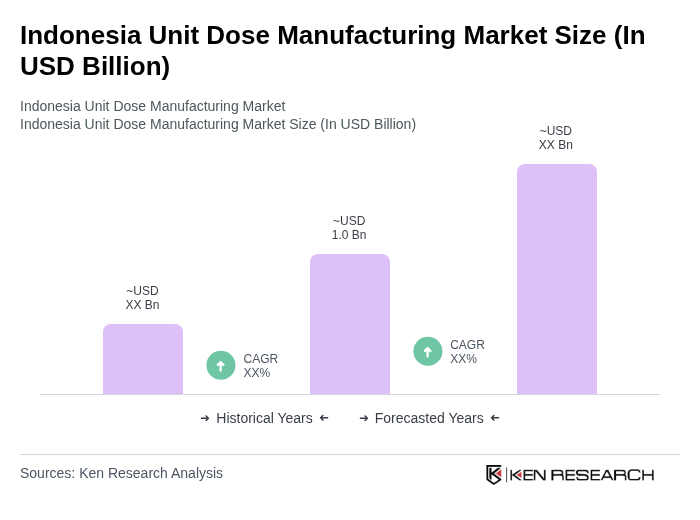
- The Indonesia Unit Dose Manufacturing Market is valued at USD 1.0 billion, based on a five-year historical analysis and triangulation with the size of the Indonesia generic drug market and the global unit dose manufacturing and packaging space. This growth is primarily driven by the increasing demand for precise medication dosing, advancements in pharmaceutical manufacturing and packaging technologies, the expansion of the domestic generic medicines market, and a growing emphasis on patient safety and compliance in hospitals and pharmacies. The rise in chronic diseases such as diabetes and cardiovascular conditions, along with the need for effective long-term medication management and adherence programs, has further fueled the market's expansion.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their robust healthcare infrastructure, concentration of pharmaceutical and packaging companies, and access to a large patient population. Jakarta, being the capital, serves as a hub for healthcare innovation, pharmaceutical headquarters, and investment, while Surabaya and Bandung contribute significantly through their local manufacturing capabilities, distribution networks, and healthcare services.
- In 2023, the Indonesian government strengthened enforcement of Good Manufacturing Practices (GMP) requirements for all pharmaceutical manufacturers through the implementation of the Indonesian Good Manufacturing Practice for Pharmaceuticals standard CPOB (Cara Pembuatan Obat yang Baik) as regulated by the Indonesian Food and Drug Authority (Badan POM) under the Regulation of the Head of BPOM on Good Manufacturing Practices for Pharmaceuticals, which mandates that manufacturers comply with GMP to ensure the quality and safety of medicines, including unit dose presentations. This framework aims to enhance the overall standards of pharmaceutical production and protect public health by ensuring that all products meet stringent quality, documentation, and validation criteria across solid, liquid, semi-solid, and parenteral dosage forms.
