Region:Asia
Author(s):Shubham
Product Code:KRAC2260
Pages:83
Published On:October 2025
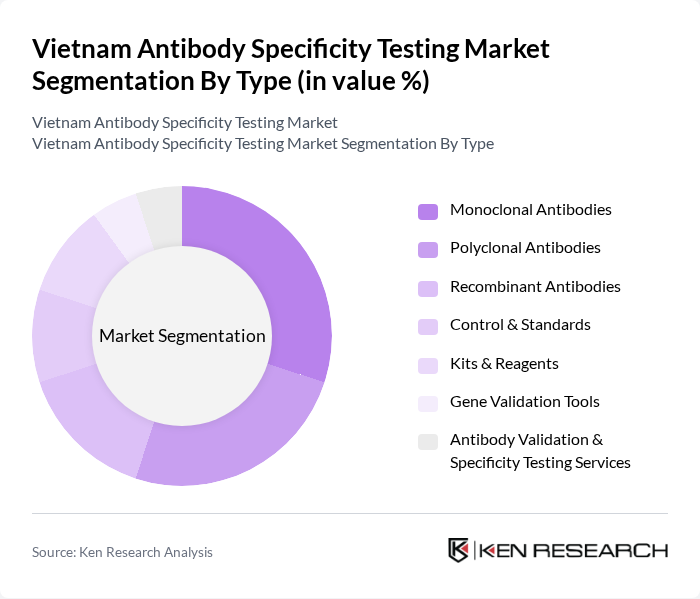
By Type:The market is segmented into various types, including Monoclonal Antibodies, Polyclonal Antibodies, Recombinant Antibodies, Control & Standards, Kits & Reagents, Gene Validation Tools, and Antibody Validation & Specificity Testing Services. Each of these subsegments plays a crucial role in the overall market dynamics, with monoclonal and polyclonal antibodies dominating due to their widespread use in diagnostics and therapeutics. Recombinant antibodies are gaining traction for their batch-to-batch consistency and specificity, while kits and reagents support standardized workflows in both research and clinical laboratories. Control & standards and gene validation tools ensure the reliability and reproducibility of testing outcomes, and specialized services address complex validation requirements for novel antibody products .
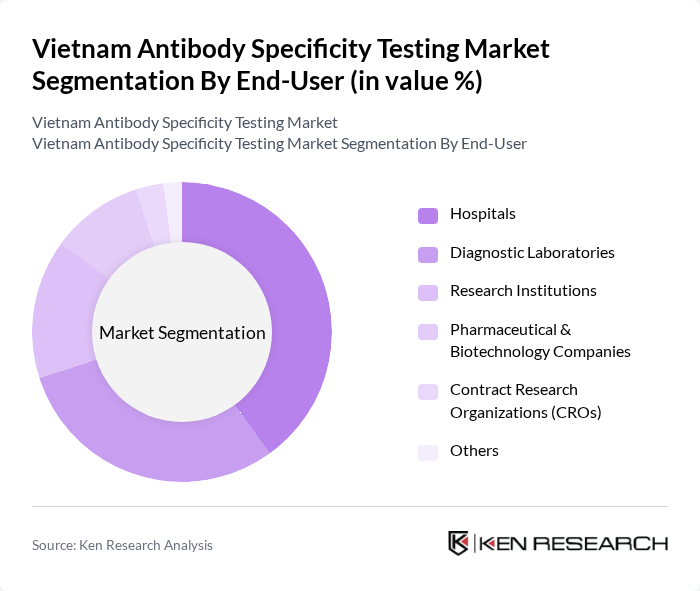
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Others. Hospitals and diagnostic laboratories account for the largest share, driven by the need for accurate and timely diagnostics. Research institutions and pharmaceutical/biotechnology companies are significant contributors due to ongoing R&D in antibody-based therapeutics and diagnostics. CROs and other specialized entities support outsourced testing and validation services, reflecting the increasing complexity and regulatory requirements in antibody development .
The Vietnam Antibody Specificity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Merck KGaA, Becton, Dickinson and Company, GenScript Biotech Corporation, PerkinElmer, Inc., Abcam plc, QIAGEN N.V., Agilent Technologies, Inc., Fujifilm Wako Pure Chemical Corporation, VWR International, LLC (Avantor), SeraCare Life Sciences, Inc. (LGC Clinical Diagnostics), BioSystems Company Limited (Vietnam), Mediplantex National Pharmaceutical Joint Stock Company (Vietnam), Viet A Corporation (Vietnam) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam antibody specificity testing market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The government’s commitment to enhancing healthcare infrastructure, with an expected $1 billion investment in future, will facilitate better access to diagnostic tools. Additionally, the integration of artificial intelligence in testing processes is anticipated to streamline operations and improve accuracy, further propelling market growth. As awareness increases, the market is likely to expand significantly in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Polyclonal Antibodies Recombinant Antibodies Control & Standards Kits & Reagents Gene Validation Tools Antibody Validation & Specificity Testing Services |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
| By Application | Disease Diagnosis Drug Development Clinical Research Therapeutic Antibody Development Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Northern Vietnam Central Vietnam Southern Vietnam Others |
| By Price Range | Low Price Mid Price High Price |
| By Technology | Immunoassay-based Technologies (ELISA, etc.) Western Blotting Flow Cytometry Mass Spectrometry Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Diagnostic Laboratories | 60 | Laboratory Managers, Quality Control Officers |
| Healthcare Providers | 50 | Physicians, Nurse Practitioners |
| Pharmaceutical Companies | 40 | Product Managers, Regulatory Affairs Specialists |
| Public Health Officials | 40 | Health Policy Advisors, Epidemiologists |
| Biotechnology Firms | 45 | Research Scientists, Business Development Managers |
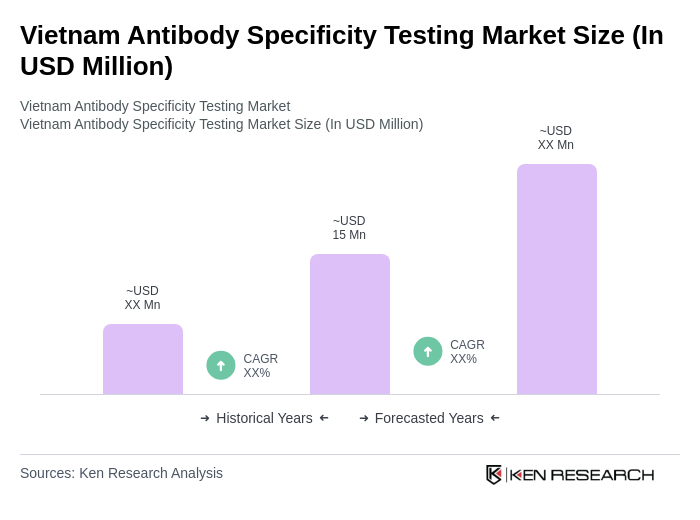
The Vietnam Antibody Specificity Testing Market is valued at approximately USD 15 million, based on a five-year historical analysis. This valuation reflects the growing demand for advanced diagnostic tools in response to increasing chronic and infectious diseases.