Middle East Inhalable Biologics Market Overview
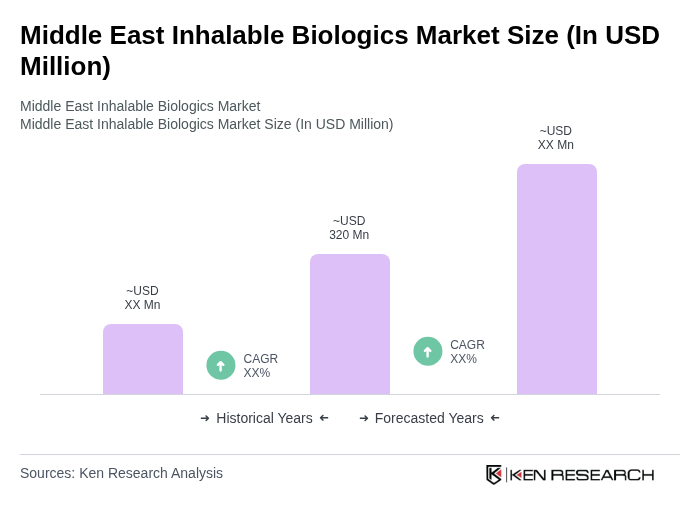
- The Middle East Inhalable Biologics Market is valued at USD 320 million, based on a five-year historical analysis and proportional regional share of the global market. This growth is primarily driven by the increasing prevalence of respiratory diseases, advancements in inhalation technology, and rising healthcare expenditure across the region. The demand for inhalable biologics is further fueled by the growing awareness of the benefits of biologics over traditional therapies, a shift toward non-invasive drug delivery, and the introduction of innovative inhalable monoclonal antibodies, peptides, and RNAi-based therapeutics. Additional drivers include patient preference for self-administration, rapid drug onset via pulmonary delivery, and strategic partnerships among regional healthcare providers and global biopharma companies .
- Countries such as Saudi Arabia, the United Arab Emirates, and Israel dominate the Middle East Inhalable Biologics Market due to their advanced healthcare infrastructure, significant investments in biotechnology, and a high prevalence of chronic respiratory diseases. These nations are also home to leading pharmaceutical companies and research institutions, which contribute to innovation and the development of new inhalable therapies. The UAE, in particular, is experiencing rapid growth in the inhalable biologics segment, driven by government healthcare initiatives and increased adoption of advanced biologic therapies .
- In 2023, the Saudi Food and Drug Authority (SFDA) issued the “Guideline for Registration of Biological Products (Biologics), 2023” to streamline the approval process for inhalable biologics. This regulation, released by the SFDA, establishes requirements for clinical trial data, product quality, and post-marketing surveillance. The guideline aims to expedite regulatory review timelines, enhance patient access to innovative therapies, and ensure safety and efficacy of inhalable biologics in the Saudi market .
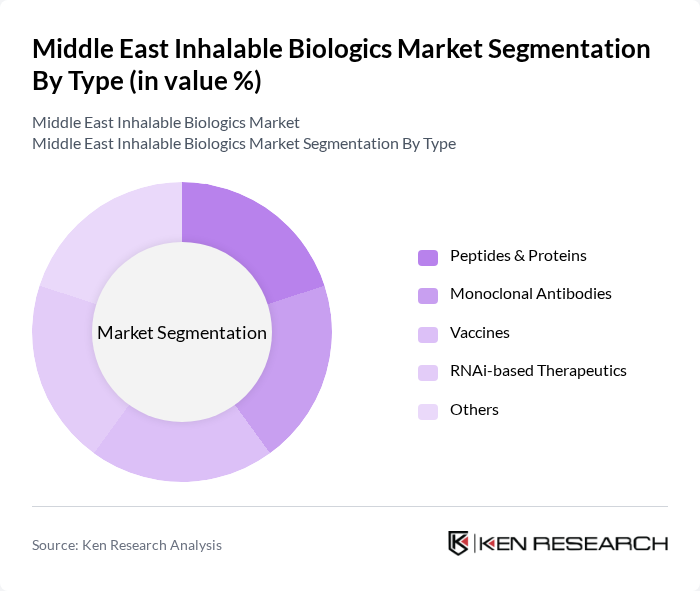
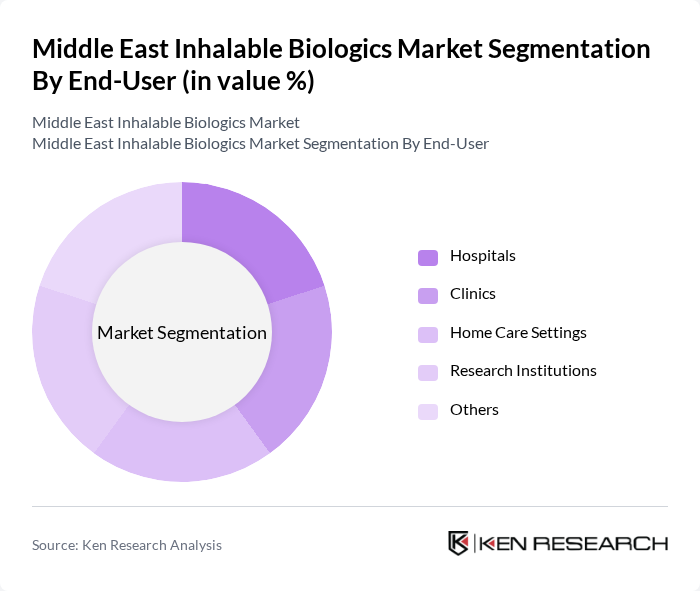
Middle East Inhalable Biologics Market Segmentation
By Type:The market is segmented into various types of inhalable biologics, including Monoclonal Antibodies, Peptides, Proteins, Vaccines, RNAi-based Therapeutics, and Others. Among these, Peptides and Proteins collectively lead the market due to their broad therapeutic applications, improved stability, and efficient pulmonary absorption. Monoclonal antibodies are also gaining traction for their targeted therapeutic effects, particularly in chronic respiratory and immunological conditions .
