About the Report
Base Year 2024Middle East Emergency Contraceptive Pills Market Overview
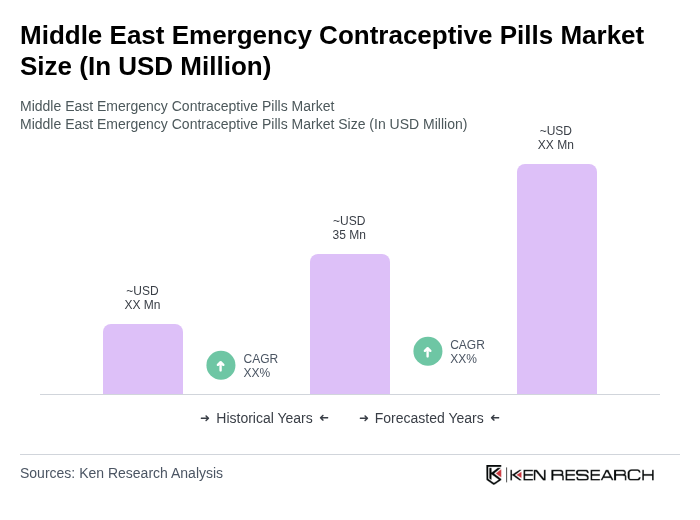
- The Middle East Emergency Contraceptive Pills Market is valued at USD 35 million, based on a five-year historical analysis and its share within the global emergency contraceptive pills market, which is estimated in the mid-hundreds of millions of USD globally. This growth is primarily driven by increasing awareness of reproductive health, rising incidences of unprotected intercourse, and the growing acceptance of emergency contraceptive methods among women, in line with global trends where education campaigns and advocacy have expanded access and normalized ECP use. The market has seen a significant uptick in demand due to educational campaigns, broader over?the?counter availability in several countries, digital/telehealth-based contraceptive counseling, and improved access to contraceptive options through pharmacy and online channels.
- Key players in this market include Saudi Arabia, the United Arab Emirates, and Turkey. These countries contribute a substantial share of regional demand due to relatively higher healthcare spending, expanding private pharmacy chains, and national reproductive health programs that include modern contraception. Urbanization, higher disposable incomes, and increasing participation of women in the workforce in these markets also support the growing demand for emergency contraceptive pills and broader hormonal contraceptive use.
- In Saudi Arabia, emergency contraceptive pills are regulated as prescription medicines under the national drug and pharmacy framework administered by the Saudi Food and Drug Authority (SFDA); the SFDA’s Pharmaceutical Products Registration guidelines and the Law of Pharmaceutical Establishments and Preparations (Council of Ministers, Saudi Arabia, 2004; SFDA implementing rules) govern registration, classification, and dispensing conditions for hormonal and contraceptive products, including emergency pills, requiring that any switch to over?the?counter status be explicitly approved through SFDA reclassification procedures. While global practice in several countries has moved toward non?prescription access for levonorgestrel ECPs, public SFDA regulations and regional policy reviews do not document a 2023 Saudi regulation that universally authorizes over?the?counter sales of emergency contraceptive pills without a prescription, and ECP availability in the Kingdom remains subject to SFDA classification and pharmacy practice standards.
