About the Report
Base Year 2024New Zealand Sternal Closure Systems Market Overview
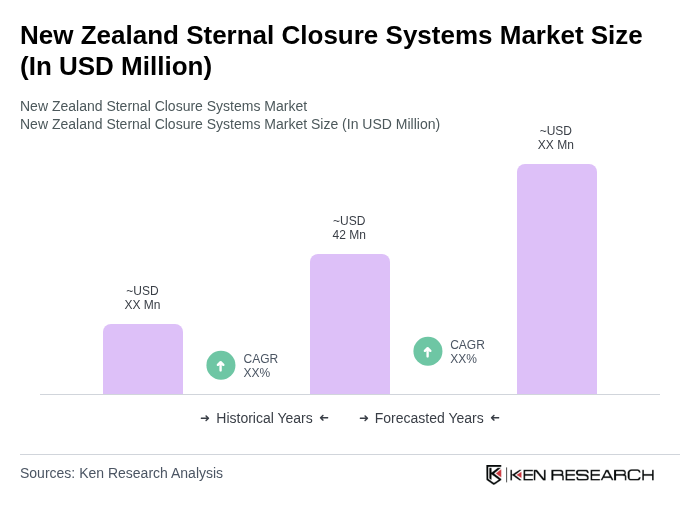
- The New Zealand Sternal Closure Systems Market is valued at USD 42 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cardiovascular diseases and the rising number of cardiac surgeries performed in the country. The demand for effective sternal closure systems is further supported by advancements in surgical techniques and materials, enhancing patient outcomes and recovery times.
- Key cities such as Auckland and Wellington dominate the market due to their advanced healthcare infrastructure and concentration of specialized medical facilities. These urban centers are equipped with state-of-the-art hospitals and surgical centers, attracting both local and international patients seeking cardiac procedures, thereby driving the demand for sternal closure systems.
- The Medicines Act 1981, issued by the Ministry of Health, governs the regulation of therapeutic devices including sternal closure systems. This binding instrument requires all medical devices used in public hospitals to hold registration with Medsafe, meet safety and performance standards under Section 21, and comply with essential principles for design, manufacturing, and clinical evaluation, ensuring only certified high-quality systems are utilized in cardiac surgeries.
New Zealand Sternal Closure Systems Market Segmentation
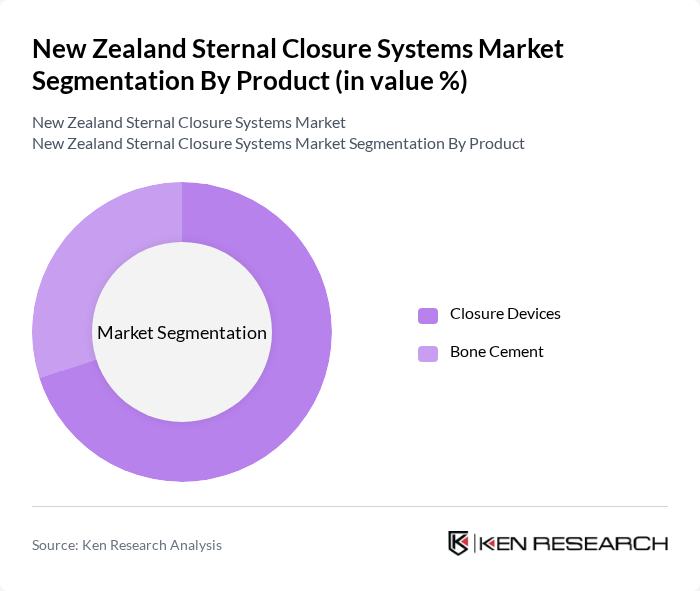
By Product:The product segmentation includes Closure Devices and Bone Cement. Closure devices are essential for securely closing the sternum after cardiac surgery, while bone cement is used to enhance the stability of the closure. The market is witnessing a growing preference for advanced closure devices due to their effectiveness and ease of use, which is driving their demand in surgical settings.
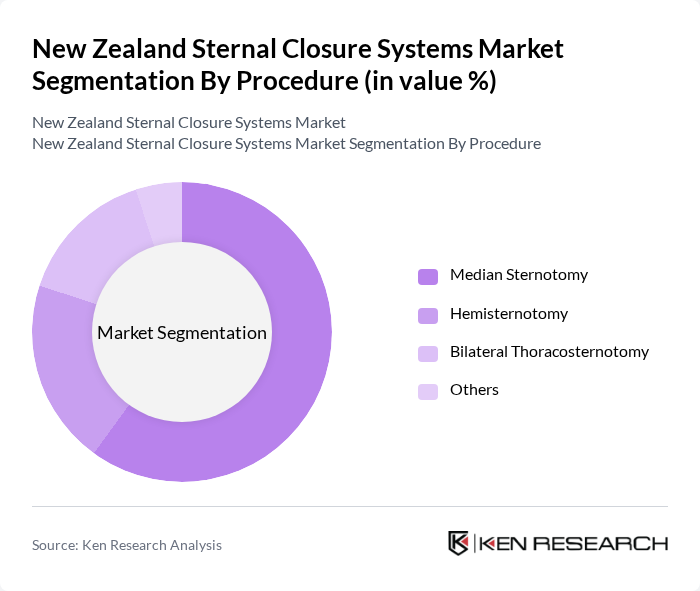
By Procedure:The procedure segmentation encompasses Median Sternotomy, Hemisternotomy, Bilateral Thoracosternotomy, and Others. Median sternotomy is the most commonly performed procedure, accounting for a significant portion of the market due to its widespread use in various cardiac surgeries. The increasing number of surgeries performed using this technique is a key driver for the demand for sternal closure systems.
