About the Report
Base Year 2024Italy Medical Devices and Surgical Equipment Market Overview
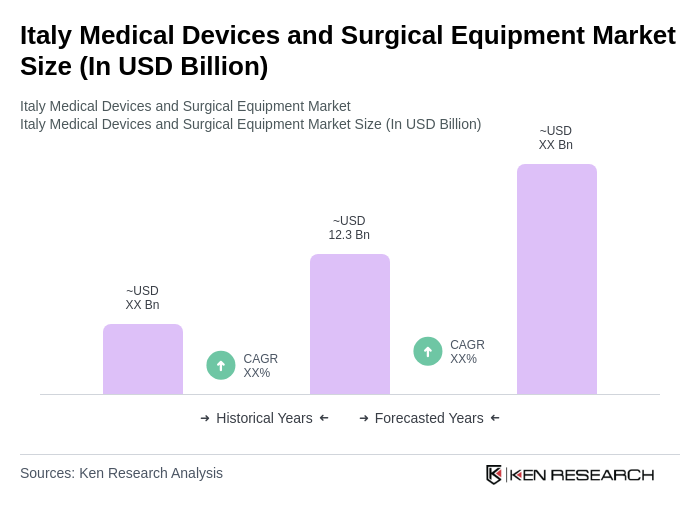
- The Italy Medical Devices and Surgical Equipment Market is valued at USD 12.3 billion, based on a five-year historical analysis. This growth is primarily driven by an aging population, increasing prevalence of chronic diseases, and advancements in medical technology. The demand for innovative medical devices and surgical equipment has surged, reflecting the need for improved healthcare services and patient outcomes .
- Key players in this market include major cities such as Milan, Rome, and Bologna, which dominate due to their robust healthcare infrastructure, presence of leading medical institutions, and a concentration of research and development activities. These cities are also home to numerous medical device manufacturers and suppliers, facilitating a vibrant ecosystem for innovation and collaboration .
- The Medical Device Regulation (EU) 2017/745, implemented in Italy by the Ministry of Health in 2021, mandates stricter compliance and safety standards for medical devices. This regulation requires all medical devices to undergo rigorous conformity assessment, registration in the EUDAMED database, and adherence to post-market surveillance requirements before being marketed, thereby enhancing patient safety and fostering trust in the healthcare system .
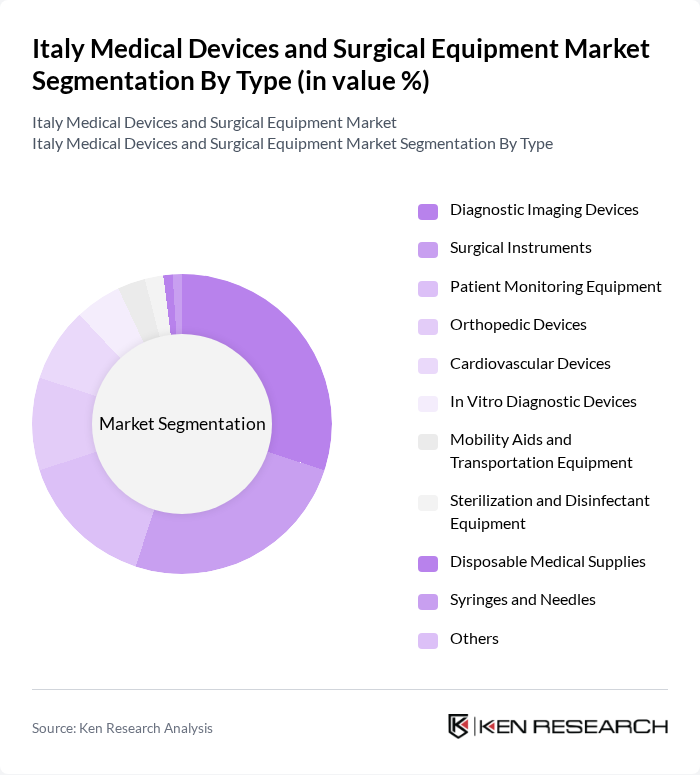
Italy Medical Devices and Surgical Equipment Market Segmentation
By Type:The market is segmented into various types of medical devices and surgical equipment, including diagnostic imaging devices, surgical instruments, patient monitoring equipment, orthopedic devices, cardiovascular devices, in vitro diagnostic devices, mobility aids and transportation equipment, sterilization and disinfectant equipment, disposable medical supplies, syringes and needles, and others. Among these, diagnostic imaging devices and surgical instruments are particularly prominent due to their essential roles in modern healthcare practices .
