Region:Middle East
Author(s):Rebecca
Product Code:KRAD2397
Pages:95
Published On:January 2026
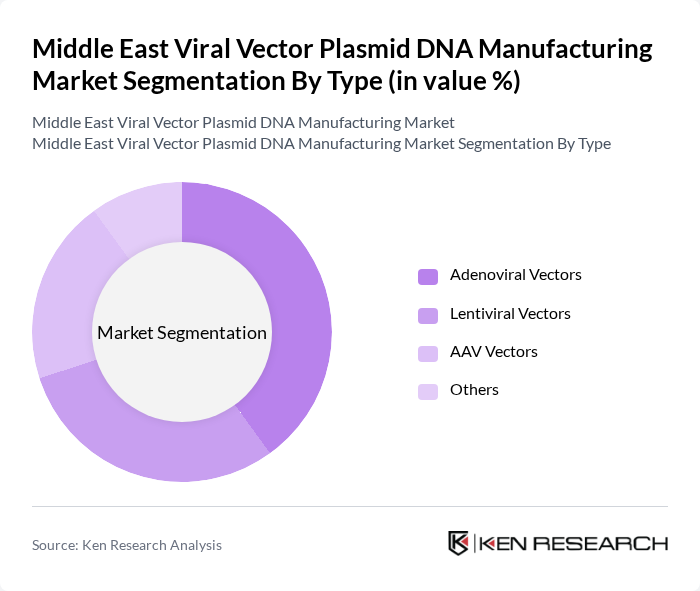
By Type:The market is segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV Vectors, and Others. Among these, Lentiviral Vectors hold a noticeable share due to their high transduction efficiency and ability to deliver large genetic payloads. This makes them particularly suitable for applications in gene therapy and vaccine development. The increasing focus on personalized medicine and the growing number of clinical trials utilizing these vectors are further driving their dominance.
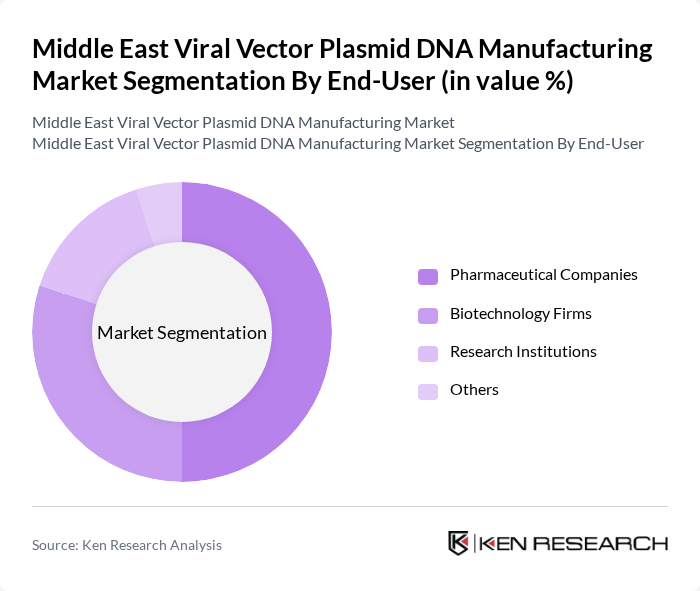
By End-User:The end-user segmentation includes Pharmaceutical Companies, Research Institutions, Contract Research Organizations (CROs), and Others. Pharmaceutical Companies are the dominant end-users in this market, driven by their extensive R&D capabilities and the need for advanced therapeutic solutions. The increasing collaboration between pharmaceutical firms and research institutions to develop innovative therapies is also contributing to the growth of this segment.
The Middle East Viral Vector Plasmid DNA Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group, WuXi AppTec, Catalent, Inc., ACGT, Inc., Oxford Biomedica, Virovek, GenScript, Sarepta Therapeutics, Amgen, Novartis, Merck KGaA, BioNTech, GSK, Takeda Pharmaceutical Company, Pfizer contribute to innovation, geographic expansion, and service delivery in this space.
The future outlook for the Middle East viral vector plasmid DNA manufacturing market appears promising, driven by ongoing advancements in biotechnology and increasing collaboration between public and private sectors. As the region continues to invest in research and development, the integration of digital technologies and automation is expected to enhance production efficiency. Furthermore, the growing focus on personalized medicine will likely create new avenues for innovation, positioning the Middle East as a competitive player in the global market landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV Vectors Others |
| By End-User | Pharmaceutical Companies Research Institutions Contract Research Organizations (CROs) Others |
| By Application | Gene Therapy Vaccine Development Cell Therapy Others |
| By Delivery Method | In Vivo Ex Vivo Others |
| By Region | GCC Countries Levant Region North Africa |
| By Manufacturing Process | Plasmid DNA Production Vector Production Purification Others |
| By Regulatory Compliance | FDA Compliance EMA Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Applications | 100 | Clinical Researchers, Biotech Executives |
| Vaccine Development Processes | 80 | Pharmaceutical Scientists, Regulatory Affairs Managers |
| Manufacturing Technologies | 70 | Production Managers, Quality Control Specialists |
| Market Entry Strategies | 60 | Business Development Managers, Market Analysts |
| Investment Trends in Biotechnology | 90 | Venture Capitalists, Financial Analysts |
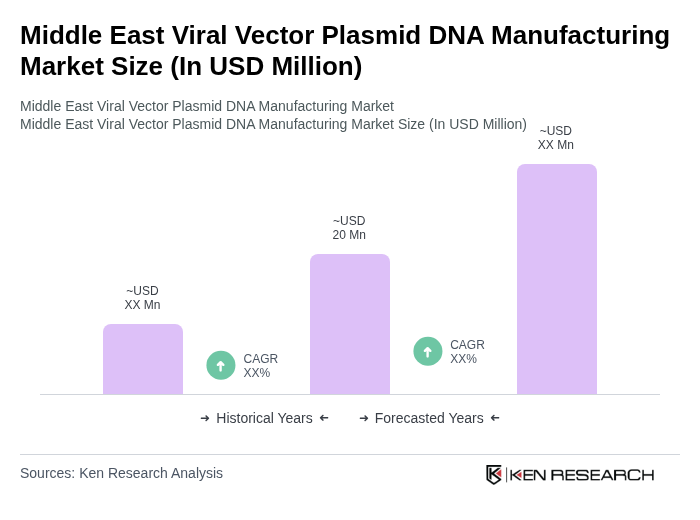
The Middle East Viral Vector Plasmid DNA Manufacturing Market is valued at approximately USD 20 million, driven by increasing demand for gene therapies and vaccines, alongside significant investments in biotechnology and pharmaceuticals in the region.