Region:Middle East
Author(s):Dev
Product Code:KRAA8210
Pages:96
Published On:November 2025
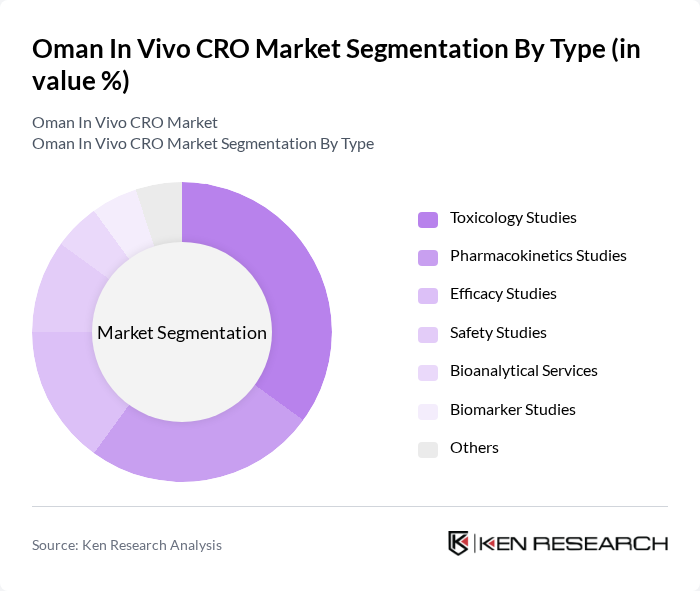
By Type:The market is segmented into various types of services offered by CROs, including toxicology studies, pharmacokinetics studies, efficacy studies, safety studies, bioanalytical services, biomarker studies, and others. Among these, toxicology studies are currently leading the market due to the increasing focus on drug safety and regulatory requirements. The demand for pharmacokinetics studies remains significant, driven by the need for understanding drug absorption, distribution, metabolism, and excretion in clinical settings. The rise of personalized medicine and biologics is also increasing the demand for efficacy and biomarker studies, as sponsors seek more targeted and data-driven approaches to drug development.
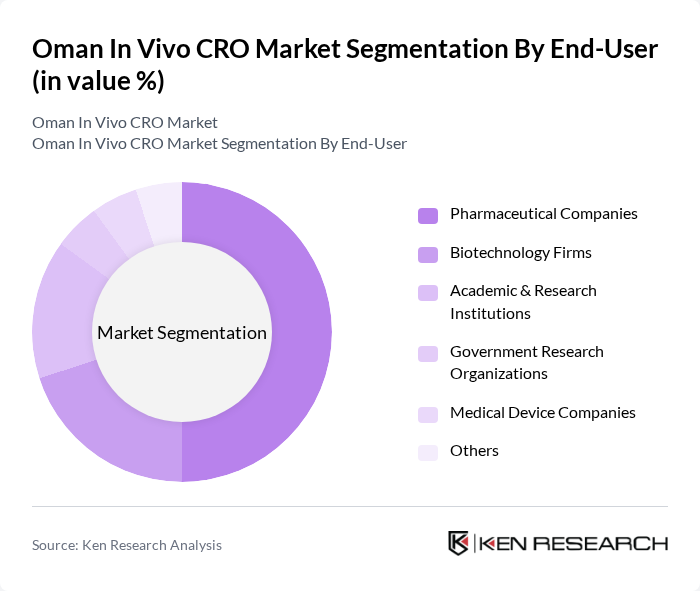
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, academic and research institutions, government research organizations, medical device companies, and others. Pharmaceutical companies dominate this segment as they are the primary clients for CROs, seeking specialized services for drug development and clinical trials. Biotechnology firms are also significant contributors, driven by the need for innovative therapies and personalized medicine. Academic and research institutions are increasingly partnering with CROs for translational research and early-stage studies, while government research organizations and medical device companies utilize CRO expertise to meet regulatory and evidence requirements for new products.
The Oman In Vivo CRO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Synergy CRO, Muscat Life Sciences, Gulf Pharmaceutical Industries (Julphar), Oman Medical Products, Al Nahda International, Muscat Pharmaceuticals, Oman Biotech, Al Jazeera Pharmaceutical, Dhofar Pharmaceutical, Oman Health Services, Al Batinah Pharmaceuticals, Oman Medical Supplies, Aster DM Healthcare, Al Hekma Pharmaceuticals, United Pharmaceuticals, Al Mufeedah Pharmaceuticals, Oman Clinical Research Center, QuintilesIMS (IQVIA Oman), Parexel International Oman, LabCorp Drug Development (Covance Oman) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman in vivo CRO market appears promising, driven by technological advancements and a growing emphasis on personalized medicine. As the healthcare landscape evolves, CROs are expected to adopt innovative methodologies, including digital technologies and data analytics, to enhance research efficiency. Furthermore, collaborations with academic institutions are likely to foster knowledge exchange and accelerate drug development processes, positioning Oman as a key player in the regional CRO ecosystem.
| Segment | Sub-Segments |
|---|---|
| By Type | Toxicology Studies Pharmacokinetics Studies Efficacy Studies Safety Studies Bioanalytical Services Biomarker Studies Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Government Research Organizations Medical Device Companies Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Metabolic Disorders Rare Diseases Others |
| By Study Phase | Preclinical Phase Phase I Phase II Phase III Phase IV Others |
| By Service Type | Full-Service CROs Functional Service Providers (FSPs) Hybrid Models Consulting & Regulatory Services Data Management & Biostatistics Others |
| By Geographic Focus | Local Studies Regional Studies International Studies Others |
| By Client Type | Startups Established Firms Government Entities Multinational Corporations Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 60 | Clinical Research Associates, Regulatory Affairs Managers |
| Biotechnology Research Services | 50 | Biotech Project Managers, R&D Directors |
| Medical Device Testing | 40 | Quality Assurance Managers, Product Development Leads |
| Academic Research Collaborations | 45 | University Researchers, Lab Managers |
| Regulatory Compliance Services | 55 | Compliance Officers, Legal Advisors in Healthcare |
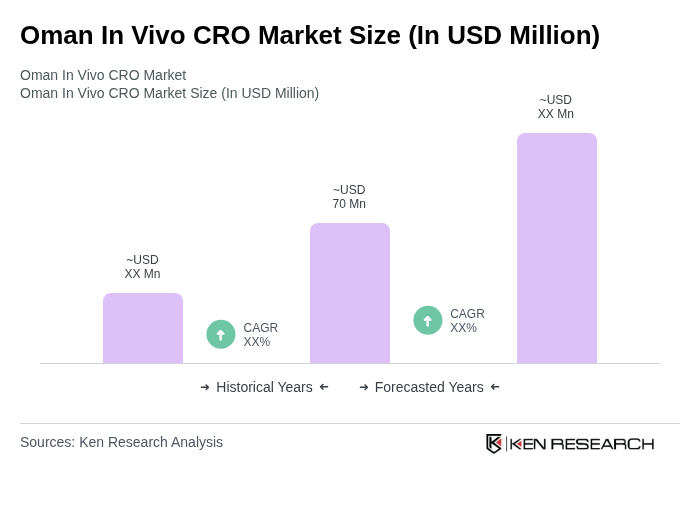
The Oman In Vivo CRO Market is valued at approximately USD 70 million, reflecting a five-year historical analysis. This growth is driven by increasing demand for clinical trials, research services, and investments in healthcare and pharmaceuticals.