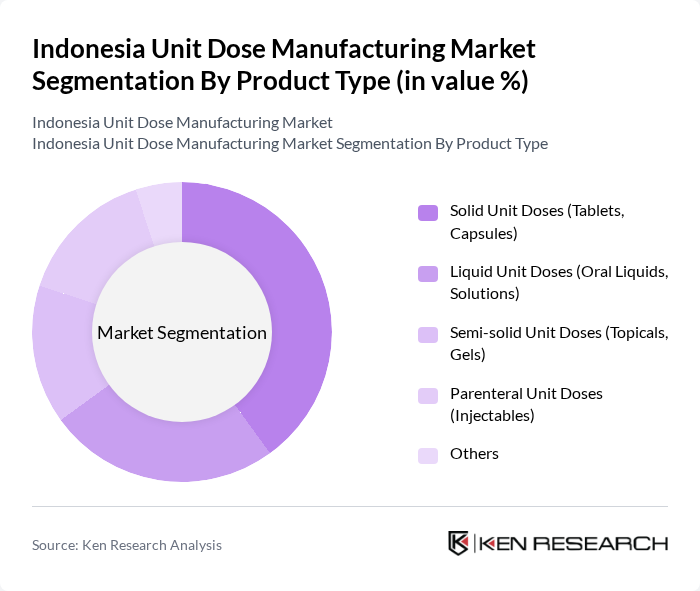
Indonesia Unit Dose Manufacturing Market Segmentation
By Product Type:The product type segmentation includes various forms of unit doses, each catering to specific patient needs and preferences. Solid unit doses, such as tablets and capsules, are widely used due to their convenience, stability, ease of large-scale manufacture, and strong presence of generic oral formulations in Indonesia. Liquid unit doses, including oral liquids and solutions, are preferred for pediatric and geriatric patients who may have difficulty swallowing solids and are increasingly used for chronic therapies and hospital dispensing because of dosing accuracy and reduced medication errors. Semi-solid unit doses, like topicals and gels, are utilized for localized treatment and are supported by the broader growth in dermatology and pain-relief formulations, while parenteral unit doses, such as injectables, are critical for immediate therapeutic effects, particularly in hospital settings and for biologics and sterile injectables. The "Others" category encompasses various innovative delivery systems that are emerging in the market, including unit-dose sachets, ophthalmic single-use vials, and advanced packaging formats aligned with global unit dose packaging trends toward convenience and safety.
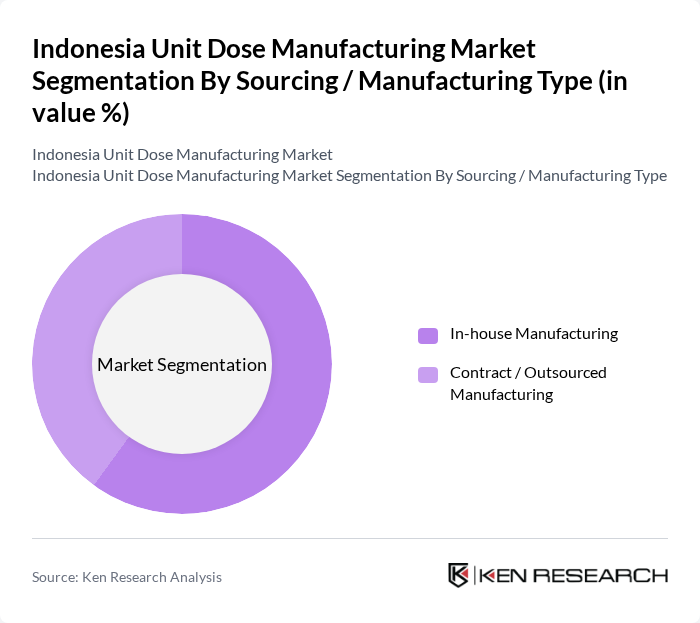
By Sourcing / Manufacturing Type:The sourcing and manufacturing type segmentation reflects the operational strategies adopted by companies in the unit dose manufacturing market. In-house manufacturing allows companies to maintain control over production quality, regulatory compliance, and processes, which is a common model among leading Indonesian pharmaceutical manufacturers that operate integrated production plants. Contract or outsourced manufacturing provides flexibility and cost efficiency, and its adoption is supported by the broader growth of contract development and manufacturing and pharmaceutical packaging services in Asia-Pacific, including sterile injectables CDMOs in Indonesia. The trend towards outsourcing is growing as companies seek to leverage specialized expertise, advanced packaging technologies, and automation while reducing capital expenditure and operational costs, leading to a more competitive landscape and a wider base of regional CDMOs and packaging partners.
Indonesia Unit Dose Manufacturing Market Competitive Landscape
The Indonesia Unit Dose Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Indofarma Tbk, PT Phapros Tbk, PT Sanbe Farma, PT Dexa Medica, PT Ferron Par Pharmaceuticals, PT Aventis Pharma (Sanofi Indonesia), PT Pfizer Indonesia, PT Novartis Indonesia, PT AstraZeneca Indonesia, PT Bayer Indonesia, PT GlaxoSmithKline Indonesia, PT Boehringer Ingelheim Indonesia, and selected regional CDMOs & packaging partners contribute to innovation, geographic expansion, and service delivery in this space through investments in GMP-compliant facilities, expansion of generic and branded portfolios, and adoption of modern unit dose packaging technologies aligned with global best practices.
