Region:Middle East
Author(s):Rebecca
Product Code:KRAD4970
Pages:87
Published On:December 2025
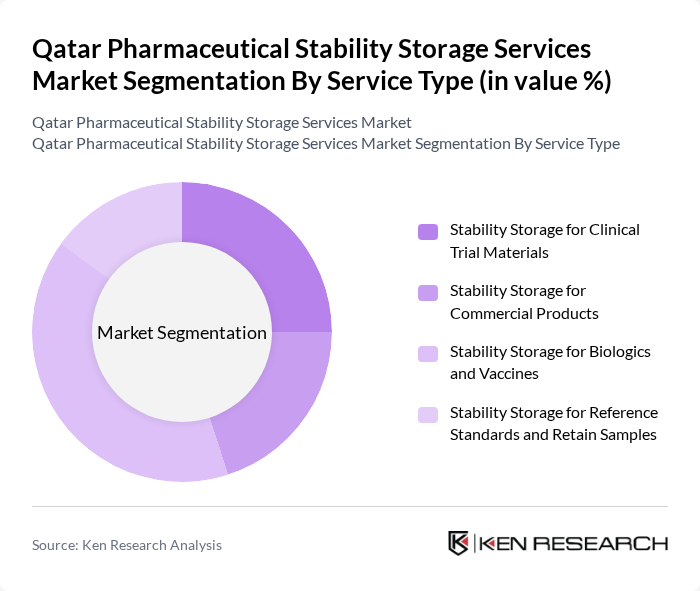
By Service Type:The service type segmentation includes various categories that cater to different pharmaceutical storage needs. The subsegments are Stability Storage for Clinical Trial Materials, Stability Storage for Commercial Products, Stability Storage for Biologics and Vaccines, and Stability Storage for Reference Standards and Retain Samples. Among these, Stability Storage for Biologics and Vaccines is currently dominating the market due to the increasing focus on vaccine development and the growing biopharmaceutical sector. The demand for specialized storage conditions for biologics, which require strict temperature and humidity controls, is driving this segment's growth.
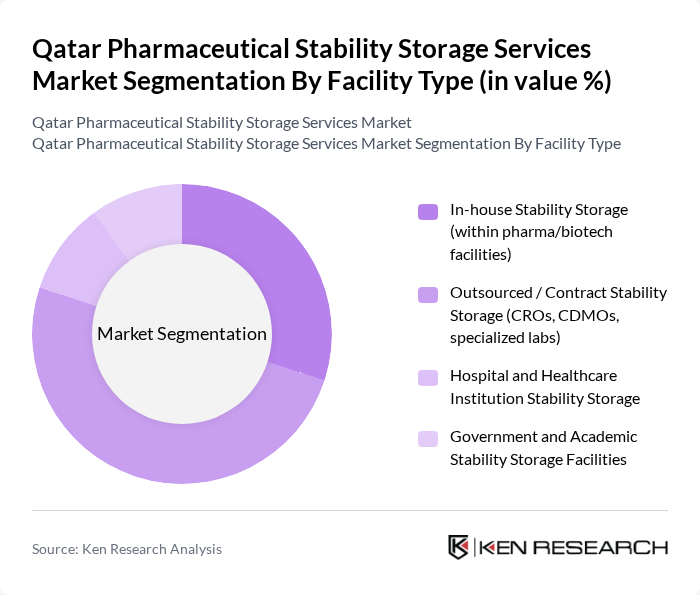
By Facility Type:The facility type segmentation encompasses various storage solutions, including In-house Stability Storage, Outsourced/Contract Stability Storage, Hospital and Healthcare Institution Stability Storage, and Government and Academic Stability Storage Facilities. The Outsourced/Contract Stability Storage segment is leading the market, driven by the increasing trend of pharmaceutical companies outsourcing their storage needs to specialized providers. This allows companies to focus on core activities while ensuring compliance with stringent storage regulations.
