Region:Asia
Author(s):Dev
Product Code:KRAC8697
Pages:94
Published On:November 2025
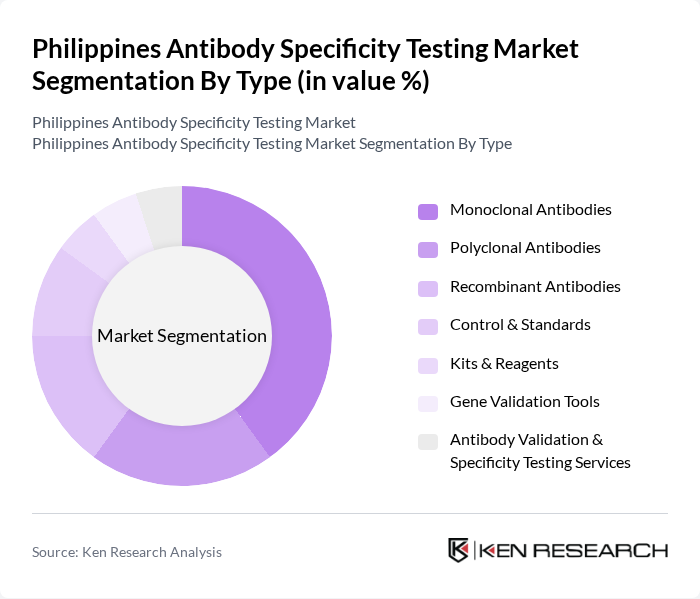
By Type:The market is segmented into Monoclonal Antibodies, Polyclonal Antibodies, Recombinant Antibodies, Control & Standards, Kits & Reagents, Gene Validation Tools, and Antibody Validation & Specificity Testing Services. Monoclonal Antibodies lead the market due to their superior specificity, reproducibility, and effectiveness in both diagnostic and research applications. The adoption of pre-validated kits and integration with automated platforms is enhancing efficiency and accuracy in antibody validation workflows .
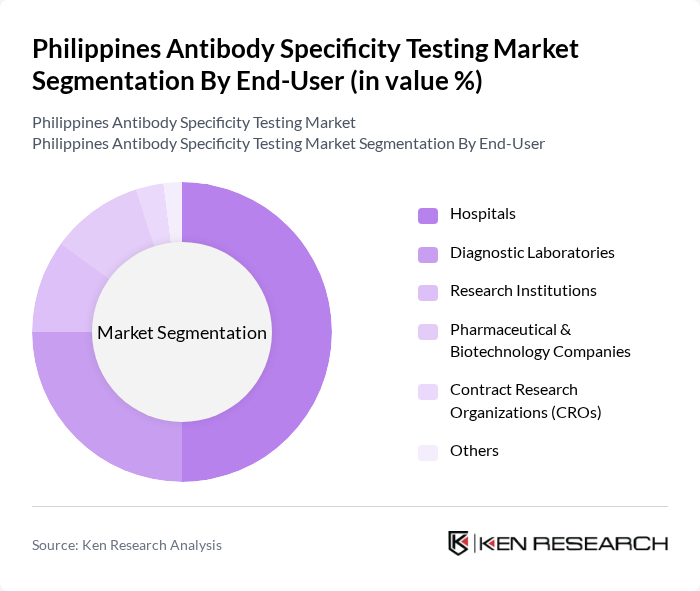
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Others. Hospitals are the leading end-users, reflecting their central role in patient diagnosis and treatment. The increasing adoption of antibody-based diagnostics in hospital laboratories and the expansion of research collaborations with pharmaceutical and biotechnology companies are further driving demand for antibody specificity testing services .
The Philippines Antibody Specificity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Merck KGaA, GenScript Biotech Corporation, Becton, Dickinson and Company (BD), Ortho Clinical Diagnostics, Fujirebio, PerkinElmer, QuidelOrtho Corporation, DiaSorin S.p.A., Hologic, Inc., and Grifols, S.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the antibody specificity testing market in the Philippines appears promising, driven by ongoing technological advancements and increasing healthcare investments. As the government continues to enhance healthcare infrastructure, the accessibility of testing is expected to improve. Additionally, the integration of artificial intelligence in diagnostics will likely streamline processes, making testing more efficient. These developments will foster a more robust healthcare environment, ultimately benefiting patient outcomes and driving market growth.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Polyclonal Antibodies Recombinant Antibodies Control & Standards Kits & Reagents Gene Validation Tools Antibody Validation & Specificity Testing Services |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
| By Application | Infectious Disease Testing Autoimmune Disease Testing Cancer Diagnostics Research & Development Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Luzon Visayas Mindanao |
| By Technology | ELISA Western Blotting Immunofluorescence Multiplex Assays Automated Immunoassay Platforms Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 100 | Laboratory Managers, Clinical Pathologists |
| Diagnostic Centers | 60 | Operations Managers, Medical Technologists |
| Research Institutions | 40 | Research Scientists, Immunologists |
| Healthcare Providers | 75 | General Practitioners, Infectious Disease Specialists |
| Regulatory Bodies | 40 | Policy Makers, Health Regulators |
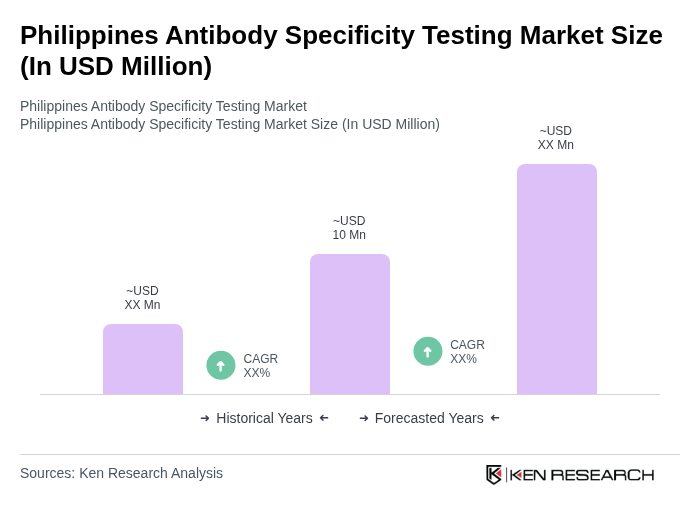
The Philippines Antibody Specificity Testing Market is valued at approximately USD 10 million, reflecting its share within the rapidly growing Asia Pacific region, driven by advancements in biopharmaceutical research and increased demand for diagnostic solutions.