Region:Europe
Author(s):Geetanshi
Product Code:KRAA4789
Pages:97
Published On:September 2025
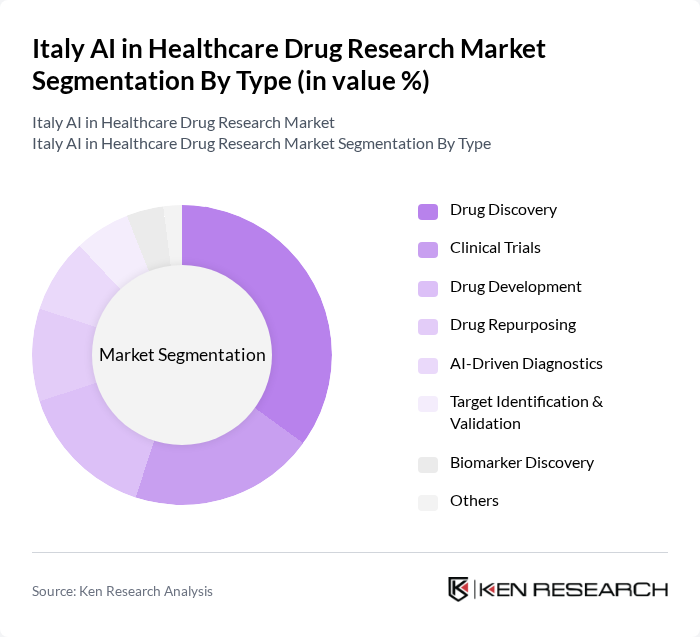
By Type:The market can be segmented into various types, including Drug Discovery, Clinical Trials, Drug Development, Drug Repurposing, AI-Driven Diagnostics, Target Identification & Validation, Biomarker Discovery, and Others. Among these, Drug Discovery is the leading segment, driven by the increasing need for efficient drug development processes and the growing adoption of AI technologies to streamline research methodologies. AI-driven platforms are increasingly used for small molecule identification, virtual screening, and predictive modeling, which accelerates early-stage drug research .
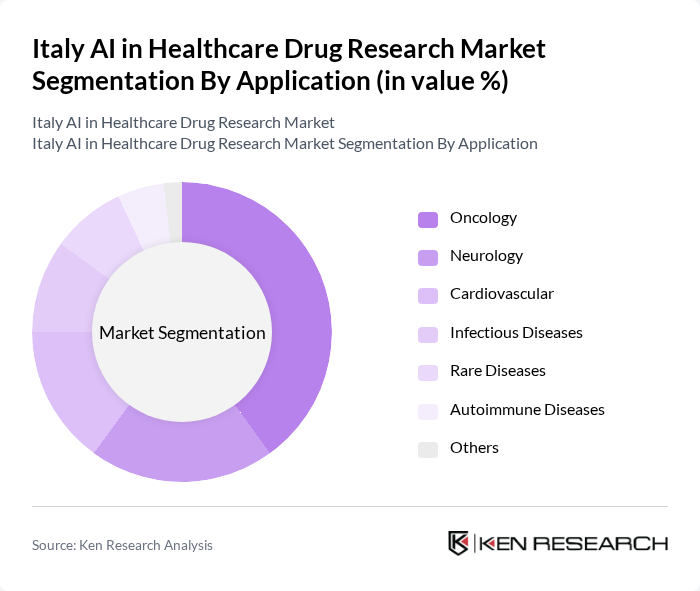
By Application:The applications of AI in healthcare drug research include Oncology, Neurology, Cardiovascular, Infectious Diseases, Rare Diseases, Autoimmune Diseases, and Others. Oncology is the dominant application area, driven by the increasing prevalence of cancer and the need for innovative treatment options. AI technologies are being utilized to analyze vast datasets, identify potential drug candidates, and personalize treatment plans for patients. Immuno-oncology is the fastest-growing sub-segment, reflecting the surge in AI-enabled cancer research and clinical trial optimization .
The Italy AI in Healthcare Drug Research Market is characterized by a dynamic mix of regional and international players. Leading participants such as IBM Watson Health, Siemens Healthineers, Philips Healthcare, Roche, Novartis, AstraZeneca, GlaxoSmithKline, Merck Group, Sanofi, Biogen, Amgen, Johnson & Johnson, Eli Lilly and Company, Bayer AG, Takeda Pharmaceutical Company, Exscientia, BenevolentAI, Insilico Medicine, Evotec SE, Dompé farmaceutici S.p.A., Iktos, Biofourmis, Medidata Solutions (Dassault Systèmes), Microsoft, NVIDIA contribute to innovation, geographic expansion, and service delivery in this space.
The future of the AI in healthcare drug research market in Italy appears promising, driven by technological advancements and increased collaboration among stakeholders. As regulatory frameworks evolve, the integration of AI into clinical trials and drug development processes is expected to accelerate. Furthermore, the focus on real-world evidence generation will enhance the credibility of AI applications, fostering greater acceptance among healthcare professionals and patients alike, ultimately leading to improved healthcare outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Drug Discovery Clinical Trials Drug Development Drug Repurposing AI-Driven Diagnostics Target Identification & Validation Biomarker Discovery Others |
| By Application | Oncology Neurology Cardiovascular Infectious Diseases Rare Diseases Autoimmune Diseases Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Contract Research Organizations (CROs) Healthcare Providers Others |
| By Sales Channel | Direct Sales Distributors Online Platforms Others |
| By Region | Northern Italy Central Italy Southern Italy Islands |
| By Investment Source | Private Investments Government Grants Venture Capital EU Funding Others |
| By Policy Support | Subsidies for AI Research Tax Incentives for R&D Regulatory Support Initiatives Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| AI in Drug Discovery | 60 | Pharmaceutical Researchers, AI Specialists |
| Clinical Trials Optimization | 50 | Clinical Trial Managers, Data Scientists |
| Patient Management Systems | 40 | Healthcare Administrators, IT Managers |
| Regulatory Compliance in AI | 45 | Regulatory Affairs Specialists, Compliance Officers |
| AI Technology Providers | 55 | Product Managers, Business Development Executives |
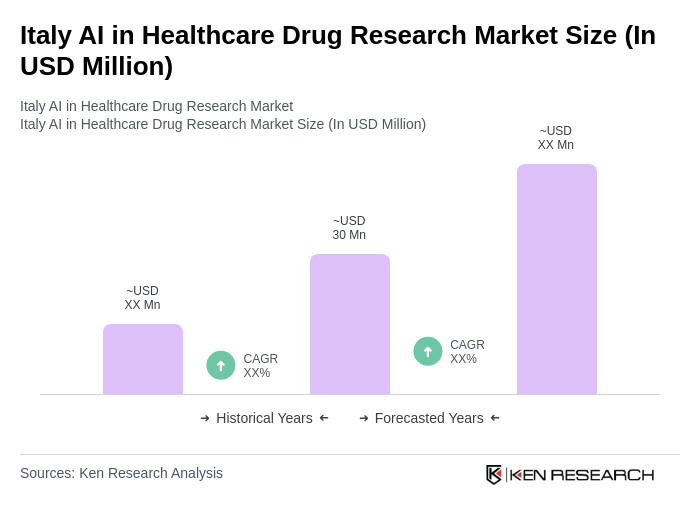
The Italy AI in Healthcare Drug Research Market is valued at approximately USD 30 million, reflecting significant growth driven by advancements in AI technologies, increased investments in healthcare innovation, and the rising demand for personalized medicine.