Region:Middle East
Author(s):Geetanshi
Product Code:KRAD3734
Pages:96
Published On:November 2025
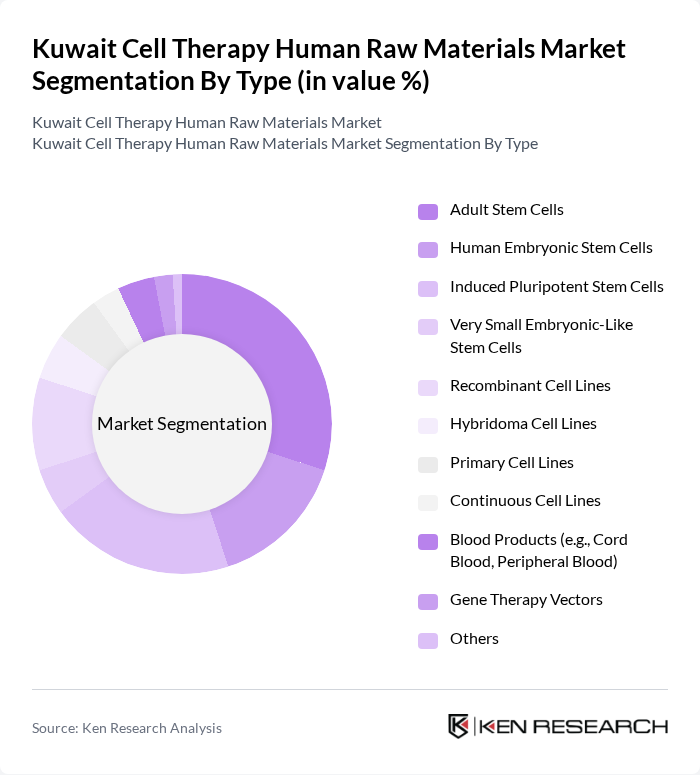
By Type:The market is segmented into various types of human raw materials, including Adult Stem Cells, Human Embryonic Stem Cells, Induced Pluripotent Stem Cells, Very Small Embryonic-Like Stem Cells, Recombinant Cell Lines, Hybridoma Cell Lines, Primary Cell Lines, Continuous Cell Lines, Blood Products (e.g., Cord Blood, Peripheral Blood), Gene Therapy Vectors, and Others. Among these, Adult Stem Cells are currently dominating the market due to their versatility, lower ethical concerns, and wide application in regenerative medicine and clinical research .
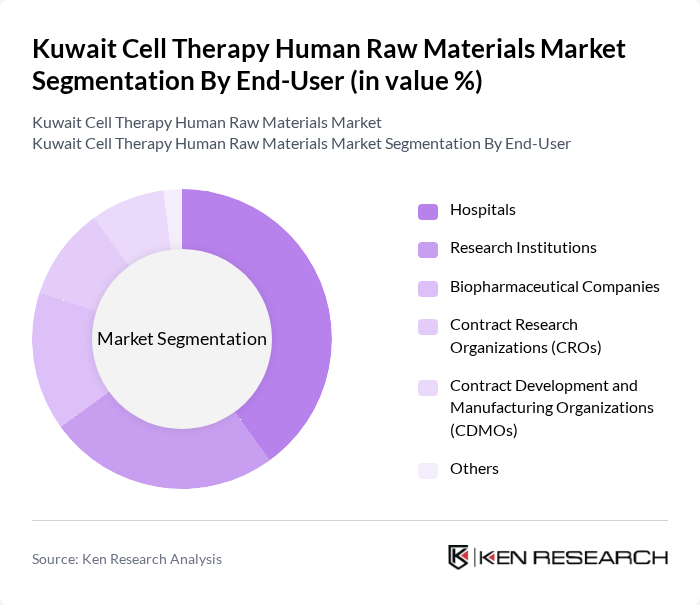
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Biopharmaceutical Companies, Contract Research Organizations (CROs), Contract Development and Manufacturing Organizations (CDMOs), and Others. Hospitals are the leading end-user segment, driven by the increasing adoption of cell therapies for treating various diseases, the expansion of clinical trial activity, and the integration of advanced cell-based treatments into standard care protocols .