By End-User:The end-user segmentation includes Hospitals, Clinics, Home Care Settings, Research Institutions, and Others. Hospitals remain the dominant end-user segment, driven by the increasing number of patients requiring inhalable biologics for chronic respiratory conditions and the availability of advanced medical facilities. Clinics and home care settings are witnessing growth due to the rising trend of outpatient and self-administered therapies .
Middle East Inhalable Biologics Market Competitive Landscape
The Middle East Inhalable Biologics Market is characterized by a dynamic mix of regional and international players. Leading participants such as AstraZeneca, Novartis, Sanofi, Boehringer Ingelheim, GSK, Teva Pharmaceutical Industries, Merck & Co., Pfizer, AbbVie, Regeneron Pharmaceuticals, Amgen, Eli Lilly and Company, Roche, Bayer AG, Biogen, MannKind Corporation, Kamada Ltd., Cipla Ltd., Chiesi Farmaceutici S.p.A., Hikma Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Inhalable Biologics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Respiratory Diseases:The Middle East has witnessed a significant rise in respiratory diseases, with the World Health Organization reporting that asthma affects approximately8–10% of the population in countries like Saudi Arabia and the UAE. Additionally, chronic obstructive pulmonary disease (COPD) cases are projected to reacharound 3 millionby the future. This growing health burden drives the demand for inhalable biologics, as they offer targeted treatment options for these conditions, enhancing patient outcomes and quality of life.
- Advancements in Inhalable Drug Delivery Technologies:The region is experiencing rapid advancements in inhalable drug delivery systems, with investments in research and development reachingUSD 1 billionin the future. Innovations such as smart inhalers and nebulizers are improving drug efficacy and patient adherence. These technologies are crucial for delivering biologics effectively, as they enhance bioavailability and minimize systemic side effects, thus fostering a favorable environment for market growth in inhalable biologics.
- Rising Demand for Non-Invasive Treatment Options:The demand for non-invasive treatment options is surging, particularly among patients seeking alternatives to injections. Inhalable biologics provide a less painful and more convenient method of administration. According to a report by the Middle East Health Authority,patient preference for inhalation therapies is increasing. This shift in patient preference is driving pharmaceutical companies to invest in developing inhalable biologics, further propelling market growth.
Market Challenges
- High Development Costs of Inhalable Biologics:The development of inhalable biologics is associated with high costs, often exceedingUSD 500 million per product. This financial burden can deter smaller companies from entering the market, limiting innovation and competition. Furthermore, the lengthy development timelines, which can span over a decade, exacerbate the financial risks involved, making it challenging for firms to justify investments in this sector.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape for inhalable biologics poses significant challenges. The Middle East regulatory bodies, such as the Saudi Food and Drug Authority, have stringent guidelines that can delay product approvals byup to 18 months. Compliance with these regulations requires substantial resources and expertise, which can be particularly challenging for new entrants, thereby hindering market growth and innovation.
Middle East Inhalable Biologics Market Future Outlook
The future of the Middle East inhalable biologics market appears promising, driven by technological advancements and increasing healthcare investments. As healthcare infrastructure expands, particularly in emerging economies, the accessibility of inhalable biologics is expected to improve. Additionally, the integration of digital health technologies will enhance patient engagement and adherence, further propelling market growth. The focus on personalized medicine will also drive innovation, leading to the development of tailored inhalable therapies that meet specific patient needs.
Market Opportunities
- Expansion of Healthcare Infrastructure:The ongoing expansion of healthcare infrastructure in the Middle East, with investments projected to reachUSD 10 billion in future, presents significant opportunities for inhalable biologics. Enhanced facilities and increased access to healthcare services will facilitate the adoption of advanced treatment options, including inhalable therapies, thereby driving market growth.
- Collaborations with Research Institutions:Collaborations between pharmaceutical companies and research institutions are becoming increasingly common, withover 30 partnershipsestablished in the future. These collaborations foster innovation in inhalable biologics, enabling the development of novel therapies and improving the overall market landscape. Such partnerships can accelerate research and development processes, leading to faster product launches and increased market penetration.