Indonesia Unit Dose Manufacturing Market Industry Analysis
Growth Drivers
- Increasing Demand for Patient-Centric Solutions:The Indonesian healthcare sector is witnessing a significant shift towards patient-centric solutions, driven by a growing population of over 270 million. In future, healthcare expenditure is projected to reach approximately IDR 1,193 trillion (USD 84 billion), reflecting a 9% increase from the previous period. This trend is fostering the development of unit dose systems that enhance medication adherence and safety, catering to the needs of patients and healthcare providers alike.
- Rising Healthcare Expenditure:Indonesia's healthcare expenditure is on an upward trajectory, with the government allocating around IDR 193 trillion (USD 13.5 billion) for health in the state budget in future, which corresponds to about 9–10 percent of total government expenditure. This rise in spending is facilitating investments in advanced manufacturing technologies, including unit dose systems, which are essential for improving drug delivery and patient outcomes in a rapidly evolving healthcare landscape.
- Technological Advancements in Drug Delivery:The Indonesian unit dose manufacturing market is benefiting from rapid technological advancements, particularly in drug delivery systems. In future, the adoption of innovative technologies, such as smart packaging and automated dispensing systems, is expected to increase by 15%, driven by the need for enhanced efficiency and accuracy in medication administration, ultimately improving patient safety and treatment efficacy across healthcare facilities.
Market Challenges
- Regulatory Compliance Complexity:Navigating the regulatory landscape in Indonesia poses significant challenges for unit dose manufacturers. The country has stringent regulations, with over 200 compliance requirements for drug manufacturing. In future, the cost of compliance is estimated to exceed IDR 50 billion (USD 3.5 million) for mid-sized manufacturers, creating barriers to entry and increasing operational costs, which can hinder market growth.
- High Initial Investment Costs:The initial investment required for establishing unit dose manufacturing facilities in Indonesia is substantial, often exceeding IDR 100 billion (USD 7 million). This high capital requirement can deter new entrants and limit the expansion of existing players. In future, the financial burden associated with advanced manufacturing technologies and compliance with Good Manufacturing Practices (GMP) will continue to challenge market participants, impacting overall growth.
Indonesia Unit Dose Manufacturing Market Future Outlook
The future of the Indonesia unit dose manufacturing market appears promising, driven by increasing healthcare investments and a growing emphasis on patient safety. As the government continues to enhance healthcare access, the demand for innovative drug delivery systems is expected to rise. Additionally, the integration of digital health solutions and personalized medicine will likely reshape the market landscape, fostering collaboration among manufacturers and healthcare providers to improve patient outcomes and operational efficiency.
Market Opportunities
- Expansion of E-Pharmacies:The rise of e-pharmacies in Indonesia presents a significant opportunity for unit dose manufacturers. With online pharmacy sales projected to reach IDR 20 trillion (USD 1.4 billion) in future, manufacturers can leverage this trend to enhance distribution channels and improve access to unit dose medications, ultimately increasing market penetration and consumer reach.
