Region:Middle East
Author(s):Dev
Product Code:KRAA3880
Pages:99
Published On:January 2026
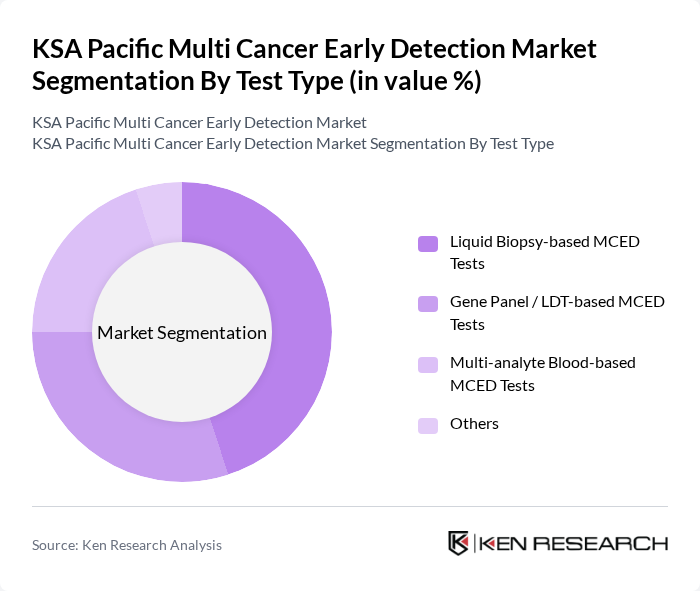
By Test Type:The test type segmentation includes various methodologies for cancer detection. The subsegments are Liquid Biopsy-based MCED Tests, Gene Panel / LDT-based MCED Tests, Multi-analyte Blood-based MCED Tests, and Others. This structure is consistent with major global and Asia Pacific MCED and liquid biopsy classifications that distinguish between circulating tumor DNA assays, gene panel / laboratory-developed tests, and multi-analyte blood-based platforms. Among these, Liquid Biopsy-based MCED Tests are gaining traction due to their non-invasive nature and ability to provide real-time insights into tumor dynamics, which mirrors global trends where liquid biopsy and blood-based MCED tests are increasingly adopted for multi-cancer screening and monitoring. This segment is particularly favored by healthcare providers and patients alike for its convenience and accuracy, especially when integrated with advanced genomic profiling and methylation-based signatures.
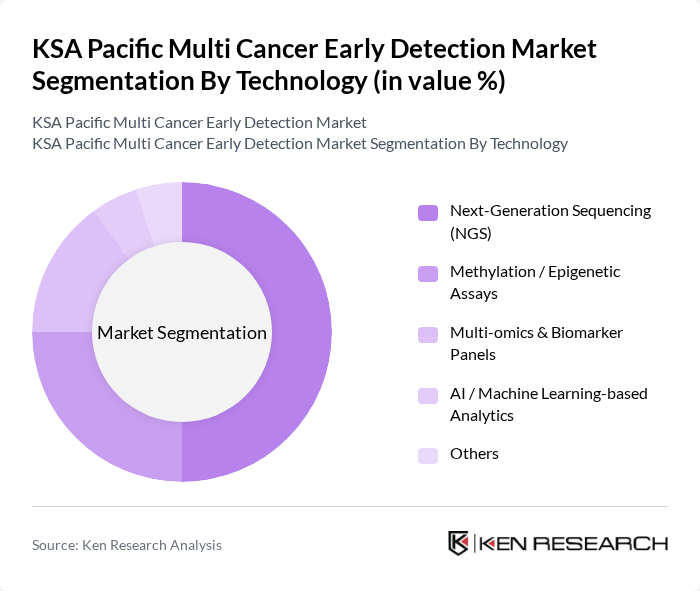
By Technology:The technology segmentation encompasses various innovative approaches used in cancer detection. This includes Next-Generation Sequencing (NGS), Methylation / Epigenetic Assays, Multi-omics & Biomarker Panels, AI / Machine Learning-based Analytics, and Others. This typology aligns with key MCED platforms that use NGS for genomic alterations, methylation profiling for tissue-of-origin, and integrated multi-omics signatures for broad cancer coverage. The Next-Generation Sequencing (NGS) segment is currently leading the market due to its high throughput and accuracy in detecting multiple cancer types simultaneously, making it a preferred choice for diagnostic laboratories and central reference labs involved in MCED and comprehensive genomic profiling.
