About the Report
Base Year 2024Saudi Arabia Biobanks Industry Market Overview
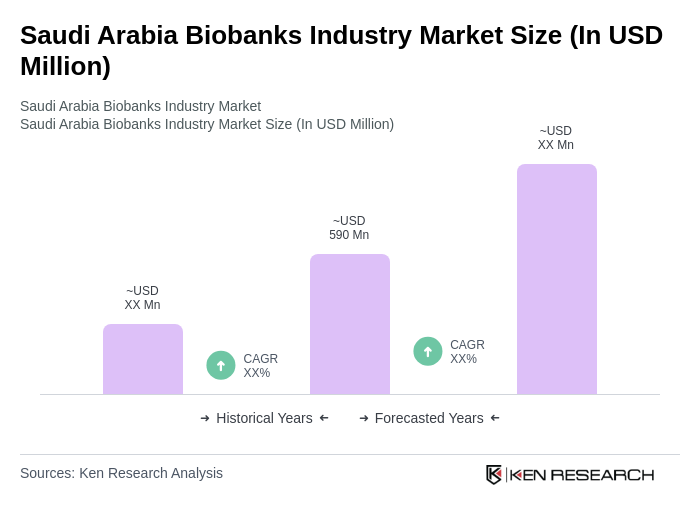
- The Saudi Arabia Biobanks Industry Market is valued at USD 590 million, based on a five-year historical analysis. This growth is primarily driven by increasing investments in healthcare infrastructure, advancements in genomics, and a rising demand for personalized medicine. The integration of biobanks into clinical research and drug development processes has further fueled market expansion, as they provide essential resources for understanding diseases and developing targeted therapies. The increasing prevalence of chronic diseases, such as cancer, diabetes, and cardiovascular disorders, has amplified the demand for high-quality biological specimens for understanding disease mechanisms and developing targeted therapies.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the Saudi Arabia Biobanks Industry Market due to their robust healthcare systems and research institutions. Riyadh, being the capital, hosts numerous medical facilities and universities that contribute to biobanking activities. The city is home to major biobanking initiatives including the National Biobank launched in December 2022 to enhance the country's public health system capabilities, and the Saudi Biobank project undertaken by the King Abdullah International Medical Research Center, which aims at building a comprehensive biobanking platform for the country with enrollment of over 200,000 individuals. Jeddah and Dammam also play significant roles, with their strategic locations and access to diverse populations, enhancing the collection of biological samples for research and clinical applications.
- The Saudi Ministry of Health issued the Executive Regulations for Biobanks, 2022, establishing a comprehensive framework for biobank operations in the kingdom. These regulations, administered by the Saudi Food and Drug Authority (SFDA), mandate licensing requirements for all biobanking facilities, specify standards for sample collection, processing, storage, and distribution, and set forth ethical guidelines for informed consent and data protection. The regulations require biobanks to obtain operational licenses, maintain international quality standards (ISO 20387:2018 for biobanking), implement robust sample tracking systems, and establish governance structures with ethics committees. Facilities must comply with minimum infrastructure requirements including temperature-controlled storage units, backup power systems, and comprehensive documentation protocols. The regulations also outline provisions for cross-border sample sharing, commercial use of biological materials, and penalties for non-compliance, thereby creating a structured environment to support research and innovation in healthcare while protecting donor rights and specimen integrity.
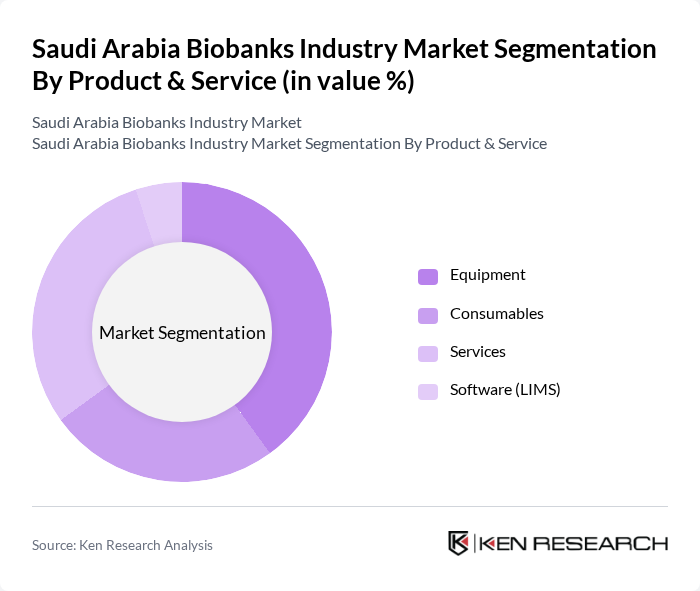
Saudi Arabia Biobanks Industry Market Segmentation
By Product & Service:The biobanks industry in Saudi Arabia is segmented into four main categories: Equipment, Consumables, Services, and Software (LIMS). Among these, the Equipment segment is currently leading the market due to the increasing demand for advanced storage and processing technologies. Biobanking equipment represents the largest revenue-generating segment, accounting for over half of the market share, driven by the need for sophisticated cold storage systems, automated sample processing equipment, and sample management technologies. The rise in research activities and clinical trials has also contributed to the growth of the Services segment, as biobanks provide essential support for sample management and analysis. Meanwhile, LIMS (Laboratory Information Management Systems) represents the fastest-growing product segment, reflecting the increasing emphasis on digital transformation and data management capabilities in biobanking operations.