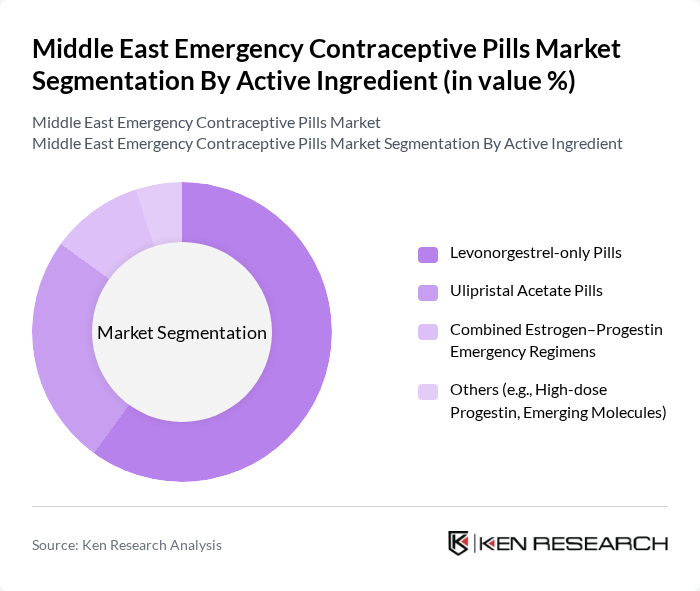
Middle East Emergency Contraceptive Pills Market Segmentation
By Active Ingredient:The market is segmented into Levonorgestrel-only Pills, Ulipristal Acetate Pills, Combined Estrogen–Progestin Emergency Regimens, and Others (e.g., High-dose Progestin, Emerging Molecules). This schema is aligned with global clinical and regulatory recognition of levonorgestrel, ulipristal acetate, and certain combined oral contraceptives as emergency methods. Among these, Levonorgestrel-only Pills dominate the market due to their widespread availability, inclusion on essential medicines lists in many countries, established efficacy, and lower cost versus newer molecules. They are preferred by consumers and providers for their single-dose regimens, favorable safety profile, and rapid time-to-dispensing through retail pharmacies and, in some markets, e?pharmacies.
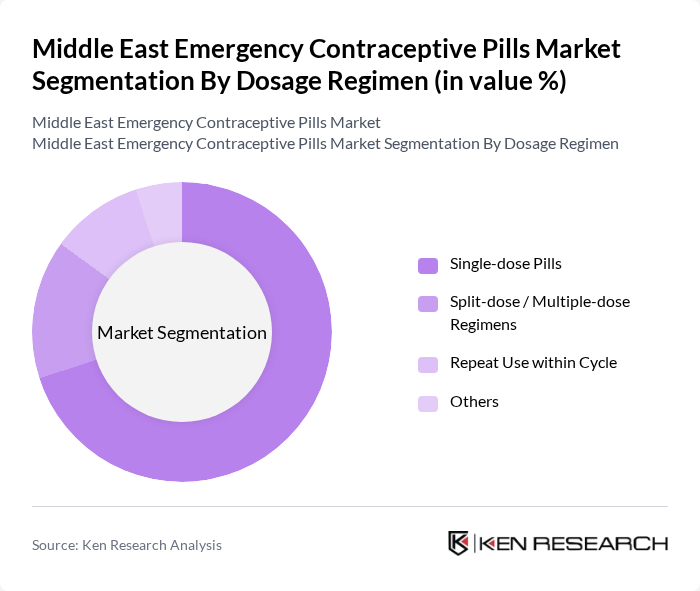
By Dosage Regimen:The market is categorized into Single-dose Pills, Split-dose / Multiple-dose Regimens, Repeat Use within Cycle, and Others. This categorization reflects prevalent clinical use patterns, where most commercially available levonorgestrel and ulipristal acetate products are single-dose, while some combined hormonal regimens follow split?dose protocols. The Single-dose Pills segment leads the market, as they offer convenience, simplified adherence, and immediate effectiveness, appealing to consumers who seek quick solutions for emergency contraception. This segment's popularity is further bolstered by pharmacy-level counseling, online awareness campaigns, and telehealth platforms that stress the importance of taking emergency contraception as soon as possible after unprotected intercourse.
Middle East Emergency Contraceptive Pills Market Competitive Landscape
The Middle East Emergency Contraceptive Pills Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bayer AG, Gedeon Richter Plc., HRA Pharma (Perrigo Company plc), Teva Pharmaceutical Industries Ltd., Pfizer Inc., Mylan N.V. (Viatris Inc.), Sun Pharmaceutical Industries Ltd., Cipla Limited, Glenmark Pharmaceuticals Ltd., Zydus Lifesciences Limited (formerly Zydus Cadila), HLL Lifecare Limited, Sandoz (Novartis Division / Sandoz Group), Ferring Pharmaceuticals, Afaxys Pharma LLC, Foundation Consumer Healthcare LLC contribute to innovation, geographic expansion, and service delivery in this space, supported by global portfolios of levonorgestrel and ulipristal acetate brands and extensive distribution relationships with retail and hospital pharmacies.
