Region:Middle East
Author(s):Geetanshi
Product Code:KRAA9078
Pages:87
Published On:November 2025
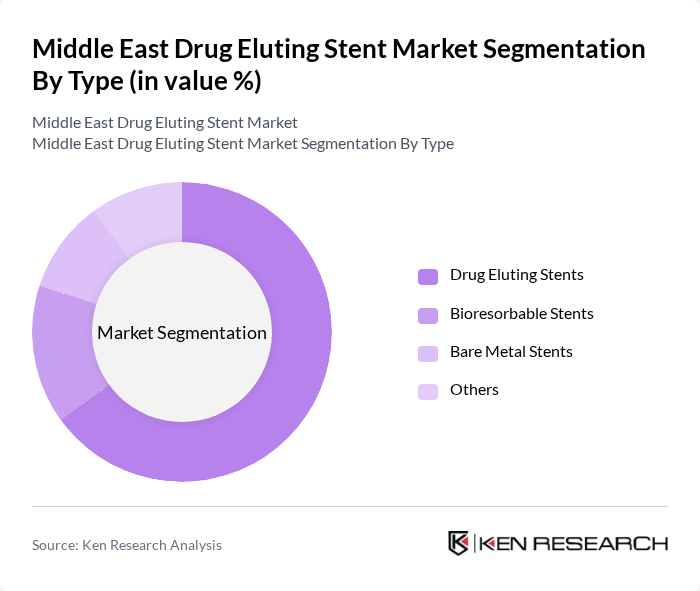
By Type:The market is segmented into various types of stents, including Drug Eluting Stents, Bioresorbable Stents, Bare Metal Stents, and Others. Among these, Drug Eluting Stents are the most widely used due to their ability to release medication that helps prevent the re-narrowing of arteries. Bioresorbable Stents are gaining traction as they dissolve over time, reducing long-term complications and improving vessel healing. Bare Metal Stents, while still in use, are less favored due to higher rates of restenosis and limited long-term efficacy.
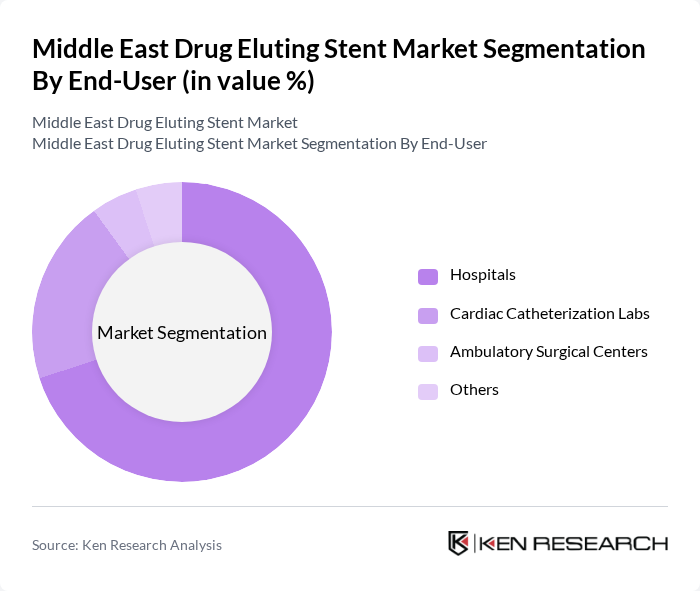
By End-User:The end-user segmentation includes Hospitals, Cardiac Catheterization Labs, Ambulatory Surgical Centers, and Others. Hospitals are the primary end-users, as they provide comprehensive cardiac care and have the necessary infrastructure for complex procedures. Cardiac Catheterization Labs are also significant users, focusing on minimally invasive procedures and advanced interventional cardiology. Ambulatory Surgical Centers are emerging as a viable option for outpatient procedures, contributing to the market's growth, especially for elective and lower-risk cases.
The Middle East Drug Eluting Stent Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Boston Scientific Corporation, Medtronic plc, Johnson & Johnson (Biosense Webster/Cordis), B. Braun SE, Terumo Corporation, Cook Medical, Cardinal Health, Lepu Medical Technology (Beijing) Co., Ltd., Meril Life Sciences Pvt. Ltd., MicroPort Scientific Corporation, Biotronik SE & Co. KG, Biosensors International Group, Ltd., Elixir Medical Corporation, Medinol Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East drug eluting stent market appears promising, driven by ongoing advancements in medical technology and increasing healthcare investments. As healthcare infrastructure expands, particularly in emerging economies, access to innovative cardiovascular treatments will improve. Additionally, the growing emphasis on preventive care and early intervention is likely to enhance patient outcomes, fostering a more proactive approach to managing cardiovascular diseases in the region, ultimately benefiting both patients and healthcare providers.
| Segment | Sub-Segments |
|---|---|
| By Type | Drug Eluting Stents Bioresorbable Stents Bare Metal Stents Others |
| By End-User | Hospitals Cardiac Catheterization Labs Ambulatory Surgical Centers Others |
| By Material | Stainless Steel Cobalt Chromium Platinum Chromium Others |
| By Delivery Method | Balloon-Expandable Stents Self-Expanding Stents Others |
| By Coating Type | Polymer Coated Stents Non-Polymer Coated Stents Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Application | Coronary Artery Disease Peripheral Artery Disease Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists in Major Hospitals | 60 | Interventional Cardiologists, Electrophysiologists |
| Procurement Managers in Healthcare Facilities | 50 | Hospital Procurement Officers, Supply Chain Managers |
| Medical Device Sales Representatives | 40 | Sales Managers, Territory Representatives |
| Healthcare Policy Makers | 40 | Health Economists, Regulatory Affairs Specialists |
| Clinical Researchers in Cardiology | 45 | Clinical Trial Coordinators, Research Scientists |
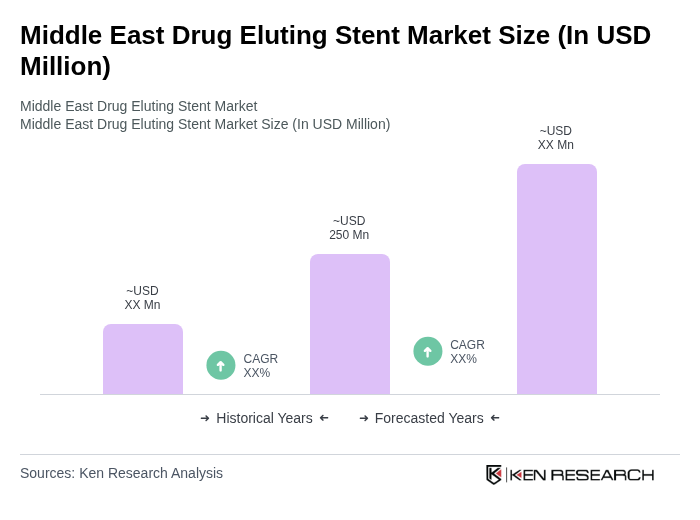
The Middle East Drug Eluting Stent Market is valued at approximately USD 250 million, driven by the rising prevalence of cardiovascular diseases and advancements in stent technology, among other factors.