Region:Middle East
Author(s):Geetanshi
Product Code:KRAA3491
Pages:80
Published On:January 2026
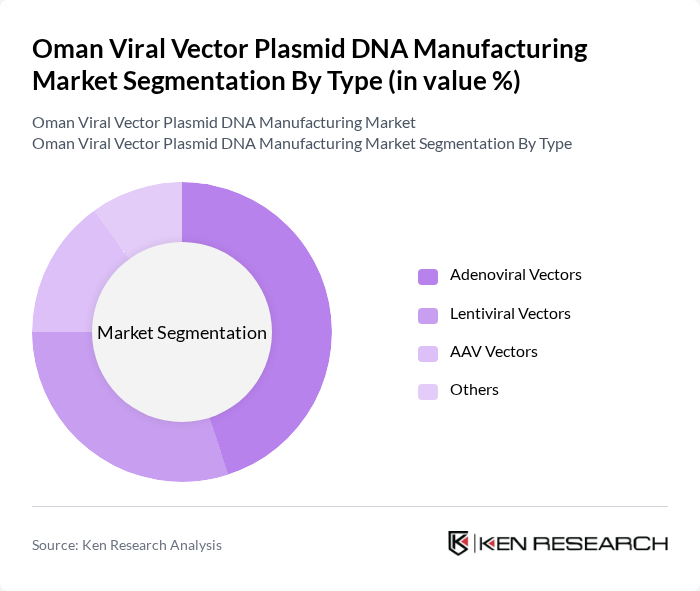
By Type:The market is segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV Vectors, and Others. Adenoviral vectors are widely used due to their high transduction efficiency and ability to accommodate large genetic payloads, making them suitable for various therapeutic applications. Lentiviral vectors are also gaining traction, particularly in gene therapy for chronic diseases, while AAV vectors are favored for their safety profile and long-term expression capabilities.
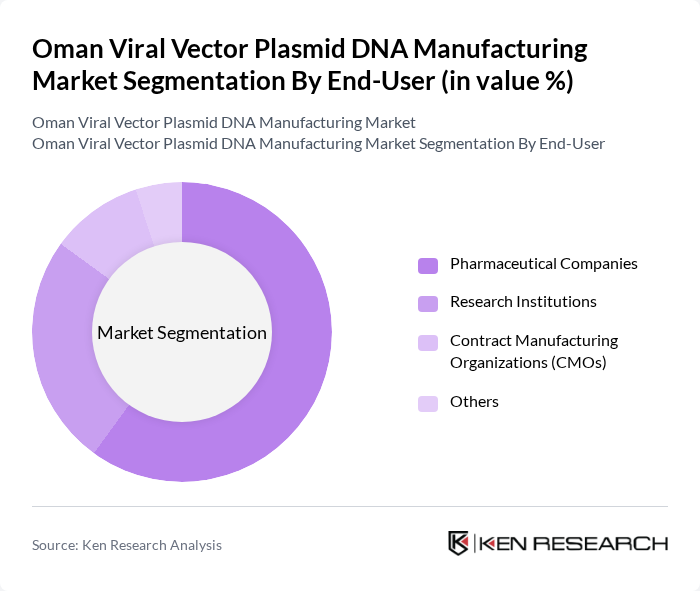
By End-User:The end-user segmentation includes Pharmaceutical Companies, Research Institutions, Biotechnology Firms, and Others. Pharmaceutical companies dominate the market due to their extensive resources and capabilities in developing and commercializing viral vector-based therapies. Research institutions play a crucial role in advancing scientific knowledge and innovation, while biotechnology firms contribute to the development of specialized applications and technologies.
The Oman Viral Vector Plasmid DNA Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oxford Biomedica, Novartis, GSK, Merck KGaA, WuXi AppTec, Catalent, Lonza, AAVLife, GenScript, Synlogic, Cellectis, Bluebird Bio, Amgen, Sangamo Therapeutics, Kite Pharma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the viral vector plasmid DNA manufacturing market in Oman appears promising, driven by increasing investments in biotechnology and a growing focus on personalized medicine. As the demand for gene therapies rises, manufacturers are likely to adopt advanced biomanufacturing technologies and automation to enhance efficiency. Additionally, collaborations between industry players and academic institutions will foster innovation, ensuring that Oman remains competitive in the global biotechnology landscape while addressing local healthcare needs effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV Vectors Others |
| By End-User | Pharmaceutical Companies Research Institutions Biotechnology Firms Others |
| By Application | Gene Therapy Vaccine Development Cell Therapy Others |
| By Production Scale | Small Scale Medium Scale Large Scale Others |
| By Region | Muscat Salalah Sohar Others |
| By Technology | Transfection Electroporation Viral Infection Others |
| By Investment Source | Private Investments Government Funding International Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotechnology Firms in Oman | 100 | R&D Managers, Product Development Leads |
| Pharmaceutical Companies | 80 | Regulatory Affairs Specialists, Quality Control Managers |
| Academic Institutions | 60 | Research Professors, Lab Directors |
| Healthcare Providers | 70 | Clinical Researchers, Medical Directors |
| Government Health Agencies | 50 | Policy Makers, Health Program Managers |
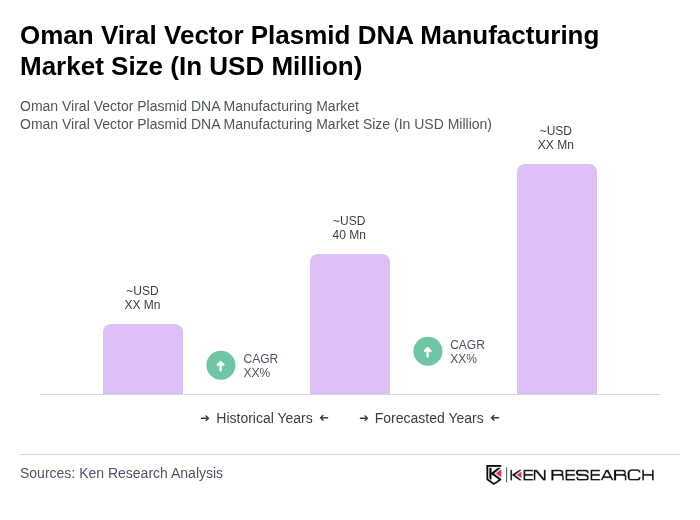
The Oman Viral Vector Plasmid DNA Manufacturing Market is valued at approximately USD 50 million, reflecting a robust growth driven by the increasing demand for gene therapies and vaccines, as well as advancements in biomanufacturing technologies.