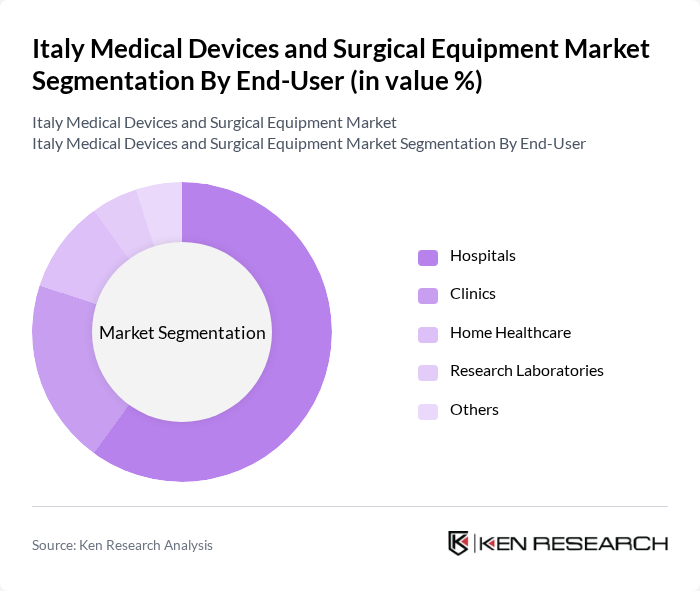
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, research laboratories, and others. Hospitals are the dominant end-user segment, driven by the increasing number of surgical procedures and the need for advanced medical technologies to improve patient care and operational efficiency .
Italy Medical Devices and Surgical Equipment Market Competitive Landscape
The Italy Medical Devices and Surgical Equipment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Siemens Healthineers, Philips Healthcare, GE Healthcare, Johnson & Johnson, Stryker Corporation, B. Braun Melsungen AG, Abbott Laboratories, Boston Scientific, Zimmer Biomet, Olympus Corporation, Terumo Corporation, Smith & Nephew, Hologic, Inc., Canon Medical Systems, Esaote S.p.A., Sorin Group (now LivaNova PLC), Althea Group S.p.A., Menarini Diagnostics, Diasorin S.p.A. contribute to innovation, geographic expansion, and service delivery in this space.
Italy Medical Devices and Surgical Equipment Market Industry Analysis
Growth Drivers
- Increasing Aging Population:Italy's population aged 65 and older is projected to reach 15.1 million in future, representing approximately 24% of the total population. This demographic shift drives demand for medical devices and surgical equipment, particularly in geriatrics and chronic disease management. The aging population necessitates advanced healthcare solutions, leading to increased investments in medical technologies to cater to age-related health issues, thereby boosting market growth significantly.
- Technological Advancements in Medical Devices:The Italian medical devices sector is witnessing rapid technological innovations, with over €1.6 billion invested in R&D in future. Innovations such as robotic-assisted surgeries and smart implants are enhancing surgical outcomes and patient care. These advancements not only improve efficiency but also reduce recovery times, making them attractive to healthcare providers. Consequently, the integration of cutting-edge technologies is a key driver of market expansion in Italy.
- Rising Healthcare Expenditure:Italy's healthcare expenditure is expected to reach €210 billion in future, reflecting a 5% increase from previous levels. This rise is driven by government initiatives to improve healthcare access and quality. Increased funding allows for the procurement of advanced medical devices and surgical equipment, facilitating better healthcare delivery. As healthcare budgets expand, the demand for innovative medical solutions is anticipated to grow, further propelling market dynamics.
Market Challenges
- Stringent Regulatory Requirements:The Italian medical devices market faces rigorous regulatory frameworks, including compliance with the Medical Device Regulation (MDR). Companies must navigate complex approval processes, which can take up to 12 months for new devices. These stringent regulations can delay product launches and increase operational costs, posing significant challenges for manufacturers aiming to introduce innovative solutions in a timely manner.
- High Competition and Market Saturation:The Italian medical devices market is characterized by intense competition, with over 1,600 companies operating in the sector. This saturation leads to price wars and reduced profit margins, making it difficult for new entrants to establish a foothold. Established players dominate the market, creating barriers for innovation and limiting opportunities for smaller firms to compete effectively in this crowded landscape.