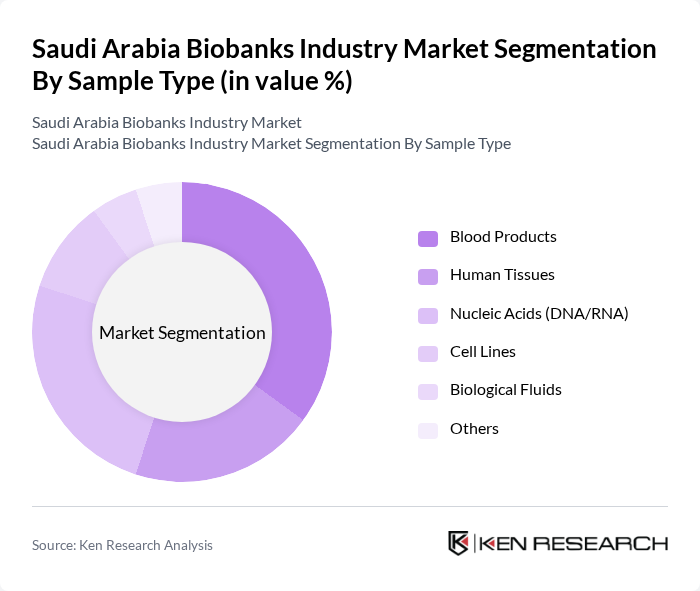
By Sample Type:The market is also segmented by sample type, which includes Blood Products, Human Tissues, Nucleic Acids (DNA/RNA), Cell Lines, Biological Fluids, and Others. Blood Products dominate this segment due to their critical role in clinical research and diagnostics. The increasing prevalence of chronic diseases has led to a higher demand for blood samples, while Nucleic Acids are gaining traction due to advancements in genetic research and personalized medicine. The Saudi Human Genome Program and other national genomic initiatives have significantly increased the demand for DNA and RNA samples, supporting research into genetic predispositions to diseases prevalent in the Saudi population.
Saudi Arabia Biobanks Industry Market Competitive Landscape
The Saudi Arabia Biobanks Industry Market is characterized by a dynamic mix of regional and international players. Leading participants such as King Faisal Specialist Hospital & Research Centre, King Abdullah International Medical Research Center (KAIMRC), King Abdulaziz City for Science and Technology (KACST), Saudi Biobank, Saudi Human Genome Program (SHGP), King Saud University - Medical City Biobank, King Abdulaziz University Hospital Biorepository, National Center for Genomic Technology (NCGT), Prince Sultan Military Medical City Biobank, National Guard Health Affairs Research & Innovation Center, Thermo Fisher Scientific Inc. (Saudi Arabia Operations), Merck KGaA (Saudi Arabia), Qiagen N.V. (Saudi Arabia Distribution), STEMCELL Technologies (Regional Partner), PromoCell GmbH (Middle East Distributor) contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Biobanks Industry Market Industry Analysis
Growth Drivers
- Increasing Demand for Personalized Medicine:The Saudi healthcare sector is witnessing a surge in personalized medicine, driven by a 12% annual increase in genomic testing. The Ministry of Health's investment of approximately SAR 2 billion in genomic research initiatives in the future highlights the commitment to tailoring treatments based on individual genetic profiles. This trend is expected to enhance the demand for biobanks, which are essential for storing genetic material and facilitating research in personalized therapies.
- Government Initiatives to Support Biobanking:The Saudi government has allocated SAR 3 billion for biobanking infrastructure development as part of its Vision 2030 initiative. This funding aims to establish state-of-the-art biobanks across the country, enhancing research capabilities. Additionally, the establishment of the National Biobank has set a precedent, with over 150,000 samples collected, fostering collaboration between public and private sectors to advance biobanking practices.
- Rising Prevalence of Chronic Diseases:Chronic diseases, such as diabetes and cardiovascular conditions, affect over 8 million people in Saudi Arabia, representing a significant public health challenge. The increasing burden of these diseases has prompted a focus on research and development, with the government investing SAR 1.5 billion in chronic disease management programs in the future. Biobanks play a crucial role in understanding disease mechanisms and developing targeted therapies, driving their growth in the healthcare landscape.
