Region:Middle East
Author(s):Dev
Product Code:KRAC2069
Pages:90
Published On:October 2025
By Product:The product segmentation includes essential components for CRISPR diagnostics, such as enzymes (Cas9, Cas12, Cas13), kits and reagents, software and libraries, and instruments. Enzymes are critical for the gene-editing and detection process, kits and reagents streamline laboratory workflows and point-of-care testing, software and libraries enable data analysis and assay design, while instruments provide the necessary hardware for sample processing and detection. User preferences are shaped by the need for accuracy, speed, and scalability in clinical and research settings .
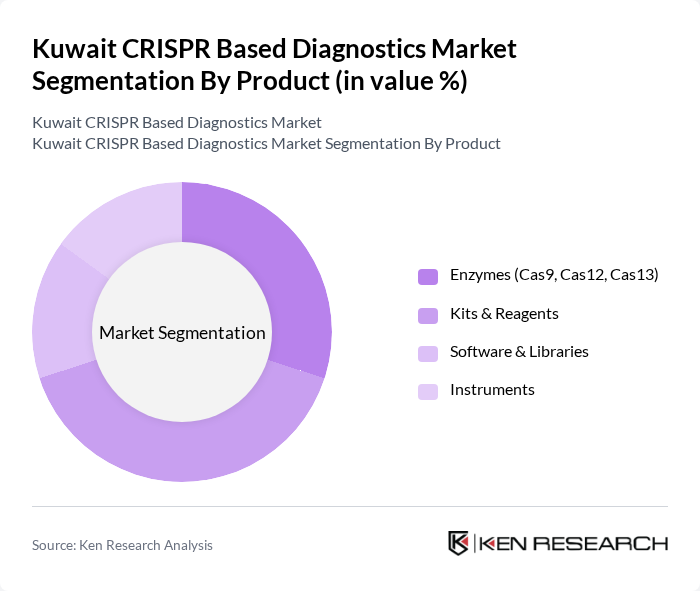
The kits and reagents sub-segment leads the market, reflecting their indispensable role in enabling rapid, accurate CRISPR-based diagnostic tests. These products are essential for both laboratory and point-of-care settings, supporting the growing demand for scalable, user-friendly solutions. The rise in genetic screening programs and infectious disease surveillance has further increased the adoption of kits and reagents, positioning this segment as the market leader .
By Technology:The technology segmentation covers CRISPR-Cas9, Cas12, Cas13, and emerging alternatives. CRISPR-Cas9 is widely used for its versatility in DNA targeting, Cas12 for its rapid detection capabilities, and Cas13 for RNA-based diagnostics. Each technology addresses distinct diagnostic needs, from hereditary disease screening to infectious pathogen detection .
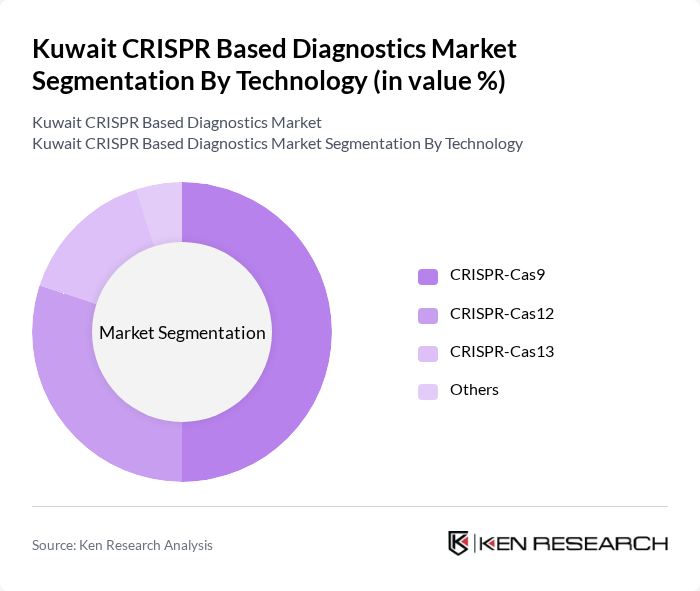
CRISPR-Cas9 technology dominates the market due to its broad applicability in genetic diagnostics and research. Its precision in targeting specific DNA sequences makes it highly effective for identifying hereditary conditions and infectious agents. The increasing adoption of personalized medicine and targeted therapies continues to drive demand for CRISPR-Cas9-based diagnostics, ensuring its leading position in the market .
The Kuwait CRISPR Based Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Integrated DNA Technologies, Inc., Sherlock Biosciences, Mammoth Biosciences, Danaher Corporation, Molbio Diagnostics, Bio-Rad Laboratories, Inc., QIAGEN N.V., New England Biolabs, Inc., Caribou Biosciences, Inc., Synthego Corporation, ToolGen Inc., Horizon Discovery (Revvity Inc.), CrisprBits Private Limited, GenScript Biotech Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the CRISPR-based diagnostics market in Kuwait appears promising, driven by technological advancements and increasing healthcare investments. As the government emphasizes biotechnology research, collaboration between public and private sectors is expected to flourish. Additionally, the integration of artificial intelligence in diagnostics will enhance accuracy and efficiency. These trends indicate a robust growth trajectory, positioning Kuwait as a potential leader in the region for innovative diagnostic solutions, particularly in personalized medicine and rapid testing technologies.
| Segment | Sub-Segments |
|---|---|
| By Product | Enzymes (Cas9, Cas12, Cas13) Kits & Reagents Software & Libraries Instruments |
| By Technology | CRISPR-Cas9 CRISPR-Cas12 CRISPR-Cas13 Others |
| By Application | Infectious Disease Detection Genetic Disease Screening Cancer Diagnostics Agricultural Diagnostics Others |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Academic & Research Institutions Biotechnology & Pharmaceutical Companies Others |
| By Test Type | Point-of-Care (POC) Testing Laboratory-Based Testing At-Home Testing |
| By Disease Type | Viral Infections (COVID-19, Dengue, Zika) Bacterial Infections Genetic Disorders Oncology Others |
| By Distribution Channel | Direct Sales Distributors & Suppliers Online Channels |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 60 | Laboratory Directors, Diagnostic Technologists |
| Healthcare Providers | 50 | Physicians, Medical Researchers |
| Biotechnology Firms | 40 | Product Managers, R&D Scientists |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Academic Institutions | 50 | Professors, Graduate Researchers |
The Kuwait CRISPR Based Diagnostics Market is valued at approximately USD 135 million, reflecting a strong demand for advanced molecular diagnostics due to the high prevalence of genetic disorders and a growing focus on precision medicine in the region.