Italy Medical Devices and Surgical Equipment Market Future Outlook
The future of the Italian medical devices and surgical equipment market appears promising, driven by ongoing technological advancements and a growing emphasis on preventive healthcare. As the healthcare landscape evolves, the integration of digital health solutions and telemedicine is expected to reshape service delivery. Additionally, the focus on personalized medicine will likely lead to tailored healthcare solutions, enhancing patient outcomes and driving further investment in innovative medical technologies.
Market Opportunities
- Growth in Telemedicine and Remote Monitoring:The telemedicine market in Italy is projected to reach €1.5 billion in future, driven by increased adoption of remote monitoring technologies. This growth presents opportunities for medical device manufacturers to develop compatible devices that enhance patient care and accessibility, particularly in rural areas where healthcare access is limited.
- Expansion of Home Healthcare Services:The home healthcare market in Italy is expected to grow to €6 billion in future, fueled by an aging population and a preference for at-home care. This trend creates opportunities for medical device companies to innovate and provide home-use devices, catering to the rising demand for convenient and effective healthcare solutions outside traditional clinical settings.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Imaging Devices Surgical Instruments Patient Monitoring Equipment Orthopedic Devices Cardiovascular Devices In Vitro Diagnostic Devices Mobility Aids and Transportation Equipment Sterilization and Disinfectant Equipment Disposable Medical Supplies Syringes and Needles Others |
| By End-User | Hospitals Clinics Home Healthcare Research Laboratories Others |
| By Application | Surgical Procedures Diagnostic Procedures Therapeutic Procedures Rehabilitation Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Price Range | Low-End Devices Mid-Range Devices High-End Devices |
| By Regulatory Compliance | CE Marked Devices Non-CE Marked Devices |
| By Technology | Traditional Technology Advanced Technology Digital Technology |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Italian Medicines Agency, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Trade Associations
Health Insurance Companies
Clinical Research Organizations
Players Mentioned in the Report:
Medtronic
Siemens Healthineers
Philips Healthcare
GE Healthcare
Johnson & Johnson
Stryker Corporation
B. Braun Melsungen AG
Abbott Laboratories
Boston Scientific
Zimmer Biomet
Olympus Corporation
Terumo Corporation
Smith & Nephew
Hologic, Inc.
Canon Medical Systems
Esaote S.p.A.
Sorin Group (now LivaNova PLC)
Althea Group S.p.A.
Menarini Diagnostics
Diasorin S.p.A.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Italy Medical Devices and Surgical Equipment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Italy Medical Devices and Surgical Equipment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Italy Medical Devices and Surgical Equipment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Aging Population
3.1.2 Technological Advancements in Medical Devices
3.1.3 Rising Healthcare Expenditure
3.1.4 Demand for Minimally Invasive Procedures
3.2 Market Challenges
3.2.1 Stringent Regulatory Requirements
3.2.2 High Competition and Market Saturation
3.2.3 Cost Containment Measures by Healthcare Providers
3.2.4 Supply Chain Disruptions
3.3 Market Opportunities
3.3.1 Growth in Telemedicine and Remote Monitoring
3.3.2 Expansion of Home Healthcare Services
3.3.3 Increasing Focus on Preventive Healthcare
3.3.4 Development of Personalized Medicine
3.4 Market Trends
3.4.1 Integration of AI and Machine Learning in Devices
3.4.2 Shift Towards Sustainable Medical Practices
3.4.3 Rise of Digital Health Solutions
3.4.4 Growing Importance of Cybersecurity in Medical Devices
3.5 Government Regulation
3.5.1 Medical Device Regulation (MDR) Compliance
3.5.2 CE Marking Requirements
3.5.3 Post-Market Surveillance Obligations
3.5.4 Reimbursement Policies for Medical Devices
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Italy Medical Devices and Surgical Equipment Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Italy Medical Devices and Surgical Equipment Market Segmentation
8.1 By Type
8.1.1 Diagnostic Imaging Devices
8.1.2 Surgical Instruments
8.1.3 Patient Monitoring Equipment
8.1.4 Orthopedic Devices
8.1.5 Cardiovascular Devices
8.1.6 In Vitro Diagnostic Devices
8.1.7 Mobility Aids and Transportation Equipment
8.1.8 Sterilization and Disinfectant Equipment
8.1.9 Disposable Medical Supplies
8.1.10 Syringes and Needles
8.1.11 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Home Healthcare
8.2.4 Research Laboratories