The Qatar Pharmaceutical Stability Storage Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qatar Pharma, Hamad Medical Corporation (HMC), Sidra Medicine, Primary Health Care Corporation (PHCC), Qatar Biobank, Qatar University – Biomedical & Pharmaceutical Research Facilities, Ministry of Public Health (MoPH) – Central Medical Stores & Reference Laboratories, Qatar National Central Pharmacy, Qatar Red Crescent Society – Medical Warehousing & Cold Chain, DHL Global Forwarding Qatar – Life Sciences & Healthcare Logistics, Aramex Qatar – Healthcare & Cold Chain Solutions, GWC (Gulf Warehousing Company) – Pharma and Cold Chain Logistics, Doha Drug Store, Naseem Healthcare Group – Central Pharmacy & Storage Facilities, Aster DM Healthcare Qatar – Pharmacy and Cold Storage Network contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar pharmaceutical stability storage services market appears promising, driven by increasing investments in healthcare infrastructure and technological advancements. As the demand for biopharmaceuticals continues to rise, companies are likely to prioritize compliance and efficiency in their storage solutions. Furthermore, the integration of IoT technologies for real-time monitoring will enhance operational capabilities, ensuring product integrity. This evolving landscape presents significant opportunities for growth and innovation in the sector, positioning Qatar as a regional leader in pharmaceutical storage solutions.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Stability Storage for Clinical Trial Materials Stability Storage for Commercial Products Stability Storage for Biologics and Vaccines Stability Storage for Reference Standards and Retain Samples |
| By Facility Type | In-house Stability Storage (within pharma/biotech facilities) Outsourced / Contract Stability Storage (CROs, CDMOs, specialized labs) Hospital and Healthcare Institution Stability Storage Government and Academic Stability Storage Facilities |
| By Storage Condition | Refrigerated Storage (2°C to 8°C) Freezer and Deep Freezer Storage (-20°C to -40°C) Ultra-low Temperature Storage (-70°C to -90°C) Controlled Room Temperature Storage (15°C to 25°C) Specialized / Custom Conditions (e.g., photostability, humidity-controlled) |
| By Application | New Drug Development and Registration (ICH Stability Studies) Ongoing Product Quality / Shelf-life Management Vaccine and Biologic Storage for National Immunization and Specialty Programs Investigational Medicinal Products for Clinical Trials |
| By Ownership | Public Sector (MoPH, Hamad Medical Corporation, government labs) Private Sector (pharma, distributors, private hospitals, independent providers) Joint Ventures and PPP-operated Facilities Regional / GCC-focused Third-party Providers |
| By Temperature-Control Infrastructure | Cold Rooms and Walk-in Chambers Reach-in Stability Chambers and Incubators Automated / High-density Storage Systems Transport-linked Stability Storage (integrated with cold chain logistics) |
| By Compliance Standards | WHO and ICH Stability Guidelines-compliant Facilities Good Distribution Practice (GDP) Compliant Storage Good Manufacturing Practice (GMP) / Good Laboratory Practice (GLP) Compliant Storage ISO 9001 / ISO 13485 and Other Quality-certified Facilities |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 60 | Quality Assurance Managers, Production Supervisors |
| Logistics Providers | 50 | Operations Managers, Supply Chain Directors |
| Healthcare Institutions | 40 | Pharmacy Directors, Procurement Officers |
| Regulatory Bodies | 40 | Compliance Officers, Policy Makers |
| Research Institutions | 40 | Pharmaceutical Researchers, Academic Professors |
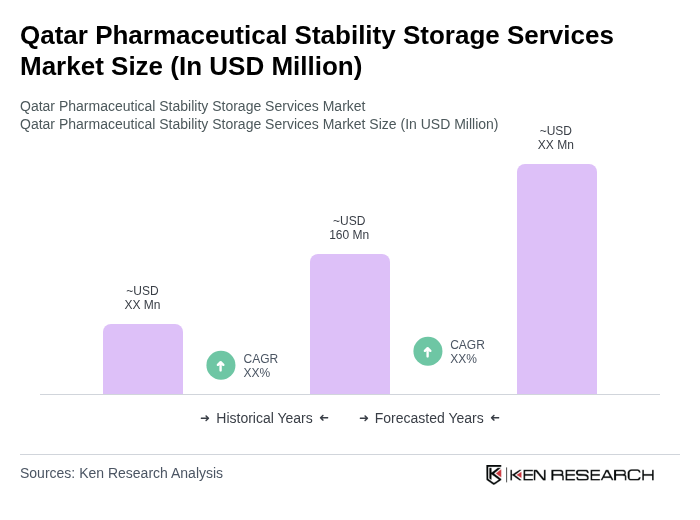
The Qatar Pharmaceutical Stability Storage Services Market is valued at approximately USD 160 million, reflecting the growth driven by increasing demand for pharmaceutical products, clinical trials, and stringent storage conditions necessary for maintaining drug efficacy and safety.