Region:Middle East
Author(s):Geetanshi
Product Code:KRAD4826
Pages:80
Published On:December 2025
By Treatment Type:
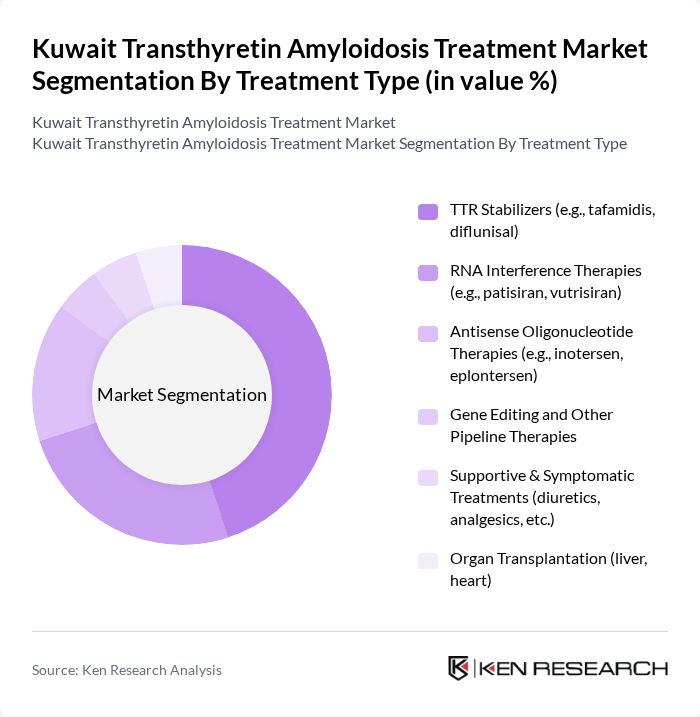
The treatment type segmentation includes TTR Stabilizers, RNA Interference Therapies, Antisense Oligonucleotide Therapies, Gene Editing and Other Pipeline Therapies, Supportive & Symptomatic Treatments, and Organ Transplantation. TTR Stabilizers, particularly tafamidis, are widely regarded as a cornerstone therapy for ATTR cardiomyopathy due to robust evidence demonstrating reduction in all-cause mortality and cardiovascular-related hospitalizations and slowing of functional decline. The increasing adoption of tafamidis in the Gulf region, supported by guideline recommendations and funding decisions for selected patients, has significantly contributed to the prominence of this class. RNA Interference Therapies such as patisiran and vutrisiran are also gaining traction for hereditary ATTR polyneuropathy, driven by their gene-silencing mechanism, sustained TTR knockdown, and data showing improvements in neuropathy impairment scores, quality of life, and certain cardiac parameters. Antisense oligonucleotide therapies like inotersen and eplontersen provide an additional disease-modifying option, particularly for patients with hATTR polyneuropathy, and are increasingly positioned alongside RNAi agents in international treatment algorithms.
By Disease Type:
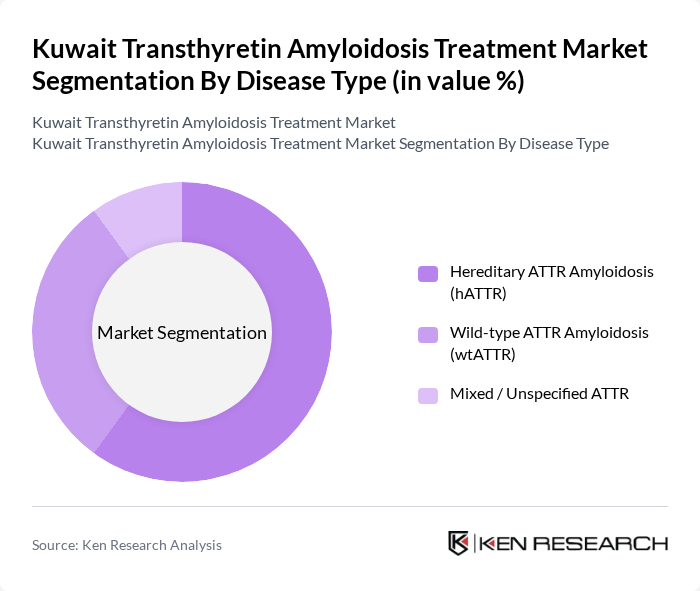
This segmentation includes Hereditary ATTR Amyloidosis (hATTR), Wild-type ATTR Amyloidosis (wtATTR), and Mixed / Unspecified ATTR. Hereditary ATTR amyloidosis is driven by pathogenic TTR variants, with certain founder mutations such as Val30Met and others reported at higher frequencies in specific populations in the Middle East and Mediterranean regions, contributing to clustering of hATTR cases in extended families. Growing availability of genetic testing, family screening, and multidisciplinary amyloidosis clinics has increased the recognition of hATTR and the use of targeted disease-modifying therapies. Wild-type ATTR, typically associated with elderly males and presenting predominantly as cardiomyopathy, is also clinically important and increasingly diagnosed worldwide with advances in bone scintigraphy and cardiac imaging; however, hereditary forms remain a major focus of clinical research and therapeutic development globally.
