Region:Middle East
Author(s):Rebecca
Product Code:KRAD1555
Pages:89
Published On:November 2025
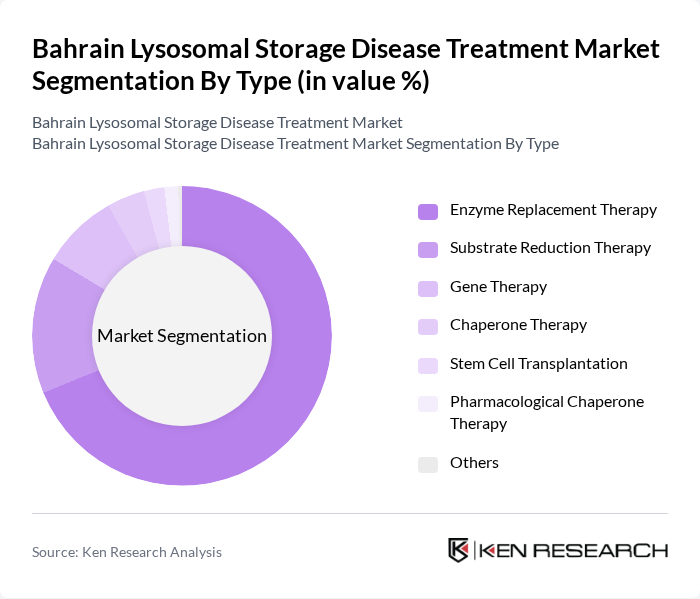
By Type:The market is segmented into various treatment types, each catering to specific needs of patients with lysosomal storage diseases. The dominant sub-segment is Enzyme Replacement Therapy, which is widely recognized for its effectiveness in managing symptoms and improving quality of life for patients. Other therapies, such as Gene Therapy and Substrate Reduction Therapy, are gaining traction due to ongoing research and development efforts aimed at providing more targeted and effective treatment options.
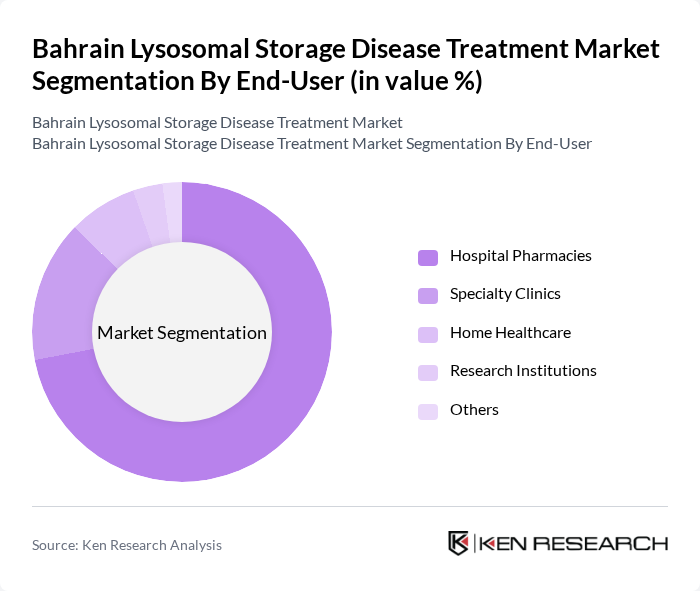
By End-User:The end-user segmentation highlights the various healthcare settings where treatments are administered. Hospital Pharmacies are the leading end-user segment, accounting for the majority of market revenue, primarily due to their capacity to provide comprehensive care and access to specialized medical professionals. Specialty clinics and home healthcare services are also significant, as they cater to patients requiring ongoing management and support for their conditions.
The Bahrain Lysosomal Storage Disease Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi Genzyme, Takeda Pharmaceutical Company Limited, Pfizer Inc., Johnson & Johnson (Janssen/Actelion), BioMarin Pharmaceutical Inc., Amicus Therapeutics, Inc., Orchard Therapeutics plc, Ultragenyx Pharmaceutical Inc., Alexion Pharmaceuticals, Inc. (AstraZeneca Rare Disease), Sobi (Swedish Orphan Biovitrum AB), Horizon Therapeutics plc, Novartis AG, Eli Lilly and Company, Regeneron Pharmaceuticals, Inc., Chiesi Farmaceutici S.p.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain lysosomal storage disease treatment market appears promising, driven by ongoing advancements in medical research and technology. The integration of personalized medicine and genetic testing into treatment plans is expected to enhance patient outcomes significantly. Furthermore, the rise of telemedicine solutions will facilitate better access to specialists, particularly in remote areas. As government funding continues to support healthcare infrastructure, the market is poised for growth, addressing both patient needs and treatment accessibility challenges.
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme Replacement Therapy Substrate Reduction Therapy Gene Therapy Chaperone Therapy Stem Cell Transplantation Pharmacological Chaperone Therapy Others |
| By End-User | Hospitals Specialty Clinics Home Healthcare Research Institutions Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Treatment Setting | Inpatient Treatment Outpatient Treatment Home-Based Treatment Others |
| By Distribution Channel | Direct Sales Online Pharmacies Retail Pharmacies Hospital Pharmacies Others |
| By Geography | Northern Governorate Southern Governorate Capital Governorate Muharraq Governorate Others |
| By Research and Development Focus | Clinical Trials Drug Development Patient Registries Real-World Evidence Studies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 90 | Geneticists, Pediatricians, Rare Disease Specialists |
| Patient Advocacy Groups | 60 | Leaders of Patient Organizations, Caregivers |
| Pharmaceutical Distributors | 40 | Sales Representatives, Market Access Managers |
| Health Insurance Companies | 50 | Policy Analysts, Claims Managers |
| Regulatory Bodies | 40 | Health Policy Makers, Regulatory Affairs Specialists |
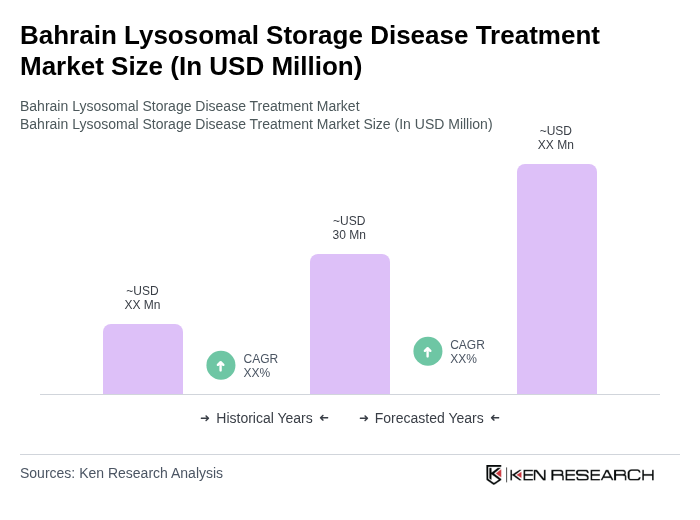
The Bahrain Lysosomal Storage Disease Treatment Market is valued at approximately USD 30 million, reflecting a five-year historical analysis. This growth is attributed to increased awareness, advancements in treatment options, and a rising prevalence of genetic disorders in the region.