Region:Middle East
Author(s):Rebecca
Product Code:KRAA9284
Pages:94
Published On:November 2025
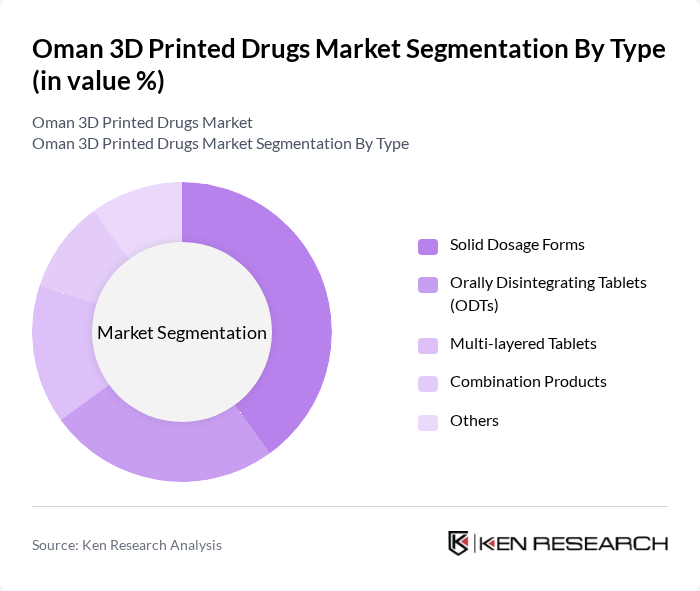
By Type:The market is segmented into solid dosage forms, orally disintegrating tablets (ODTs), multi-layered tablets, combination products, and others. Solid dosage forms are leading due to their established manufacturing processes, stability, and patient familiarity. Orally disintegrating tablets are gaining traction, driven by patient preference for ease of administration and rapid onset of action. Multi-layered tablets and combination products are increasingly adopted for complex therapies requiring controlled release or multi-drug regimens .
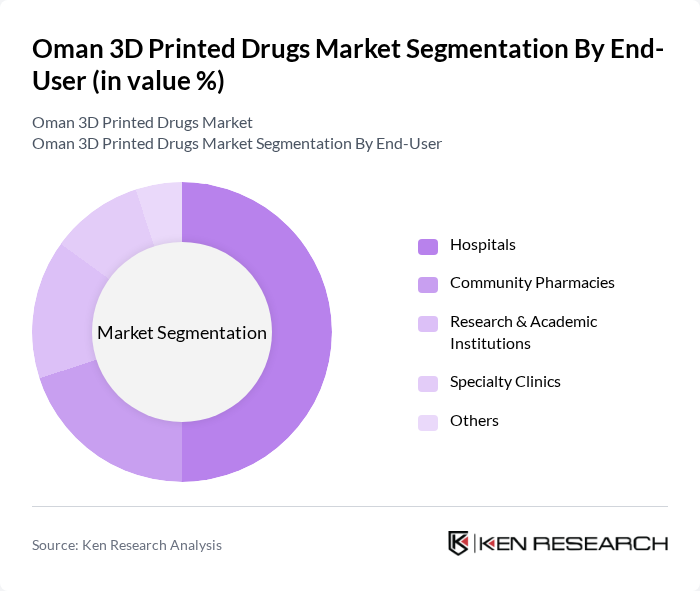
By End-User:The end-user segmentation includes hospitals, community pharmacies, research and academic institutions, specialty clinics, and others. Hospitals are the dominant segment, driven by early adoption of advanced technologies and the need for personalized therapies in critical care and chronic disease management. Community pharmacies are expanding their role in personalized medicine, while research and academic institutions are pivotal in clinical validation and technology transfer. Specialty clinics and other healthcare providers are increasingly integrating 3D printed drugs for niche therapeutic needs .
The Oman 3D Printed Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Aprecia Pharmaceuticals, FabRx Ltd., 3D Bioprinting Solutions, GlaxoSmithKline plc, Merck Group, Novartis AG, Pfizer Inc., Siemens Healthineers AG, Stratasys Ltd., T3D Therapeutics, 3D Systems Corporation, Organovo Holdings, Inc., Medtronic plc, Stryker Corporation, Aspect Biosystems Ltd., Oman Pharmaceutical Products Co. LLC, Oman Medical Devices Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the 3D printed drugs market in Oman appears promising, driven by technological advancements and increasing healthcare demands. As the government continues to support innovation through funding and regulatory improvements, the market is likely to see a surge in new entrants and product offerings. Additionally, the integration of AI in drug development and a focus on sustainable practices will further enhance the market landscape, positioning Oman as a key player in the pharmaceutical industry.
| Segment | Sub-Segments |
|---|---|
| By Type | Solid Dosage Forms Orally Disintegrating Tablets (ODTs) Multi-layered Tablets Combination Products Others |
| By End-User | Hospitals Community Pharmacies Research & Academic Institutions Specialty Clinics Others |
| By Therapeutic Area | Neurological Disorders Oncology Cardiovascular Diseases Infectious Diseases Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Material Used | Pharmaceutical Polymers Cellulose-based Materials Biodegradable Materials Others |
| By Region | Muscat Salalah Sohar Others |
| By Regulatory Compliance | Oman Ministry of Health Standards FDA Compliance EMA Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 45 | Production Managers, R&D Directors |
| Healthcare Providers | 50 | Pharmacists, Physicians |
| Regulatory Bodies | 25 | Regulatory Affairs Specialists, Compliance Officers |
| Patients Using 3D Printed Drugs | 40 | Chronic Disease Patients, General Consumers |
| Industry Experts and Analysts | 30 | Market Analysts, Industry Consultants |
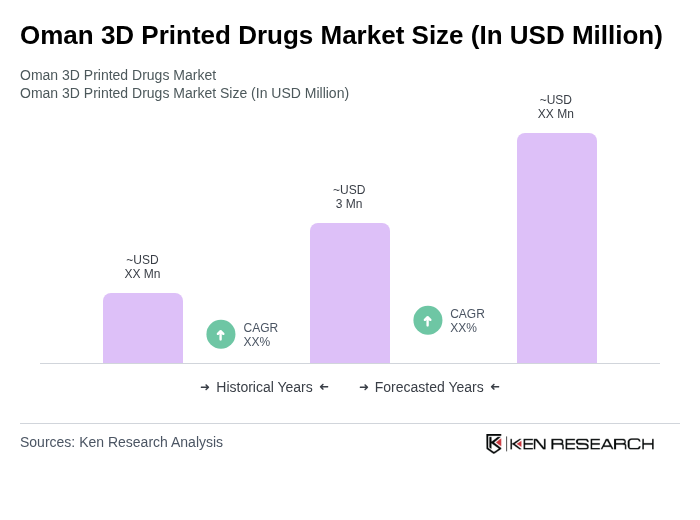
The Oman 3D Printed Drugs Market is valued at approximately USD 3 million, reflecting a growing interest in personalized medicine and advancements in 3D printing technology within the region's healthcare sector.