The Kuwait Transthyretin Amyloidosis Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Alnylam Pharmaceuticals, Inc., Pfizer Inc., Ionis Pharmaceuticals, Inc., AstraZeneca plc, Akcea Therapeutics, Inc. (Ionis Pharmaceuticals subsidiary), BridgeBio Pharma, Inc. (including Eidos Therapeutics), Prothena Corporation plc, Sorrento Therapeutics, Inc., Corino Therapeutics, Inc., Alexion Pharmaceuticals, Inc. (AstraZeneca Rare Disease), Novartis AG, Takeda Pharmaceutical Company Limited, Roche Holding AG, Sanofi, Kuwait Ministry of Health & Kuwait Cancer Control Center (as key institutional stakeholders) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait Transthyretin Amyloidosis treatment market appears promising, driven by ongoing advancements in medical technology and increased healthcare investments. The government is expected to enhance healthcare infrastructure, facilitating better access to specialized treatments. Additionally, collaborations between local healthcare providers and international pharmaceutical companies will likely foster innovation in therapy development. As awareness continues to grow, the market is poised for significant expansion, ultimately improving patient outcomes and treatment accessibility in Kuwait.
| Segment | Sub-Segments |
|---|---|
| By Treatment Type | TTR Stabilizers (e.g., tafamidis, diflunisal) RNA Interference Therapies (e.g., patisiran, vutrisiran) Antisense Oligonucleotide Therapies (e.g., inotersen, eplontersen) Gene Editing and Other Pipeline Therapies Supportive & Symptomatic Treatments (diuretics, analgesics, etc.) Organ Transplantation (liver, heart) |
| By Disease Type | Hereditary ATTR Amyloidosis (hATTR) Wild-type ATTR Amyloidosis (wtATTR) Mixed / Unspecified ATTR |
| By Clinical Manifestation | ATTR Cardiomyopathy (ATTR-CM) ATTR Polyneuropathy (ATTR-PN) Mixed Cardio-neuropathic Involvement |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Specialty Pharmacies |
| By Treatment Setting | Tertiary Care / University Hospitals Secondary Care Hospitals Specialist Cardiology & Neurology Centers Home-based Infusion & Follow-up Care |
| By Healthcare Provider Type | Public Sector Providers (Ministry of Health hospitals) Private Hospitals & Clinics Military and Government-affiliated Hospitals Regional Referral Centers Abroad (medical tourism) |
| By Patient Demographics | Age Group (?40 years, 41–60 years, ?61 years) Gender Nationality (Kuwaiti vs Expatriate) Presence of Key Comorbidities (heart failure, CKD, diabetes, etc.) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Oncologists, Hematologists, General Practitioners |
| Pharmaceutical Stakeholders | 90 | Product Managers, Market Access Specialists |
| Patient Advocacy Groups | 60 | Patient Representatives, Support Group Leaders |
| Health Insurance Companies | 70 | Policy Analysts, Claims Managers |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
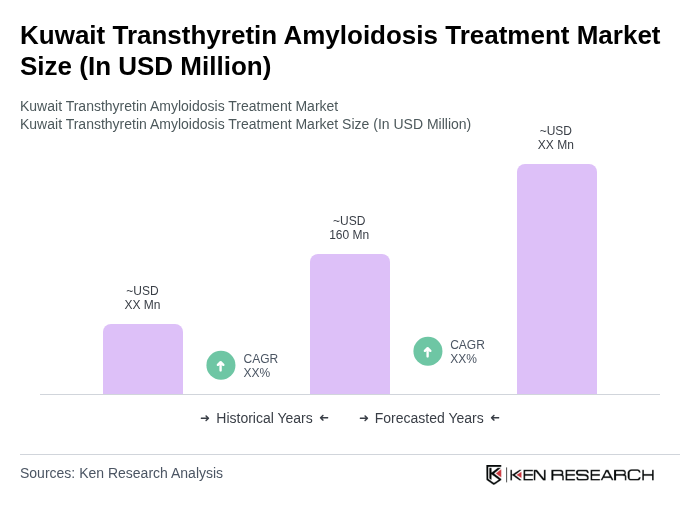
The Kuwait Transthyretin Amyloidosis Treatment Market is valued at approximately USD 160 million, reflecting a five-year historical analysis. This growth is driven by increased clinical recognition and the availability of targeted therapies for transthyretin amyloidosis (ATTR).