The Kuwait Cell Therapy Human Raw Materials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Kuwait Institute for Scientific Research (KISR), Gulf Biotechnology Company, Al-Dar Biotechnology, Kuwait Cancer Control Center, Kuwait University – Faculty of Medicine, BioKuwait, Mubarak Al-Kabeer Hospital Research Center, Dasman Diabetes Institute, Advanced Technology Company, Central Blood Bank Kuwait, Ministry of Health – Biotechnology Division, Kuwait Foundation for the Advancement of Sciences (KFAS), Kuwait Medical Genetics Center, Jaber Al-Ahmad Hospital Research Unit, Kuwait Society for Biotechnology Sciences contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait cell therapy human raw materials market appears promising, driven by ongoing advancements in biotechnology and increasing government support. As the prevalence of chronic diseases continues to rise, the demand for innovative therapies will likely escalate. Furthermore, the integration of artificial intelligence in research is expected to enhance the efficiency of cell therapy development, paving the way for more effective treatments. This evolving landscape presents significant opportunities for growth and innovation in the sector.
| Segment | Sub-Segments |
|---|---|
| By Type | Adult Stem Cells Human Embryonic Stem Cells Induced Pluripotent Stem Cells Very Small Embryonic-Like Stem Cells Recombinant Cell Lines Hybridoma Cell Lines Primary Cell Lines Continuous Cell Lines Blood Products (e.g., Cord Blood, Peripheral Blood) Gene Therapy Vectors Others |
| By End-User | Hospitals Research Institutions Biopharmaceutical Companies Contract Research Organizations (CROs) Contract Development and Manufacturing Organizations (CDMOs) Others |
| By Source | Autologous Sources Allogeneic Sources Cord Blood Human-Derived Cell Lines Animal-Derived Cell Lines Others |
| By Application | Oncology Orthopedics Cardiovascular Neurology Bioproduction (Biologics, Biosimilars, Vaccines) Drug Discovery & Development Tissue Engineering & Regenerative Medicine Diagnostics Gene Therapy Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Hospital Pharmacy Others |
| By Technology | Cell Culture Techniques Gene Editing Technologies (e.g., CRISPR/Cas9) Cell Preservation Technologies Transfection Technology Viral Vector Technology Single-Use Bioprocessing Technology Others |
| By Regulatory Compliance | FDA Approved EMA Approved Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers in Cell Therapy | 60 | Doctors, Surgeons, Medical Directors |
| Regulatory Bodies and Health Authorities | 40 | Regulatory Affairs Specialists, Policy Makers |
| Patients Receiving Cell Therapy | 50 | Patients, Caregivers, Patient Advocacy Groups |
| Research Institutions and Universities | 45 | Researchers, Professors, Graduate Students |
| Manufacturers of Cell Therapy Products | 40 | Product Managers, Quality Assurance Officers |
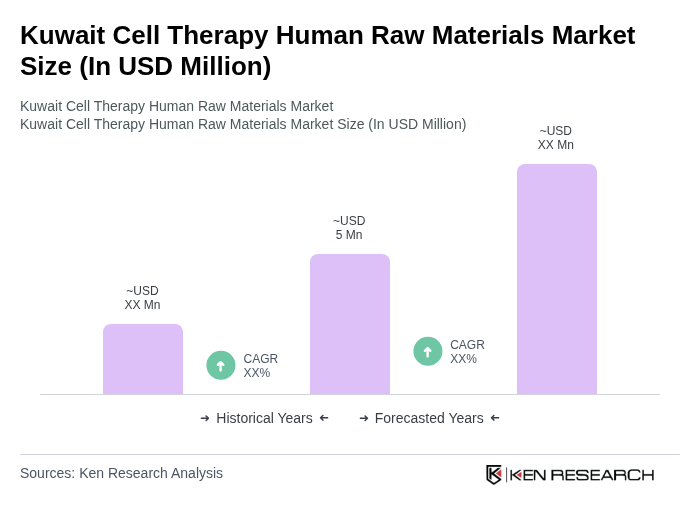
The Kuwait Cell Therapy Human Raw Materials Market is valued at approximately USD 5 million, reflecting growth driven by advancements in regenerative medicine and increasing demand for innovative therapies to treat chronic diseases.