Middle East Emergency Contraceptive Pills Market Industry Analysis
Growth Drivers
- Increasing Awareness of Reproductive Health:The Middle East has seen a significant rise in reproductive health awareness, with over 60% of women aged 15-49 now knowledgeable about emergency contraceptive options. This shift is supported by educational initiatives from organizations like the World Health Organization, which reported a 30% increase in reproductive health program funding in the region. As awareness grows, the demand for emergency contraceptive pills is expected to rise, leading to increased accessibility and usage.
- Rising Demand for Family Planning Solutions:The demand for family planning solutions in the Middle East is projected to increase, with a reported 25% rise in contraceptive use among married women from 2015 to 2023. According to the United Nations, the region's population is expected to grow by 1.5% annually, intensifying the need for effective family planning methods. This trend is driving the market for emergency contraceptive pills as a critical component of reproductive health strategies.
- Government Initiatives Promoting Contraceptive Use:Various governments in the Middle East are implementing initiatives to promote contraceptive use, with funding exceeding $200 million for reproductive health programs. Countries like Egypt and Jordan have launched campaigns to increase access to contraceptives, including emergency pills. These initiatives aim to reduce unintended pregnancies and improve maternal health, thereby fostering a more supportive environment for emergency contraceptive use.
Market Challenges
- Cultural and Religious Barriers to Contraceptive Use:Cultural and religious beliefs significantly hinder the acceptance of emergency contraceptive pills in the Middle East. A survey by the Arab Barometer indicated that over 70% of respondents in conservative regions view contraceptive use unfavorably. This stigma creates a barrier to access and education, limiting the market's growth potential and affecting women's reproductive choices across the region.
- Regulatory Hurdles in Product Approval:The regulatory landscape for emergency contraceptive pills in the Middle East is complex, with approval processes often taking over 18 months. According to the World Bank, only 40% of new contraceptive products receive timely approval due to stringent regulations. This delay in product availability can stifle market growth and limit options for consumers seeking emergency contraceptive solutions.
Middle East Emergency Contraceptive Pills Market Future Outlook
The future of the Middle East emergency contraceptive pills market appears promising, driven by increasing awareness and supportive government policies. As telemedicine and online consultations gain traction, access to emergency contraceptives is expected to improve significantly. Additionally, the shift towards over-the-counter availability will likely enhance consumer confidence and usage rates. These trends indicate a growing acceptance of emergency contraceptive pills as essential components of reproductive health strategies in the region.
Market Opportunities
- Introduction of New Product Formulations:The development of innovative product formulations, such as non-hormonal emergency contraceptives, presents a significant market opportunity. With increasing consumer demand for diverse options, companies can cater to varying preferences, potentially increasing market penetration and sales.
- Partnerships with Healthcare Providers:Collaborating with healthcare providers can enhance distribution channels and improve education on emergency contraceptive pills. By leveraging existing healthcare networks, companies can increase awareness and accessibility, ultimately driving sales and fostering a more informed consumer base.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Active Ingredient | Levonorgestrel-only Pills Ulipristal Acetate Pills Combined Estrogen–Progestin Emergency Regimens Others (e.g., High-dose Progestin, Emerging Molecules) |
| By Dosage Regimen | Single-dose Pills Split-dose / Multiple-dose Regimens Repeat Use within Cycle Others |
| By Distribution Channel | Retail Pharmacies Hospital Pharmacies Online / E-commerce Pharmacies Clinics & NGO / Community Health Channels |
| By Country | Saudi Arabia United Arab Emirates Qatar Kuwait Bahrain Oman Israel Turkey Rest of Middle East |
| By Indication / Use Case | Unprotected Intercourse Contraceptive Failure (e.g., Condom Breakage, Missed Pills) Sexual Assault-related Emergency Contraception Others |
| By Point of Purchase | Over-the-counter (OTC) Behind-the-counter / Pharmacist-mediated Prescription-based NGO / Public-sector Supply |
| By Price Tier | Economy / Generic Brands Mid-range Branded Products Premium / Imported Brands Subsidized / Donor-funded Products |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Clinics
Non-Governmental Organizations (NGOs) focused on women's health
Health Insurance Companies
Players Mentioned in the Report:
Bayer AG
Gedeon Richter Plc.