8.2.5 Others
8.3 By Application
8.3.1 Surgical Procedures
8.3.2 Diagnostic Procedures
8.3.3 Therapeutic Procedures
8.3.4 Rehabilitation
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Retail Pharmacies
8.4.5 Others
8.5 By Price Range
8.5.1 Low-End Devices
8.5.2 Mid-Range Devices
8.5.3 High-End Devices
8.6 By Regulatory Compliance
8.6.1 CE Marked Devices
8.6.2 Non-CE Marked Devices
8.7 By Technology
8.7.1 Traditional Technology
8.7.2 Advanced Technology
8.7.3 Digital Technology
9. Italy Medical Devices and Surgical Equipment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Annual Revenue (EUR million)
9.2.4 Revenue Growth Rate (%)
9.2.5 Market Share (%)
9.2.6 Product Portfolio Breadth (number of product categories)
9.2.7 R&D Expenditure (% of revenue)
9.2.8 Regulatory Compliance Score (CE/MDR)
9.2.9 Distribution Network Coverage (number of regions/countries)
9.2.10 Innovation Index (patents filed, new product launches)
9.2.11 Customer Satisfaction Score (Net Promoter Score or equivalent)
9.2.12 Pricing Strategy (premium, value, competitive)
9.2.13 Brand Recognition (market surveys)
9.2.14 After-Sales Service Quality (response time, support ratings)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic
9.5.2 Siemens Healthineers
9.5.3 Philips Healthcare
9.5.4 GE Healthcare
9.5.5 Johnson & Johnson
9.5.6 Stryker Corporation
9.5.7 B. Braun Melsungen AG
9.5.8 Abbott Laboratories
9.5.9 Boston Scientific
9.5.10 Zimmer Biomet
9.5.11 Olympus Corporation
9.5.12 Terumo Corporation
9.5.13 Smith & Nephew
9.5.14 Hologic, Inc.
9.5.15 Canon Medical Systems
9.5.16 Esaote S.p.A.
9.5.17 Sorin Group (now LivaNova PLC)
9.5.18 Althea Group S.p.A.
9.5.19 Menarini Diagnostics
9.5.20 Diasorin S.p.A.
10. Italy Medical Devices and Surgical Equipment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Procurement Processes
10.1.3 Key Decision-Makers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Infrastructure Development Plans
10.2.3 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Clinics
10.3.3 Home Healthcare Challenges
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development Opportunities
11. Italy Medical Devices and Surgical Equipment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from industry associations such as Assobiomedica
- Review of government publications and healthcare expenditure reports from the Italian Ministry of Health
- Examination of academic journals and white papers on medical device innovations and trends
Primary Research
- Interviews with key opinion leaders in the medical device sector, including surgeons and hospital administrators
- Surveys conducted with procurement managers in hospitals and clinics across Italy
- Focus group discussions with healthcare professionals to gather insights on surgical equipment usage
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including trade publications and market surveys
- Triangulation of qualitative insights from interviews with quantitative data from market reports
- Sanity checks through expert panel reviews comprising industry veterans and regulatory experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare spending and device import/export data
- Segmentation of the market by product categories such as diagnostic devices, surgical instruments, and orthopedic implants
- Incorporation of demographic trends and aging population statistics influencing device demand
Bottom-up Modeling
- Collection of sales data from leading medical device manufacturers operating in Italy
- Estimation of market share based on unit sales and revenue figures from key product segments
- Analysis of distribution channels and their impact on market penetration and growth
Forecasting & Scenario Analysis
- Development of growth projections using historical data and market trends over the past five years
- Scenario analysis based on potential regulatory changes and technological advancements in medical devices
- Creation of baseline, optimistic, and pessimistic forecasts through 2030, considering economic factors
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiovascular Devices | 100 | Cardiologists, Cardiac Surgeons |
| Orthopedic Equipment | 60 | Orthopedic Surgeons, Hospital Procurement Managers |
| Diagnostic Imaging Devices | 75 | Radiologists, Imaging Technologists |
| Surgical Instruments | 55 | Surgeons, Operating Room Managers |
| Patient Monitoring Systems | 50 | Nurses, Clinical Engineers |
Frequently Asked Questions
What is the current value of the Italy Medical Devices and Surgical Equipment Market?
The Italy Medical Devices and Surgical Equipment Market is valued at approximately USD 12.3 billion, reflecting significant growth driven by an aging population, chronic disease prevalence, and advancements in medical technology.