Region:Middle East
Author(s):Rebecca
Product Code:KRAC9745
Pages:82
Published On:November 2025
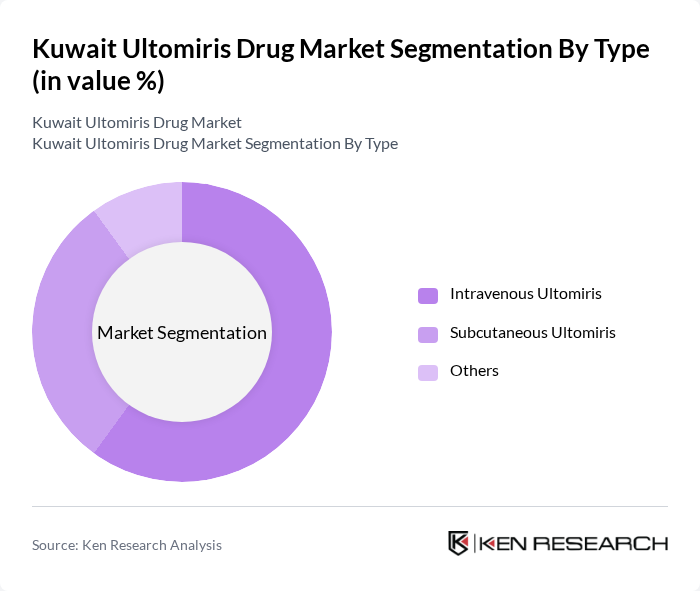
By Type:The market is segmented into three types: Intravenous Ultomiris, Subcutaneous Ultomiris, and Others. Intravenous Ultomiris is the leading sub-segment due to its established efficacy, rapid onset of action, and preference in acute care settings. Subcutaneous Ultomiris is gaining traction as it offers convenience, reduced infusion times, and improved patient adherence. The "Others" category includes less common formulations and delivery methods, such as investigational or off-label routes .
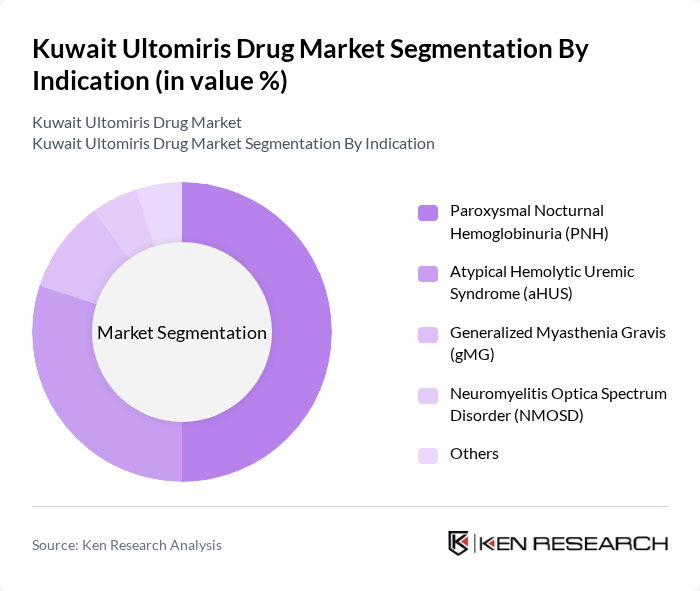
By Indication:The market is categorized into Paroxysmal Nocturnal Hemoglobinuria (PNH), Atypical Hemolytic Uremic Syndrome (aHUS), Generalized Myasthenia Gravis (gMG), Neuromyelitis Optica Spectrum Disorder (NMOSD), and Others. PNH is the dominant indication due to its higher prevalence among rare complement-mediated disorders and the critical need for effective, long-acting treatment. aHUS follows closely, with increasing awareness, improved diagnostic rates, and expanded insurance coverage driving patient identification and therapy initiation. The inclusion of gMG and NMOSD reflects recent regulatory approvals and growing clinical adoption .
The Kuwait Ultomiris Drug Market is characterized by a dynamic mix of regional and international players. Leading participants such as Alexion Pharmaceuticals (AstraZeneca Rare Disease), AstraZeneca PLC, Sobi (Swedish Orphan Biovitrum AB), Roche Holding AG, Novartis AG, Takeda Pharmaceutical Company Limited, Amgen Inc., Pfizer Inc., Sanofi S.A., AbbVie Inc., GSK (GlaxoSmithKline plc), Eli Lilly and Company, Bristol-Myers Squibb Company, Biogen Inc., Astellas Pharma Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait Ultomiris drug market appears promising, driven by increasing investments in healthcare infrastructure and a growing focus on rare diseases. As the government continues to prioritize healthcare spending, the market is likely to see enhanced access to innovative therapies. Additionally, the integration of digital health solutions and telemedicine is expected to facilitate better patient management. These trends will create a conducive environment for the growth of Ultomiris and similar therapies, ultimately improving patient outcomes in Kuwait.
| Segment | Sub-Segments |
|---|---|
| By Type | Intravenous Ultomiris Subcutaneous Ultomiris Others |
| By Indication | Paroxysmal Nocturnal Hemoglobinuria (PNH) Atypical Hemolytic Uremic Syndrome (aHUS) Generalized Myasthenia Gravis (gMG) Neuromyelitis Optica Spectrum Disorder (NMOSD) Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Region | Capital Governorate Hawalli Governorate Al Ahmadi Governorate Farwaniya Governorate Mubarak Al-Kabeer Governorate Jahra Governorate Others |
| By Treatment Setting | Inpatient Settings Outpatient Settings Home Healthcare Others |
| By Pricing Tier | Premium Tier Mid Tier Budget Tier Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinics | 60 | Oncologists, Nurse Practitioners |
| Pharmacies Dispensing Ultomiris | 50 | Pharmacists, Pharmacy Managers |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
| Healthcare Administrators | 45 | Hospital Administrators, Procurement Officers |
| Health Insurance Providers | 40 | Claims Analysts, Policy Managers |
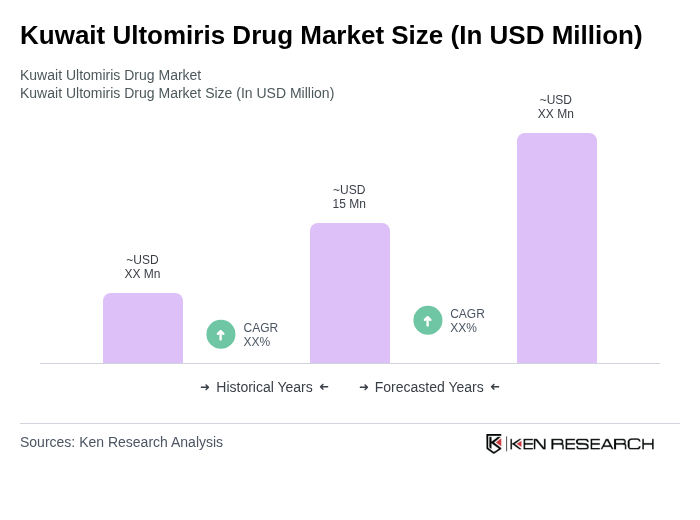
The Kuwait Ultomiris Drug Market is valued at approximately USD 15 million, driven by the increasing prevalence of rare diseases and advancements in healthcare infrastructure, alongside rising healthcare expenditure and improved diagnostic capabilities.