The KSA Pacific Multi Cancer Early Detection Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Illumina, Inc., Thermo Fisher Scientific Inc., QIAGEN N.V., Bio-Rad Laboratories, Inc., Hologic, Inc., Guardant Health, Inc., Exact Sciences Corporation, GRAIL, LLC, Freenome Holdings, Inc., Foundation Medicine, Inc., Caris Life Sciences, and other emerging MCED players in KSA & Pacific contribute to innovation, geographic expansion, and service delivery in this space, reflecting the global MCED competitive landscape where these companies are consistently profiled as core stakeholders.
The KSA Pacific Multi Cancer Early Detection Market is poised for transformative growth, driven by technological innovations and increased government support. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while personalized medicine approaches will cater to individual patient needs. Additionally, the expansion of telemedicine services will facilitate remote screenings, making early detection more accessible. These trends indicate a promising future for the market, with a focus on improving patient outcomes and reducing healthcare costs.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Liquid Biopsy-based MCED Tests Gene Panel / LDT-based MCED Tests Multi-analyte Blood-based MCED Tests Others |
| By Technology | Next-Generation Sequencing (NGS) Methylation / Epigenetic Assays Multi-omics & Biomarker Panels AI / Machine Learning-based Analytics Others |
| By Cancer Detection Scope | Pan-Cancer (Multi-site) Tests Selected High-Incidence Cancer Panels Organ-specific MCED Panels Others |
| By End-User | Hospitals & Cancer Centers Reference & Diagnostic Laboratories Specialty Clinics Corporate & Preventive Health Programs Others |
| By Distribution / Access Channel | Hospital-based Programs Laboratory Referral Model Direct-to-Consumer (DTC) / Digital Health Platforms Employer / Payer-led Programs Others |
| By Geography (KSA & Wider Pacific) | Saudi Arabia – Central Region Saudi Arabia – Eastern Region Saudi Arabia – Western Region Saudi Arabia – Southern & Other Regions Key Pacific Markets (e.g., Japan, South Korea, Australia, Others) |
| By Reimbursement & Policy Support | Government-funded Screening Programs Public & Private Insurance Coverage Out-of-Pocket / Self-pay Segment Public Health Campaigns & Incentives Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinics | 110 | Oncologists, Clinic Administrators |
| Diagnostic Laboratories | 90 | Laboratory Managers, Technicians |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Policy Analysts |
| Patient Advocacy Groups | 60 | Advocacy Leaders, Patient Representatives |
| Insurance Providers | 70 | Health Insurance Analysts, Underwriters |
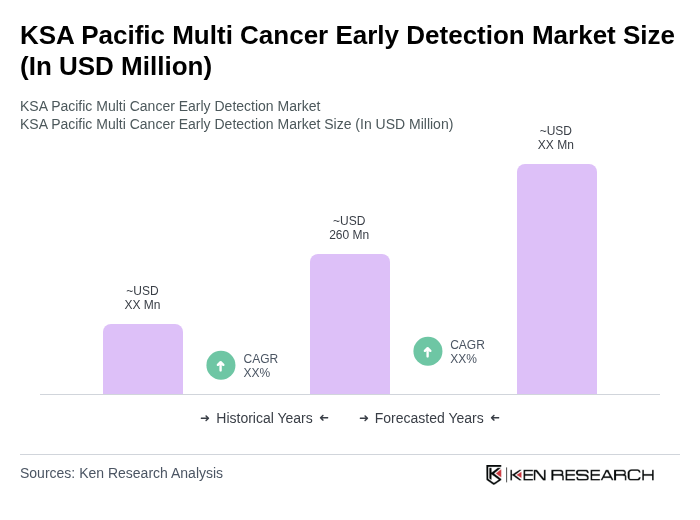
The KSA Pacific Multi Cancer Early Detection Market is valued at approximately USD 260 million, reflecting significant growth driven by increased awareness of cancer screening and advancements in diagnostic technologies.