Region:Middle East
Author(s):Dev
Product Code:KRAC1995
Pages:82
Published On:October 2025
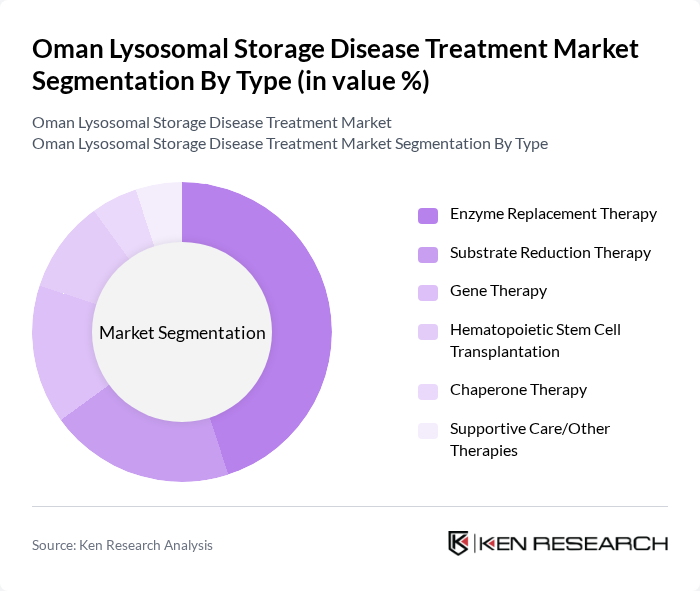
By Type:The market is segmented into various treatment types, including Enzyme Replacement Therapy, Substrate Reduction Therapy, Gene Therapy, Hematopoietic Stem Cell Transplantation, Chaperone Therapy, and Supportive Care/Other Therapies. Among these,Enzyme Replacement Therapyis the most widely adopted due to its proven efficacy and established treatment protocols for several lysosomal storage diseases. The increasing number of patients diagnosed with conditions like Gaucher and Fabry diseases has further propelled the demand for this therapy. Recent trends highlight the growing role of gene therapy and substrate reduction therapy as adjuncts or alternatives for specific patient populations .
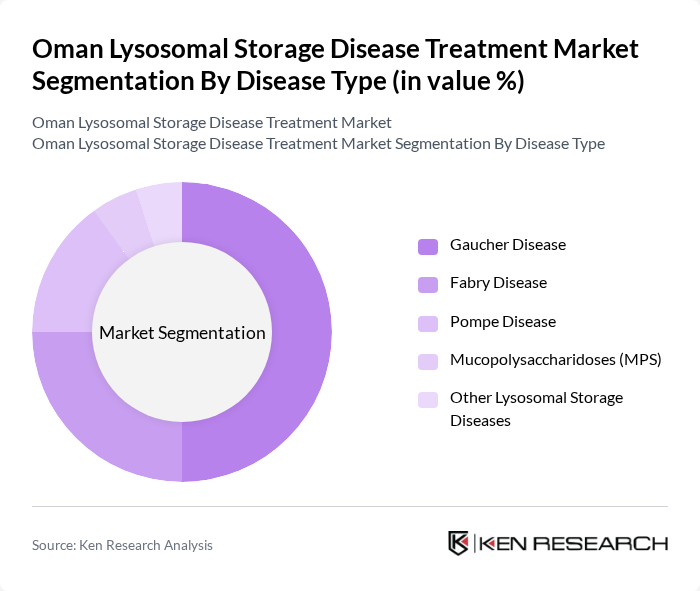
By Disease Type:The market is categorized based on the specific lysosomal storage diseases treated, including Gaucher Disease, Fabry Disease, Pompe Disease, Mucopolysaccharidoses (MPS), and Other Lysosomal Storage Diseases.Gaucher Diseaseleads the market due to its higher prevalence and the availability of effective treatment options, which has resulted in a significant patient population seeking therapy. The increasing awareness and diagnosis of Fabry and Pompe diseases are also contributing to the growth of their respective treatment segments. Enhanced genetic screening and newborn testing initiatives have further supported early diagnosis and intervention .
The Oman Lysosomal Storage Disease Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi (Sanofi Genzyme), Takeda Pharmaceutical Company Limited, BioMarin Pharmaceutical Inc., Amicus Therapeutics, Inc., Orchard Therapeutics plc, Ultragenyx Pharmaceutical Inc., JCR Pharmaceuticals Co., Ltd., Sobi (Swedish Orphan Biovitrum AB), Pfizer Inc., Chiesi Farmaceutici S.p.A., Vertex Pharmaceuticals Incorporated, Eidos Therapeutics, Inc., AVROBIO, Inc., CANbridge Pharmaceuticals Inc., WuXi Biologics contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman lysosomal storage disease treatment market appears promising, driven by ongoing advancements in medical research and technology. The anticipated increase in government funding for rare disease initiatives, projected to reach $12 million in future, will facilitate the development of new therapies. Additionally, the integration of telemedicine services is expected to enhance patient access to specialists, improving diagnosis and treatment adherence. These trends indicate a positive trajectory for the market, fostering innovation and better patient outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme Replacement Therapy Substrate Reduction Therapy Gene Therapy Hematopoietic Stem Cell Transplantation Chaperone Therapy Supportive Care/Other Therapies |
| By Disease Type | Gaucher Disease Fabry Disease Pompe Disease Mucopolysaccharidoses (MPS) Other Lysosomal Storage Diseases |
| By End-User | Hospitals Specialty Clinics Home Healthcare Research Institutions |
| By Distribution Channel | Direct Sales Distributors Online Pharmacies |
| By Patient Demographics | Pediatric Patients Adult Patients |
| By Treatment Setting | Inpatient Outpatient |
| By Geographic Region | Muscat Salalah Sohar Others |
| By Price Range | Low Price Mid Price High Price |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Doctors, Genetic Counselors, Rare Disease Specialists |
| Patient Advocacy Groups | 60 | Patient Representatives, Caregivers, Support Group Leaders |
| Pharmaceutical Companies | 45 | Product Managers, Market Access Specialists |
| Health Insurance Providers | 50 | Policy Analysts, Claims Managers |
| Regulatory Bodies | 40 | Health Policy Makers, Regulatory Affairs Managers |
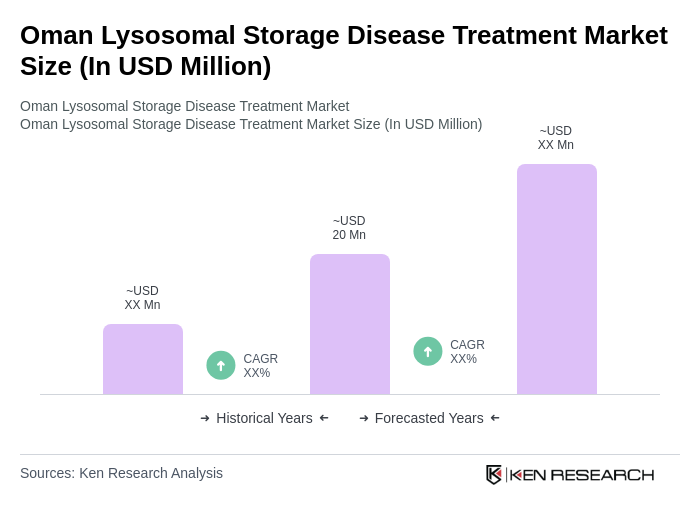
The Oman Lysosomal Storage Disease Treatment Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This growth is attributed to the rising prevalence of lysosomal storage diseases and advancements in treatment options.