Region:Middle East
Author(s):Dev
Product Code:KRAA8322
Pages:98
Published On:November 2025
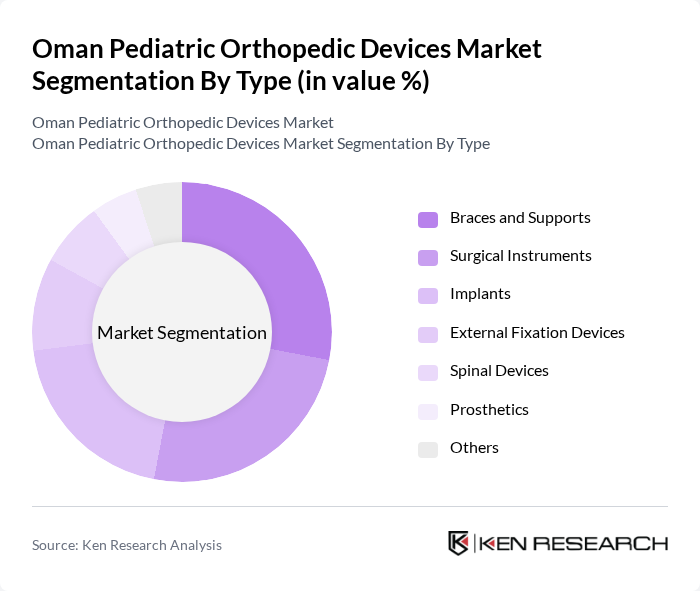
By Type:The market is segmented into several types of pediatric orthopedic devices, including braces and supports, surgical instruments, implants, external fixation devices, spinal devices, prosthetics, and others. Braces and supports are widely used for non-invasive treatment and post-operative rehabilitation in conditions such as scoliosis and juvenile arthritis. Surgical instruments and implants are essential for corrective procedures addressing fractures, deformities, and limb length discrepancies. External fixation devices and spinal devices cater to complex trauma and spinal deformities, while prosthetics and other devices address congenital limb deficiencies and rehabilitation needs.
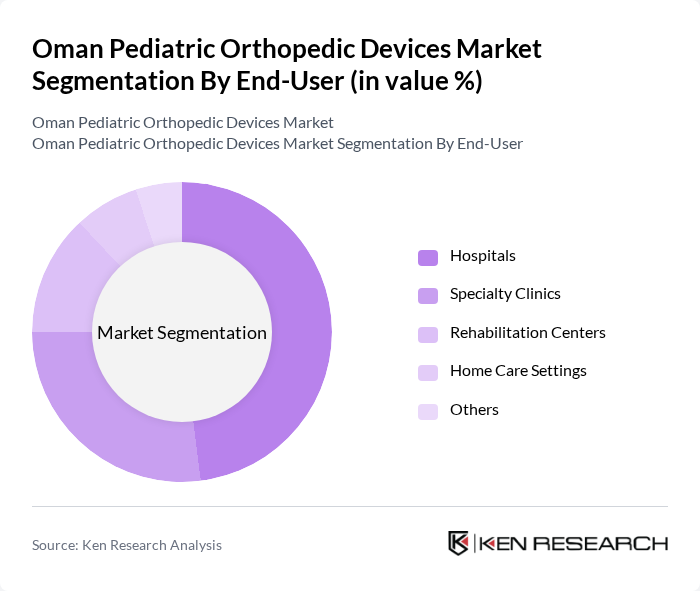
By End-User:The end-user segmentation includes hospitals, specialty clinics, rehabilitation centers, home care settings, and others. Hospitals represent the largest segment due to their comprehensive facilities, advanced surgical capabilities, and access to multidisciplinary pediatric teams. Specialty clinics and rehabilitation centers play a significant role in providing targeted orthopedic care and post-surgical rehabilitation, while home care settings and other providers support ongoing management and recovery for pediatric patients.
The Oman Pediatric Orthopedic Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stryker Corporation, DePuy Synthes (Johnson & Johnson), Zimmer Biomet Holdings, Inc., Medtronic plc, Smith & Nephew plc, Orthofix Medical Inc., NuVasive, Inc., Amedica Corporation (now SINTX Technologies, Inc.), Arthrex, Inc., K2M Group Holdings, Inc. (now part of Stryker), Wright Medical Group N.V. (now part of Stryker), CONMED Corporation, Bioventus LLC, Exactech, Inc., Medacta International SA contribute to innovation, geographic expansion, and service delivery in this space.
The future of the pediatric orthopedic devices market in Oman appears promising, driven by ongoing technological advancements and increased government support. As healthcare expenditure rises, investments in pediatric orthopedic services are expected to expand, improving access to specialized care. Additionally, the integration of telemedicine and digital health solutions will likely enhance patient consultations and follow-ups, making orthopedic care more accessible. These trends indicate a positive trajectory for the market, fostering innovation and improved patient outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Braces and Supports Surgical Instruments Implants External Fixation Devices Spinal Devices Prosthetics Others |
| By End-User | Hospitals Specialty Clinics Rehabilitation Centers Home Care Settings Others |
| By Age Group | Infants (0-1 years) Toddlers (1-3 years) Preschoolers (3-5 years) School-age Children (6-12 years) Adolescents (13-18 years) |
| By Condition | Congenital Disorders (e.g., clubfoot, hip dysplasia) Trauma and Injuries (fractures, dislocations) Neuromuscular Disorders (e.g., cerebral palsy) Skeletal Dysplasia Sports Injuries Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Retail Pharmacies Others |
| By Material | Metal Polymer/Plastic Composite Materials Bioabsorbable Materials Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Orthopedic Surgeons | 60 | Orthopedic Surgeons, Pediatric Specialists |
| Healthcare Facility Procurement Managers | 50 | Procurement Managers, Supply Chain Directors |
| Parents of Pediatric Patients | 40 | Parents, Caregivers |
| Medical Device Manufacturers | 40 | Product Managers, Sales Representatives |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
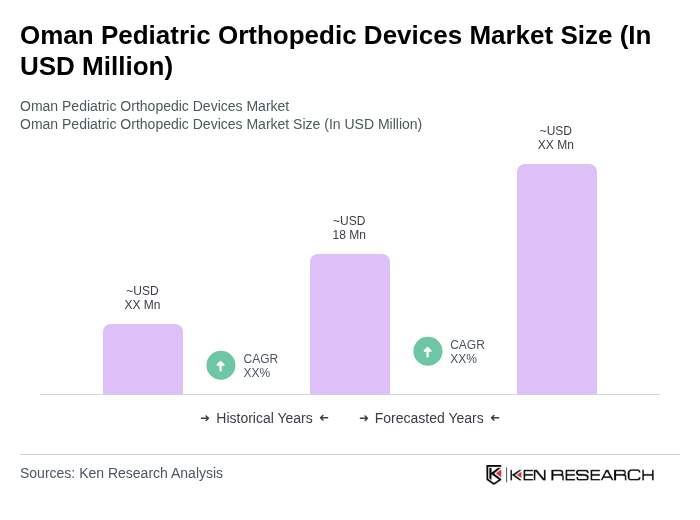
The Oman Pediatric Orthopedic Devices Market is valued at approximately USD 18 million, reflecting a significant growth driven by the rising prevalence of pediatric musculoskeletal disorders and advancements in orthopedic technology.