HRA Pharma (Perrigo Company plc)
Teva Pharmaceutical Industries Ltd.
Pfizer Inc.
Mylan N.V. (Viatris Inc.)
Sun Pharmaceutical Industries Ltd.
Cipla Limited
Glenmark Pharmaceuticals Ltd.
Zydus Lifesciences Limited (formerly Zydus Cadila)
HLL Lifecare Limited
Sandoz (Novartis Division / Sandoz Group)
Ferring Pharmaceuticals
Afaxys Pharma LLC
Foundation Consumer Healthcare LLC
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Emergency Contraceptive Pills Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Emergency Contraceptive Pills Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Emergency Contraceptive Pills Market Analysis
3.1 Growth Drivers
3.1.1 Increasing awareness of reproductive health
3.1.2 Rising demand for family planning solutions
3.1.3 Government initiatives promoting contraceptive use
3.1.4 Expansion of pharmacy networks and accessibility
3.2 Market Challenges
3.2.1 Cultural and religious barriers to contraceptive use
3.2.2 Regulatory hurdles in product approval
3.2.3 Limited availability in rural areas
3.2.4 Stigma associated with emergency contraceptive pills
3.3 Market Opportunities
3.3.1 Introduction of new product formulations
3.3.2 Partnerships with healthcare providers
3.3.3 Increasing online sales channels
3.3.4 Educational campaigns targeting young adults
3.4 Market Trends
3.4.1 Growth of telemedicine and online consultations
3.4.2 Shift towards over-the-counter availability
3.4.3 Rising interest in sustainable and organic products
3.4.4 Increased focus on women's health and empowerment
3.5 Government Regulation
3.5.1 Guidelines for emergency contraceptive distribution
3.5.2 Age restrictions on purchase
3.5.3 Labeling and advertising regulations
3.5.4 Approval processes for new products
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Emergency Contraceptive Pills Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Emergency Contraceptive Pills Market Segmentation
8.1 By Active Ingredient
8.1.1 Levonorgestrel-only Pills
8.1.2 Ulipristal Acetate Pills
8.1.3 Combined Estrogen–Progestin Emergency Regimens
8.1.4 Others (e.g., High-dose Progestin, Emerging Molecules)
8.2 By Dosage Regimen
8.2.1 Single-dose Pills
8.2.2 Split-dose / Multiple-dose Regimens
8.2.3 Repeat Use within Cycle
8.2.4 Others
8.3 By Distribution Channel
8.3.1 Retail Pharmacies
8.3.2 Hospital Pharmacies
8.3.3 Online / E-commerce Pharmacies
8.3.4 Clinics & NGO / Community Health Channels
8.4 By Country
8.4.1 Saudi Arabia
8.4.2 United Arab Emirates
8.4.3 Qatar
8.4.4 Kuwait
8.4.5 Bahrain
8.4.6 Oman
8.4.7 Israel
8.4.8 Turkey
8.4.9 Rest of Middle East
8.5 By Indication / Use Case
8.5.1 Unprotected Intercourse
8.5.2 Contraceptive Failure (e.g., Condom Breakage, Missed Pills)
8.5.3 Sexual Assault-related Emergency Contraception
8.5.4 Others
8.6 By Point of Purchase
8.6.1 Over-the-counter (OTC)
8.6.2 Behind-the-counter / Pharmacist-mediated
8.6.3 Prescription-based
8.6.4 NGO / Public-sector Supply
8.7 By Price Tier
8.7.1 Economy / Generic Brands
8.7.2 Mid-range Branded Products
8.7.3 Premium / Imported Brands
8.7.4 Subsidized / Donor-funded Products
9. Middle East Emergency Contraceptive Pills Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Regional Revenue (USD Million, Latest FY)
9.2.3 3-year Revenue CAGR in Middle East (%)