- Growth in Geriatric Population:Indonesia's geriatric population is expected to exceed 30 million by future, creating a substantial demand for unit dose systems tailored to elderly patients. This demographic shift presents an opportunity for manufacturers to develop specialized packaging and delivery solutions that cater to the unique needs of older adults, enhancing medication adherence and improving health outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Solid Unit Doses (Tablets, Capsules) Liquid Unit Doses (Oral Liquids, Solutions) Semi-solid Unit Doses (Topicals, Gels) Parenteral Unit Doses (Injectables) Others |
| By Sourcing / Manufacturing Type | In-house Manufacturing Contract / Outsourced Manufacturing |
| By Application / End-User | Public Hospitals (BPJS-affiliated) Private Hospitals & Specialty Clinics Long-term Care & Nursing Facilities Independent & Retail Pharmacies Home Healthcare Providers Others |
| By Packaging Format | Blister Packs Unit-dose Sachets & Pouches Pre-filled Syringes Vials & Ampoules Others |
| By Therapeutic Area | Cardiovascular Anti-infectives Central Nervous System Oncology Diabetes & Metabolic Disorders Others |
| By Region | Java Sumatra Bali & Nusa Tenggara Kalimantan Sulawesi & Eastern Indonesia |
| By Regulatory Compliance Level | BPOM & GMP Compliant Internationally Certified (e.g., WHO, US FDA, EMA) Partially Compliant / In Transition Non-Compliant / Unregulated |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Agency of Drug and Food Control, Ministry of Health)
Manufacturers and Producers
Pharmaceutical Distributors and Wholesalers
Healthcare Providers and Hospitals
Pharmacy Chains and Retail Pharmacies
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Financial Institutions and Banks
Players Mentioned in the Report:
PT Kalbe Farma Tbk
PT Kimia Farma Tbk
PT Indofarma Tbk
PT Phapros Tbk
PT Sanbe Farma
PT Dexa Medica
PT Ferron Par Pharmaceuticals
PT Aventis Pharma (Sanofi Indonesia)
PT Pfizer Indonesia
PT Novartis Indonesia
PT AstraZeneca Indonesia
PT Bayer Indonesia
PT GlaxoSmithKline Indonesia
PT Boehringer Ingelheim Indonesia
Selected Regional CDMOs & Packaging Partners
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Unit Dose Manufacturing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Unit Dose Manufacturing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Unit Dose Manufacturing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Patient-Centric Solutions
3.1.2 Rising Healthcare Expenditure
3.1.3 Technological Advancements in Drug Delivery
3.1.4 Government Initiatives to Enhance Healthcare Access
3.2 Market Challenges
3.2.1 Regulatory Compliance Complexity
3.2.2 High Initial Investment Costs
3.2.3 Limited Awareness Among Healthcare Providers
3.2.4 Supply Chain Disruptions
3.3 Market Opportunities
3.3.1 Expansion of E-Pharmacies
3.3.2 Growth in Geriatric Population
3.3.3 Increasing Focus on Preventive Healthcare
3.3.4 Collaborations with Local Manufacturers
3.4 Market Trends
3.4.1 Shift Towards Personalized Medicine
3.4.2 Integration of Digital Health Solutions
3.4.3 Rise of Biologics and Biosimilars
3.4.4 Emphasis on Sustainability in Packaging
3.5 Government Regulation
3.5.1 Implementation of Good Manufacturing Practices (GMP)
3.5.2 Drug Registration and Approval Processes
3.5.3 Pricing Regulations for Pharmaceuticals
3.5.4 Policies Supporting Local Manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Unit Dose Manufacturing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Unit Dose Manufacturing Market Segmentation
8.1 By Product Type
8.1.1 Solid Unit Doses (Tablets, Capsules)
8.1.2 Liquid Unit Doses (Oral Liquids, Solutions)
8.1.3 Semi-solid Unit Doses (Topicals, Gels)
8.1.4 Parenteral Unit Doses (Injectables)
8.1.5 Others
8.2 By Sourcing / Manufacturing Type
8.2.1 In-house Manufacturing
8.2.2 Contract / Outsourced Manufacturing
8.3 By Application / End-User
8.3.1 Public Hospitals (BPJS-affiliated)
8.3.2 Private Hospitals & Specialty Clinics
8.3.3 Long-term Care & Nursing Facilities
8.3.4 Independent & Retail Pharmacies
8.3.5 Home Healthcare Providers
8.3.6 Others
8.4 By Packaging Format
8.4.1 Blister Packs
8.4.2 Unit-dose Sachets & Pouches
8.4.3 Pre-filled Syringes
8.4.4 Vials & Ampoules
8.4.5 Others
8.5 By Therapeutic Area
8.5.1 Cardiovascular
8.5.2 Anti-infectives
8.5.3 Central Nervous System