New Zealand Sternal Closure Systems Market Competitive Landscape
The New Zealand Sternal Closure Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Boston Scientific, Johnson & Johnson, Abbott Laboratories, Stryker Corporation, B. Braun Melsungen AG, Terumo Corporation, Cook Medical, Zimmer Biomet, Conmed Corporation, Aesculap, Inc., Medline Industries, Inc., Integra LifeSciences, Ethicon, Inc., KLS Martin Group contribute to innovation, geographic expansion, and service delivery in this space.
New Zealand Sternal Closure Systems Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:The rise in cardiovascular diseases in New Zealand is significant, with approximately 40,000 hospitalizations annually due to heart-related issues. This growing patient population necessitates effective surgical interventions, driving demand for sternal closure systems. The New Zealand Health Survey indicates that around 1 in 20 adults are diagnosed with heart disease, highlighting the urgent need for advanced surgical solutions to address this health crisis.
- Advancements in Surgical Techniques:Innovations in surgical techniques, such as minimally invasive procedures, are transforming cardiac surgeries. In future, it is estimated that over 60% of cardiac surgeries will utilize advanced techniques, enhancing recovery times and patient outcomes. These advancements are supported by a 15% increase in funding for surgical technology from the New Zealand government, fostering the adoption of modern sternal closure systems that align with these evolving practices.
- Rising Geriatric Population:New Zealand's aging population is projected to reach 1 million individuals aged 65 and over in future, increasing the demand for surgical interventions. Older adults are more susceptible to cardiovascular diseases, necessitating effective sternal closure solutions. The Ministry of Health reports that this demographic will require more than 30% of all cardiac surgeries, further propelling the market for sternal closure systems tailored to geriatric patients' needs.
Market Challenges
- High Cost of Sternal Closure Systems:The financial burden associated with sternal closure systems poses a significant challenge, with costs ranging from NZD 1,500 to NZD 3,000 per procedure. This high expense can limit accessibility for healthcare facilities, particularly in rural areas. The New Zealand government allocates only 6% of its healthcare budget to surgical interventions, which may hinder the adoption of advanced sternal closure technologies in the market.
- Stringent Regulatory Requirements:The regulatory landscape for medical devices in New Zealand is complex, with the Therapeutic Goods Administration enforcing rigorous standards. Compliance with these regulations can delay product launches, impacting market growth. In future, it is estimated that the average time for regulatory approval for new sternal closure systems will exceed 18 months, creating barriers for manufacturers seeking to introduce innovative solutions to the market.
New Zealand Sternal Closure Systems Market Future Outlook
The future of the New Zealand sternal closure systems market appears promising, driven by technological advancements and an increasing focus on patient-centric care. As healthcare providers adopt innovative surgical techniques, the demand for bioresorbable materials and smart technologies is expected to rise. Additionally, the expansion of healthcare infrastructure will facilitate greater access to advanced surgical solutions, ultimately improving patient outcomes and enhancing the overall efficiency of cardiac surgeries in the region.
Market Opportunities
- Expansion of Healthcare Infrastructure:The New Zealand government plans to invest NZD 1 billion in healthcare infrastructure in future, enhancing access to surgical facilities. This investment will create opportunities for sternal closure system manufacturers to collaborate with hospitals and clinics, ensuring that advanced surgical solutions are readily available to meet the growing demand for cardiac procedures.
- Development of Innovative Products:There is a significant opportunity for manufacturers to develop innovative sternal closure systems that incorporate bioresorbable materials. With a projected market growth of 20% for bioresorbable products in the next five years, companies that invest in research and development can capture a substantial share of the market, addressing the evolving needs of healthcare providers and patients alike.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product | Closure Devices Bone Cement |
| By Procedure | Median Sternotomy Hemisternotomy Bilateral Thoracosternotomy Others |
| By Material | Titanium Stainless Steel Polyether Ether Ketone (PEEK) Others |
| By End-User | Hospitals Surgical Centers Others |
| By Region | North Island South Island |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Medsafe, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Wholesalers
Industry Associations (e.g., New Zealand Medical Technology Association)
Financial Institutions
Players Mentioned in the Report:
Medtronic
Boston Scientific
Johnson & Johnson
Abbott Laboratories
Stryker Corporation
B. Braun Melsungen AG
Terumo Corporation
Cook Medical
Zimmer Biomet
Conmed Corporation
Aesculap, Inc.
Medline Industries, Inc.
