Region:Middle East
Author(s):Rebecca
Product Code:KRAD1360
Pages:92
Published On:November 2025
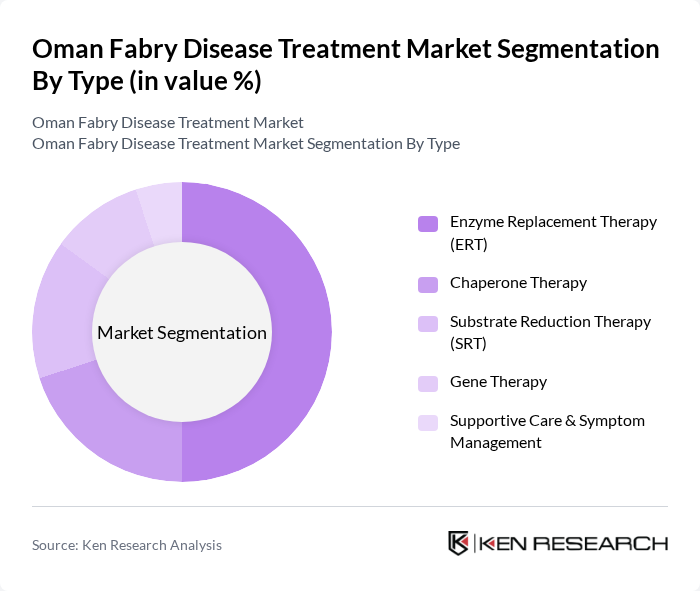
By Type:The market is segmented into various treatment types, including Enzyme Replacement Therapy (ERT), Chaperone Therapy, Substrate Reduction Therapy (SRT), Gene Therapy, and Supportive Care & Symptom Management. Among these, Enzyme Replacement Therapy (ERT) remains the most widely adopted treatment due to its proven efficacy in managing symptoms and improving patient quality of life. The increasing availability of ERT options, such as agalsidase beta and migalastat, and ongoing clinical studies further bolster its dominance in the market .
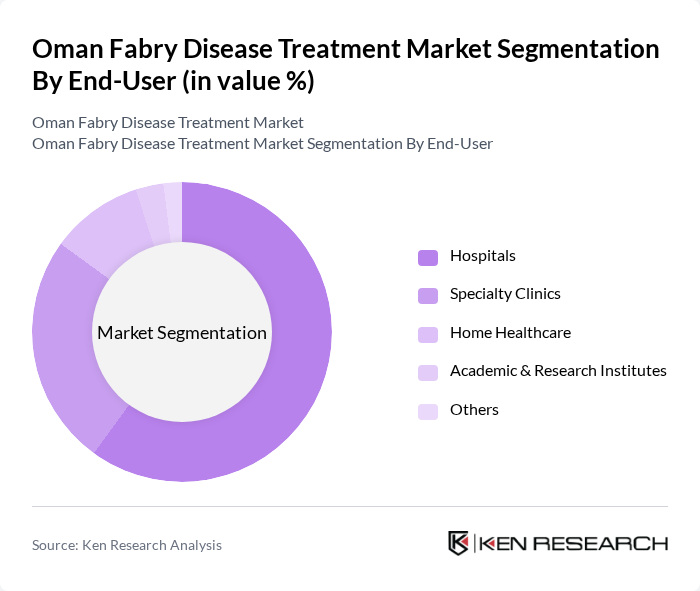
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics, Home Healthcare, Academic & Research Institutes, and Others. Hospitals are the leading end-user segment, primarily due to their comprehensive facilities and specialized staff capable of managing complex cases of Fabry disease. The trend towards integrated care models in hospitals, including multidisciplinary teams and advanced diagnostic labs, enhances patient outcomes and makes hospitals the preferred choice for treatment .
The Oman Fabry Disease Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi Genzyme, Amicus Therapeutics, Takeda Pharmaceutical Company Limited, Pfizer Inc., Idorsia Pharmaceuticals Ltd, Protalix BioTherapeutics, Inc., Avrobio, Inc., Eidos Therapeutics (now part of BridgeBio Pharma), BioMarin Pharmaceutical Inc., H. Lundbeck A/S, Sobi (Swedish Orphan Biovitrum AB), Chiesi Farmaceutici S.p.A., Ultragenyx Pharmaceutical Inc., CANbridge Pharmaceuticals Inc., JCR Pharmaceuticals Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman Fabry disease treatment market appears promising, driven by ongoing advancements in healthcare infrastructure and increased investment in rare disease research. As the government prioritizes healthcare improvements, the establishment of specialized treatment centers is expected to enhance patient access. Furthermore, collaborations with international organizations will likely facilitate the introduction of innovative therapies, ultimately improving patient outcomes and expanding market potential in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme Replacement Therapy (ERT) Chaperone Therapy Substrate Reduction Therapy (SRT) Gene Therapy Supportive Care & Symptom Management |
| By End-User | Hospitals Specialty Clinics Home Healthcare Academic & Research Institutes Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Treatment Setting | Inpatient Treatment Outpatient Treatment Home-Based Treatment Others |
| By Distribution Channel | Direct Sales Online Pharmacies Retail Pharmacies Hospital Pharmacies Others |
| By Geographic Distribution | Urban Areas Rural Areas Semi-Urban Areas Others |
| By Policy Support | Subsidies for Treatment Tax Incentives for Research Grants for Patient Support Programs Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Geneticists, Hematologists, Primary Care Physicians |
| Patient Advocacy Groups | 50 | Patient Advocates, Support Group Leaders |
| Pharmaceutical Stakeholders | 75 | Product Managers, Market Access Specialists |
| Health Insurance Representatives | 60 | Policy Analysts, Claims Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Managers, Compliance Officers |
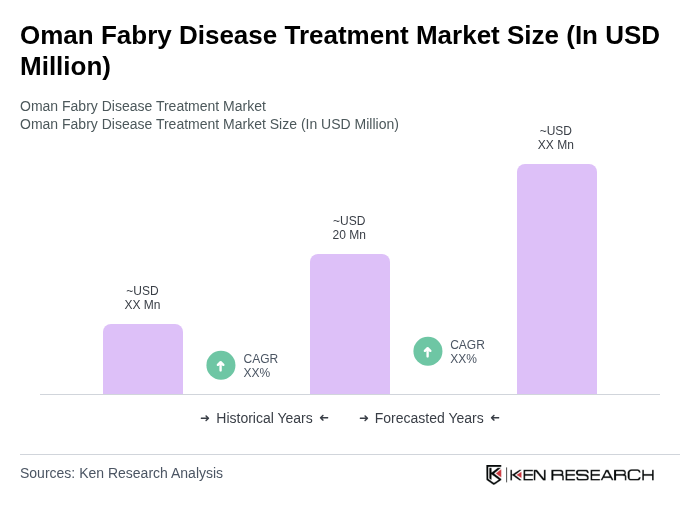
The Oman Fabry Disease Treatment Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This valuation is influenced by factors such as increased awareness of Fabry disease and advancements in treatment options.