Market Challenges
- Ethical Concerns Regarding Biobanking:Ethical issues surrounding consent and data privacy pose significant challenges for biobanks in Saudi Arabia. In the future, approximately 65% of the population expressed concerns about the use of their biological samples without adequate consent. This skepticism can hinder participation in biobanking initiatives, ultimately affecting the quality and quantity of samples available for research, which is critical for advancing medical science.
- High Operational Costs:The operational costs associated with establishing and maintaining biobanks are substantial, with estimates reaching SAR 600 million for a fully functional facility. These costs encompass advanced storage technologies, personnel training, and compliance with regulatory standards. As a result, many potential biobanks may struggle to secure funding, limiting their ability to contribute effectively to research and development in the healthcare sector.
Saudi Arabia Biobanks Industry Market Future Outlook
The future of the biobanks industry in Saudi Arabia appears promising, driven by advancements in technology and increasing collaboration between research institutions and healthcare providers. The integration of artificial intelligence in biobanking processes is expected to enhance data analysis and sample management. Furthermore, the growing emphasis on patient-centric approaches will likely lead to more inclusive biobanking practices, fostering greater public trust and participation in biobanking initiatives, ultimately benefiting the healthcare landscape.
Market Opportunities
- Expansion of Research Collaborations:There is a significant opportunity for biobanks to expand research collaborations with international institutions. In the future, Saudi Arabia aims to increase its research partnerships by 35%, leveraging biobanks to facilitate access to diverse biological samples, which can enhance the quality of research and innovation in personalized medicine.
- Development of New Biobanking Technologies:The emergence of innovative biobanking technologies presents a lucrative opportunity. Investments in automation and digital biobanking solutions are projected to grow by 30% in the future, enabling more efficient sample processing and data management. This technological advancement can significantly improve operational efficiency and reduce costs for biobanks, making them more sustainable.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product & Service | Equipment Consumables Services Software (LIMS) |
| By Sample Type | Blood Products Human Tissues Nucleic Acids (DNA/RNA) Cell Lines Biological Fluids Others |
| By Biobank Type | Population-based Biobanks Disease-oriented Biobanks Clinical Biobanks Others |
| By Application | Regenerative Medicine Life Science Research Clinical Research Drug Development Biomarker Discovery Personalized Medicine Others |
| By End-User | Academic and Research Institutions Pharmaceutical and Biotechnology Companies Hospitals and Clinical Laboratories Government Agencies Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Biotechnology and Pharmaceutical Companies
Healthcare Providers and Hospitals
Clinical Research Organizations
Biobanking Technology Providers
Non-Governmental Organizations (NGOs) focused on health research
Public Health Agencies
Players Mentioned in the Report:
King Faisal Specialist Hospital & Research Centre
King Abdullah International Medical Research Center (KAIMRC)
King Abdulaziz City for Science and Technology (KACST)
Saudi Biobank
Saudi Human Genome Program (SHGP)
King Saud University - Medical City Biobank
King Abdulaziz University Hospital Biorepository
National Center for Genomic Technology (NCGT)
Prince Sultan Military Medical City Biobank
National Guard Health Affairs Research & Innovation Center
Thermo Fisher Scientific Inc. (Saudi Arabia Operations)
Merck KGaA (Saudi Arabia)
Qiagen N.V. (Saudi Arabia Distribution)
STEMCELL Technologies (Regional Partner)
PromoCell GmbH (Middle East Distributor)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Biobanks Industry Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Biobanks Industry Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Biobanks Industry Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for personalized medicine
3.1.2 Government initiatives to support biobanking
3.1.3 Rising prevalence of chronic diseases
3.1.4 Advancements in genomic research
3.2 Market Challenges
3.2.1 Ethical concerns regarding biobanking
3.2.2 High operational costs
3.2.3 Limited public awareness
3.2.4 Regulatory hurdles
3.3 Market Opportunities
3.3.1 Expansion of research collaborations
3.3.2 Development of new biobanking technologies
3.3.3 Increased funding for biomedical research
3.3.4 Growing interest in regenerative medicine
3.4 Market Trends
3.4.1 Integration of AI in biobanking processes
3.4.2 Shift towards digital biobanks
3.4.3 Focus on patient-centric biobanking
3.4.4 Collaboration with pharmaceutical companies
3.5 Government Regulation
3.5.1 National regulations on biobanking practices
3.5.2 Data protection laws affecting biobanks