Integra LifeSciences
Ethicon, Inc.
KLS Martin Group
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. New Zealand Sternal Closure Systems Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 New Zealand Sternal Closure Systems Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. New Zealand Sternal Closure Systems Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Advancements in surgical techniques
3.1.3 Rising geriatric population
3.1.4 Growing demand for minimally invasive procedures
3.2 Market Challenges
3.2.1 High cost of sternal closure systems
3.2.2 Stringent regulatory requirements
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative closure methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in medical technology
3.3.3 Collaborations with research institutions
3.3.4 Development of innovative products
3.4 Market Trends
3.4.1 Shift towards bioresorbable materials
3.4.2 Integration of smart technologies in surgical tools
3.4.3 Focus on patient-centric solutions
3.4.4 Rise in outpatient surgical procedures
3.5 Government Regulation
3.5.1 Medical Device Regulations
3.5.2 Health and Safety Standards
3.5.3 Reimbursement Policies
3.5.4 Clinical Trial Requirements
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. New Zealand Sternal Closure Systems Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. New Zealand Sternal Closure Systems Market Segmentation
8.1 By Product
8.1.1 Closure Devices
8.1.2 Bone Cement
8.2 By Procedure
8.2.1 Median Sternotomy
8.2.2 Hemisternotomy
8.2.3 Bilateral Thoracosternotomy
8.2.4 Others
8.3 By Material
8.3.1 Titanium
8.3.2 Stainless Steel
8.3.3 Polyether Ether Ketone (PEEK)
8.3.4 Others
8.4 By End-User
8.4.1 Hospitals
8.4.2 Surgical Centers
8.4.3 Others
8.5 By Region
8.5.1 North Island
8.5.2 South Island
9. New Zealand Sternal Closure Systems Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Share (%)
9.2.3 Revenue Growth Rate (CAGR %)
9.2.4 EBITDA Margin (%)
9.2.5 R&D Investment (% of Revenue)
9.2.6 Patent Portfolio Size
9.2.7 Global Presence (No. of Countries)
9.2.8 Customer Acquisition Cost
9.2.9 Net Promoter Score (NPS)
9.2.10 Return on Equity (ROE %)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic
9.5.2 Boston Scientific
9.5.3 Johnson & Johnson
9.5.4 Abbott Laboratories
9.5.5 Stryker Corporation
9.5.6 B. Braun Melsungen AG
9.5.7 Terumo Corporation
9.5.8 Cook Medical
9.5.9 Zimmer Biomet
9.5.10 Conmed Corporation
9.5.11 Aesculap, Inc.
9.5.12 Medline Industries, Inc.
9.5.13 Integra LifeSciences
9.5.14 Ethicon, Inc.
9.5.15 KLS Martin Group
10. New Zealand Sternal Closure Systems Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Funding Sources
10.2.3 Project Prioritization
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Procurement
10.3.2 Quality Assurance Issues
10.3.3 Supply Chain Disruptions
10.3.4 Training and Support Needs
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Requirements
10.4.3 Technology Acceptance
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Case Studies
10.5.3 Scalability Potential
10.5.4 Long-term Benefits
11. New Zealand Sternal Closure Systems Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams
1.4 Cost Structure Analysis
1.5 Key Partnerships
1.6 Customer Segments
1.7 Channels
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from New Zealand health authorities and medical device associations
- Review of academic journals and publications focusing on sternal closure technologies
- Examination of market trends and forecasts from healthcare market research firms
Primary Research
- Interviews with cardiac surgeons and thoracic specialists regarding sternal closure preferences
- Surveys with hospital procurement managers to understand purchasing decisions
- Field interviews with medical device sales representatives to gather insights on market dynamics
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from clinical studies, sales data, and expert opinions
- Sanity checks through feedback from a panel of healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total surgical procedures involving sternal closure in New Zealand
- Analysis of demographic data to project patient population growth
- Incorporation of healthcare expenditure trends and government funding for cardiac care
Bottom-up Modeling
- Collection of sales data from leading sternal closure system manufacturers
- Estimation of average selling prices based on product types and market segments
- Volume estimates derived from surgical procedure data and market penetration rates
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating technological advancements and healthcare policies
- Scenario modeling based on potential changes in surgical practices and patient demographics
- Baseline, optimistic, and pessimistic forecasts through 2030 based on market drivers
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiac Surgery Departments | 45 | Cardiac Surgeons, Surgical Coordinators |
| Hospital Procurement Teams | 38 | Procurement Managers, Supply Chain Analysts |
| Medical Device Distributors | 32 | Sales Representatives, Product Managers |
| Healthcare Policy Makers | 28 | Health Economists, Policy Advisors |
| Clinical Research Organizations | 35 | Clinical Researchers, Data Analysts |
Frequently Asked Questions
What is the current value of the New Zealand Sternal Closure Systems Market?
The New Zealand Sternal Closure Systems Market is valued at approximately USD 42 million, reflecting a five-year historical analysis. This growth is driven by the increasing prevalence of cardiovascular diseases and the rising number of cardiac surgeries performed in the country.