Region:Middle East
Author(s):Rebecca
Product Code:KRAD2769
Pages:95
Published On:November 2025
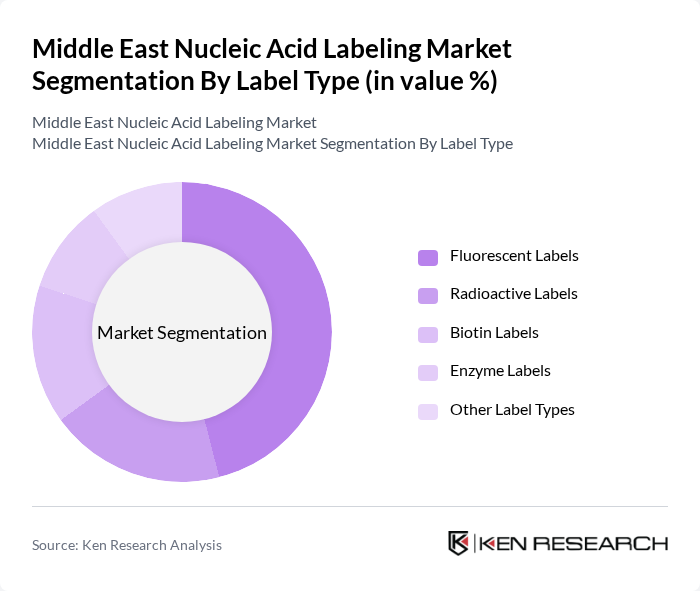
By Label Type:The market is segmented into various label types, including Fluorescent Labels, Radioactive Labels, Biotin Labels, Enzyme Labels, and Other Label Types. Among these, Fluorescent Labels are leading the market due to their high sensitivity, safety, and versatility in applications such as diagnostics and research. The increasing adoption of fluorescence-based techniques in laboratories is driving the demand for this sub-segment. Radioactive Labels, while still relevant, are facing competition from safer and more automation-ready alternatives, resulting in a gradual decline in their market share.
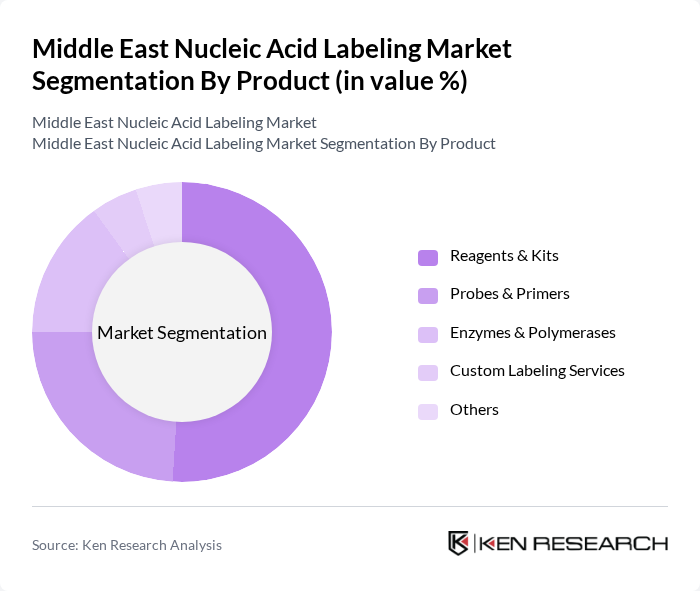
By Product:The product segmentation includes Reagents & Kits, Probes & Primers, Enzymes & Polymerases, Custom Labeling Services, and Others. Reagents & Kits dominate the market due to their essential role in nucleic acid labeling applications, including diagnostics and research. The increasing demand for ready-to-use kits in laboratories is driving this segment's growth. Probes & Primers are also gaining traction, particularly in genetic research and diagnostics, as they are crucial for specific nucleic acid detection.
The Middle East Nucleic Acid Labeling Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific, Roche Diagnostics, Agilent Technologies, Bio-Rad Laboratories, Merck KGaA, QIAGEN, Illumina, Inc., Promega Corporation, New England Biolabs, Takara Bio Inc., Sigma-Aldrich (Merck Group), PerkinElmer, Inc., Bioneer Corporation, Revvity, Inc., Eppendorf AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East nucleic acid labeling market appears promising, driven by ongoing advancements in biotechnology and increased collaboration between public and private sectors. As healthcare systems evolve, the integration of artificial intelligence and automation in laboratory processes is expected to enhance efficiency and accuracy. Furthermore, the growing focus on sustainable practices will likely lead to the development of eco-friendly labeling technologies, aligning with global trends towards environmental responsibility in healthcare.
| Segment | Sub-Segments |
|---|---|
| By Label Type | Fluorescent Labels Radioactive Labels Biotin Labels Enzyme Labels Other Label Types |
| By Product | Reagents & Kits Probes & Primers Enzymes & Polymerases Custom Labeling Services Others |
| By Application | Diagnostics Drug Development Genetic Research Forensic Science Others |
| By Technology | PCR-based Techniques Sequencing Technologies Microarray Technologies CRISPR-based Techniques Others |
| By End-User | Academic & Research Institutions Pharmaceutical & Biotechnology Companies Clinical Laboratories Contract Research Organizations (CROs) Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Pricing Model | Cost-Plus Pricing Value-Based Pricing Competitive Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Diagnostics Laboratories | 100 | Laboratory Directors, Clinical Researchers |
| Academic Research Institutions | 80 | Principal Investigators, Research Scientists |
| Pharmaceutical Companies | 70 | R&D Managers, Product Development Leads |
| Biotechnology Firms | 60 | Business Development Managers, Technical Directors |
| Government Health Agencies | 40 | Policy Makers, Health Program Coordinators |
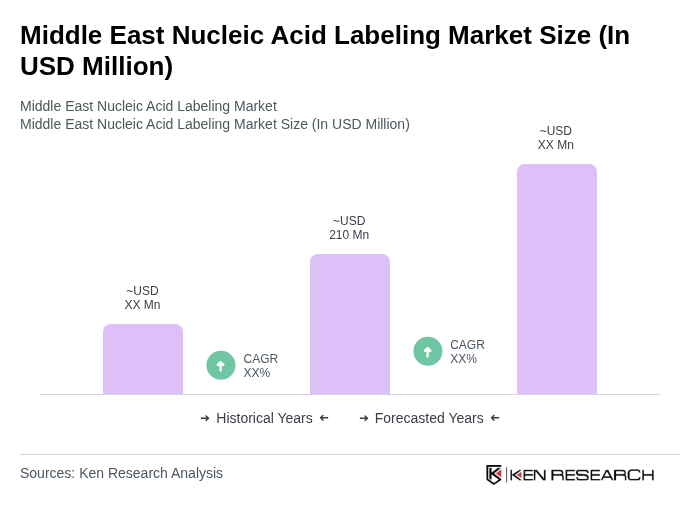
The Middle East Nucleic Acid Labeling Market is valued at approximately USD 210 million, reflecting significant growth driven by advancements in molecular biology techniques and increasing demand for personalized medicine and genetic disorder diagnostics.