3.5.3 Guidelines for ethical biobanking
3.5.4 Funding policies for biobanking initiatives
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Biobanks Industry Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Biobanks Industry Market Segmentation
8.1 By Product & Service
8.1.1 Equipment
8.1.2 Consumables
8.1.3 Services
8.1.4 Software (LIMS)
8.2 By Sample Type
8.2.1 Blood Products
8.2.2 Human Tissues
8.2.3 Nucleic Acids (DNA/RNA)
8.2.4 Cell Lines
8.2.5 Biological Fluids
8.2.6 Others
8.3 By Biobank Type
8.3.1 Population-based Biobanks
8.3.2 Disease-oriented Biobanks
8.3.3 Clinical Biobanks
8.3.4 Others
8.4 By Application
8.4.1 Regenerative Medicine
8.4.2 Life Science Research
8.4.3 Clinical Research
8.4.4 Drug Development
8.4.5 Biomarker Discovery
8.4.6 Personalized Medicine
8.4.7 Others
8.5 By End-User
8.5.1 Academic and Research Institutions
8.5.2 Pharmaceutical and Biotechnology Companies
8.5.3 Hospitals and Clinical Laboratories
8.5.4 Government Agencies
8.5.5 Others
9. Saudi Arabia Biobanks Industry Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Organization Size (Large, Medium, Small)
9.2.3 Total Sample Capacity (in units/year)
9.2.4 Number of Active Biobank Facilities
9.2.5 Research Collaboration Index
9.2.6 Technology Infrastructure Investment (USD)
9.2.7 Number of Clinical Partnerships
9.2.8 Genomic Data Repository Size
9.2.9 Regulatory Compliance Rating
9.2.10 International Accreditation Status
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 King Faisal Specialist Hospital & Research Centre
9.5.2 King Abdullah International Medical Research Center (KAIMRC)
9.5.3 King Abdulaziz City for Science and Technology (KACST)
9.5.4 Saudi Biobank
9.5.5 Saudi Human Genome Program (SHGP)
9.5.6 King Saud University - Medical City Biobank
9.5.7 King Abdulaziz University Hospital Biorepository
9.5.8 National Center for Genomic Technology (NCGT)
9.5.9 Prince Sultan Military Medical City Biobank
9.5.10 National Guard Health Affairs Research & Innovation Center
9.5.11 Thermo Fisher Scientific Inc. (Saudi Arabia Operations)
9.5.12 Merck KGaA (Saudi Arabia)
9.5.13 Qiagen N.V. (Saudi Arabia Distribution)
9.5.14 STEMCELL Technologies (Regional Partner)
9.5.15 PromoCell GmbH (Middle East Distributor)
10. Saudi Arabia Biobanks Industry Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Commerce
10.1.4 Ministry of Research and Innovation
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Biobanking Facilities
10.2.2 Funding for Research Projects
10.2.3 Expenditure on Technology Upgrades
10.3 Pain Point Analysis by End-User Category
10.3.1 Academic Institutions
10.3.2 Healthcare Providers
10.3.3 Research Organizations
10.4 User Readiness for Adoption
10.4.1 Awareness of Biobanking Benefits
10.4.2 Training and Education Needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Biobank Impact
10.5.2 Expansion into New Research Areas
10.5.3 Long-term Sustainability Plans
11. Saudi Arabia Biobanks Industry Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Key Partnerships Exploration
1.5 Cost Structure Assessment
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing biobank reports and publications from Saudi health authorities
- Review of academic journals and articles focusing on biobanking practices in the region
- Examination of market trends and forecasts from industry-specific databases and white papers
Primary Research
- Interviews with biobank directors and managers across leading healthcare institutions
- Surveys targeting researchers and scientists involved in biobanking projects
- Focus groups with stakeholders from pharmaceutical companies and biotech firms
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government and private sector reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the biobanking market size based on national healthcare expenditure data
- Segmentation of the market by biobank type (e.g., population-based, disease-specific)
- Incorporation of government initiatives and funding for biobanking projects
Bottom-up Modeling
- Collection of data on the number of biobanks and their operational capacities
- Estimation of revenue generated from biobanking services and sample storage fees
- Analysis of cost structures and funding sources for biobanks in Saudi Arabia
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth and disease prevalence rates
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Population-based Biobanks | 100 | Biobank Managers, Public Health Officials |
| Disease-specific Biobanks | 80 | Clinical Researchers, Oncologists |
| Pharmaceutical Collaborations | 60 | Pharmaceutical R&D Managers, Biotech Executives |
| Regulatory Compliance in Biobanking | 50 | Compliance Officers, Legal Advisors |
| Technological Innovations in Biobanking | 70 | IT Managers, Biobank Technologists |
Frequently Asked Questions
What is the current value of the Saudi Arabia Biobanks Industry Market?
The Saudi Arabia Biobanks Industry Market is valued at approximately USD 590 million, reflecting significant growth driven by investments in healthcare infrastructure, advancements in genomics, and the rising demand for personalized medicine.