8.5.4 Oncology
8.5.5 Diabetes & Metabolic Disorders
8.5.6 Others
8.6 By Region
8.6.1 Java
8.6.2 Sumatra
8.6.3 Bali & Nusa Tenggara
8.6.4 Kalimantan
8.6.5 Sulawesi & Eastern Indonesia
8.7 By Regulatory Compliance Level
8.7.1 BPOM & GMP Compliant
8.7.2 Internationally Certified (e.g., WHO, US FDA, EMA)
8.7.3 Partially Compliant / In Transition
8.7.4 Non-Compliant / Unregulated
9. Indonesia Unit Dose Manufacturing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Indonesia Unit Dose Revenue (Latest Year, USD Million)
9.2.4 3-year Revenue CAGR (%) – Unit Dose Portfolio
9.2.5 EBITDA Margin (%) – Unit Dose Operations
9.2.6 Capacity Utilization Rate (%)
9.2.7 Capex Intensity (% of Sales)
9.2.8 Market Share in Indonesia Unit Dose Manufacturing (%)
9.2.9 Contract Manufacturing vs In-house Revenue Mix (%)
9.2.10 Average Contract Tenure (Years)
9.2.11 On-time Delivery Rate (%)
9.2.12 Regulatory Inspection Success Rate (% Passed Without Major Observations)
9.2.13 R&D Spend (% of Sales) – Unit Dose
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Kalbe Farma Tbk
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Indofarma Tbk
9.5.4 PT Phapros Tbk
9.5.5 PT Sanbe Farma
9.5.6 PT Dexa Medica
9.5.7 PT Ferron Par Pharmaceuticals
9.5.8 PT Aventis Pharma (Sanofi Indonesia)
9.5.9 PT Pfizer Indonesia
9.5.10 PT Novartis Indonesia
9.5.11 PT AstraZeneca Indonesia
9.5.12 PT Bayer Indonesia
9.5.13 PT GlaxoSmithKline Indonesia
9.5.14 PT Boehringer Ingelheim Indonesia
9.5.15 Selected Regional CDMOs & Packaging Partners
10. Indonesia Unit Dose Manufacturing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Industry
10.1.4 Ministry of Trade
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Healthcare Providers
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Case Studies of Successful Implementations
11. Indonesia Unit Dose Manufacturing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from government bodies and trade associations related to unit dose manufacturing
- Review of published market studies and white papers focusing on pharmaceutical and consumer goods sectors in Indonesia
- Examination of regulatory frameworks and guidelines from the Indonesian Food and Drug Authority (BPOM)
Primary Research
- Interviews with key stakeholders in the pharmaceutical industry, including manufacturers and distributors
- Surveys targeting healthcare professionals to understand the demand for unit dose packaging
- Field interviews with regulatory experts to assess compliance challenges in unit dose manufacturing
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including trade publications and expert opinions
- Triangulation of market data with insights from industry conferences and seminars
- Sanity checks conducted through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in Indonesia as a basis for unit dose segment analysis
- Segmentation of the market by therapeutic categories and dosage forms to identify unit dose opportunities
- Incorporation of growth trends in healthcare expenditure and population demographics
Bottom-up Modeling
- Collection of production data from leading unit dose manufacturers to establish volume benchmarks
- Operational cost analysis based on manufacturing processes and packaging technologies
- Calculation of market size based on unit sales and average selling prices of unit dose products
Forecasting & Scenario Analysis
- Development of forecasting models using historical growth rates and market drivers
- Scenario analysis based on potential regulatory changes and shifts in consumer preferences
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 120 | Production Managers, Quality Assurance Heads |
| Healthcare Providers | 90 | Pharmacists, Hospital Administrators |
| Regulatory Bodies | 40 | Compliance Officers, Regulatory Affairs Specialists |
| Packaging Suppliers | 70 | Sales Managers, Product Development Engineers |
| Market Research Analysts | 60 | Industry Analysts, Market Strategists |
Frequently Asked Questions
What is the current value of the Indonesia Unit Dose Manufacturing Market?
The Indonesia Unit Dose Manufacturing Market is valued at approximately USD 1.0 billion, reflecting growth driven by increased demand for precise medication dosing, advancements in manufacturing technologies, and a focus on patient safety and compliance in healthcare settings.