9.2.4 Middle East Emergency EC Pills Market Share (%)
9.2.5 Volume Sold (Units / Packs, Annual)
9.2.6 Average Realized Price per Pack (USD)
9.2.7 Gross Margin (%) on EC Portfolio
9.2.8 R&D Spend on Women’s Health / EC (% of Sales)
9.2.9 Geographic Coverage (Number of Middle East Countries Served)
9.2.10 Channel Mix (% Sales via Retail, Hospital, Online, Public)
9.2.11 Regulatory / Reimbursement Approvals Count in Region
9.2.12 Brand Awareness / Preference Score (Survey-based)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Bayer AG
9.5.2 Gedeon Richter Plc.
9.5.3 HRA Pharma (Perrigo Company plc)
9.5.4 Teva Pharmaceutical Industries Ltd.
9.5.5 Pfizer Inc.
9.5.6 Mylan N.V. (Viatris Inc.)
9.5.7 Sun Pharmaceutical Industries Ltd.
9.5.8 Cipla Limited
9.5.9 Glenmark Pharmaceuticals Ltd.
9.5.10 Zydus Lifesciences Limited (formerly Zydus Cadila)
9.5.11 HLL Lifecare Limited
9.5.12 Sandoz (Novartis Division / Sandoz Group)
9.5.13 Ferring Pharmaceuticals
9.5.14 Afaxys Pharma LLC
9.5.15 Foundation Consumer Healthcare LLC
10. Middle East Emergency Contraceptive Pills Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government procurement policies
10.1.2 Budget allocation for reproductive health
10.1.3 Collaboration with NGOs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for awareness campaigns
10.2.3 Partnerships with private sectors
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Accessibility issues
10.3.2 Affordability concerns
10.3.3 Lack of information
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness levels
10.4.2 Attitudes towards contraceptive use
10.4.3 Availability of products
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of effectiveness
10.5.2 User feedback collection
10.5.3 Expansion into new markets
10.5.4 Others
11. Middle East Emergency Contraceptive Pills Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Business model development
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity planning
15.2.2 Milestone tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health organizations and NGOs focused on reproductive health in the Middle East
- Review of demographic and health surveys to understand contraceptive usage patterns
- Examination of regulatory frameworks and policies regarding emergency contraceptive pills in various Middle Eastern countries
Primary Research
- Interviews with healthcare professionals, including gynecologists and pharmacists, to gather insights on prescribing practices
- Surveys conducted with women of reproductive age to assess awareness and usage of emergency contraceptive pills
- Focus group discussions with community health workers to understand cultural perceptions and barriers to access
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government health statistics and NGO reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews involving reproductive health specialists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on population demographics and reproductive health statistics
- Segmentation of the market by country, considering variations in access and cultural acceptance of emergency contraceptive pills
- Incorporation of trends in contraceptive use and public health initiatives promoting family planning
Bottom-up Modeling
- Collection of sales data from pharmacies and healthcare providers to establish baseline consumption rates
- Estimation of market penetration rates based on awareness and accessibility of emergency contraceptive pills
- Volume x price analysis to determine revenue potential across different market segments
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating variables such as economic conditions, healthcare access, and social attitudes towards contraception
- Scenario modeling based on potential changes in regulations and public health campaigns
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Provider Insights | 120 | Gynecologists, Family Medicine Doctors |
| Pharmacy Distribution Channels | 90 | Pharmacists, Pharmacy Managers |
| Consumer Awareness Surveys | 150 | Women aged 18-45, Health-conscious Individuals |
| Cultural Perception Studies | 80 | Community Health Workers, Social Workers |
| Market Access Evaluations | 110 | Public Health Officials, NGO Representatives |
Frequently Asked Questions
What is the current value of the Middle East Emergency Contraceptive Pills Market?
The Middle East Emergency Contraceptive Pills Market is valued at approximately USD 35 million, reflecting its share within the global emergency contraceptive pills market, which is estimated in the mid-hundreds of millions of USD globally.