Region:Middle East
Author(s):Geetanshi
Product Code:KRAD3746
Pages:87
Published On:November 2025
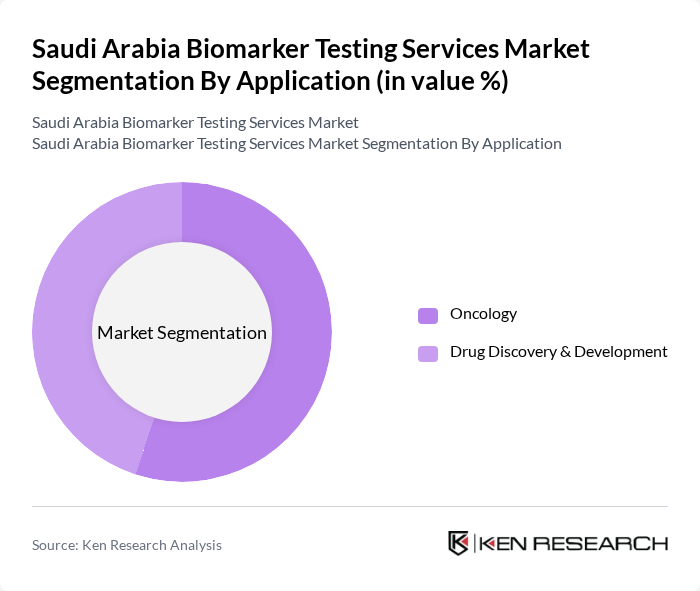
By Application:The application segment includes various areas where biomarker testing is utilized, such as oncology and drug discovery & development. Oncology remains the leading application area, driven by the rising incidence of cancer and the need for targeted therapies. Drug discovery & development is also significant, as it aids in the identification of potential drug candidates, evaluation of efficacy, and monitoring of therapeutic response. The adoption of genomics, proteomics, and metabolomics is enhancing the precision and scope of biomarker applications in both segments .
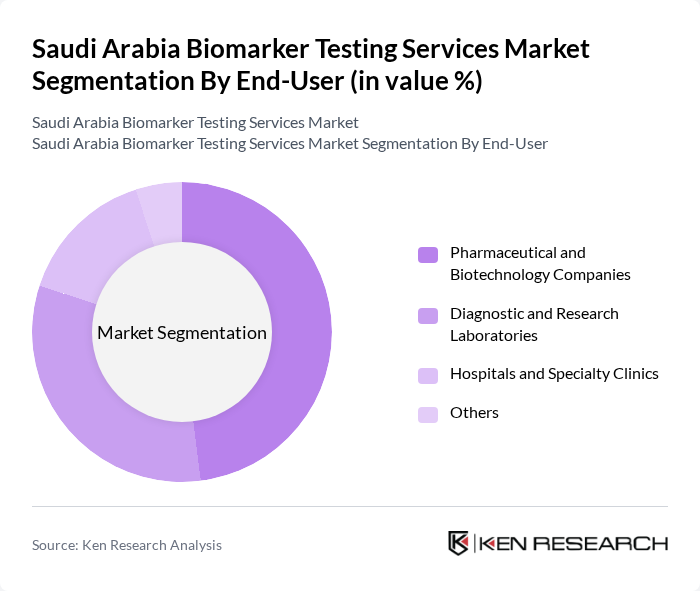
By End-User:The end-user segment comprises pharmaceutical and biotechnology companies, diagnostic and research laboratories, hospitals and specialty clinics, and others. Pharmaceutical and biotechnology companies are the dominant end-users due to their extensive reliance on biomarker testing for drug development and clinical trials. Diagnostic and research laboratories are rapidly expanding their service portfolios, while hospitals and specialty clinics are increasingly integrating biomarker testing into routine clinical practice for personalized patient care .
The Saudi Arabia Biomarker Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Eurofins Scientific SE, SGS AG, Charles River Laboratories International Inc, Labcorp Drug Development, Thermo Fisher Scientific Inc, Icon PLC, IQVIA Holdings Inc, Syneos Health, Intertek Group PLC, BioAgilytix Labs, King Faisal Specialist Hospital & Research Centre, Saudi German Hospital contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biomarker testing services market in Saudi Arabia appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The integration of artificial intelligence in biomarker analysis is expected to enhance diagnostic accuracy and efficiency. Additionally, the expansion of telemedicine services will facilitate greater access to biomarker testing, particularly in remote areas, thereby improving patient engagement and outcomes in the healthcare system.
| Segment | Sub-Segments |
|---|---|
| By Application | Oncology Drug Discovery & Development |
| By End-User | Pharmaceutical and Biotechnology Companies Diagnostic and Research Laboratories Hospitals and Specialty Clinics Others |
| By Biomarker Type | Efficacy Biomarkers Safety Biomarkers Validation Biomarkers |
| By Technology | Next-Generation Sequencing PCR-based Testing Mass Spectrometry Immunoassays Others |
| By Sample Type | Blood Samples Tissue Samples Urine Samples Others |
| By Region | Northern and Central Region Eastern Region Western Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Biomarker Testing | 100 | Oncologists, Pathologists |
| Cardiovascular Biomarker Testing | 80 | Cardiologists, General Practitioners |
| Genetic Testing Services | 70 | Genetic Counselors, Laboratory Technicians |
| Patient Awareness and Acceptance | 90 | Patients, Healthcare Advocates |
| Regulatory Compliance Insights | 60 | Regulatory Affairs Specialists, Compliance Officers |
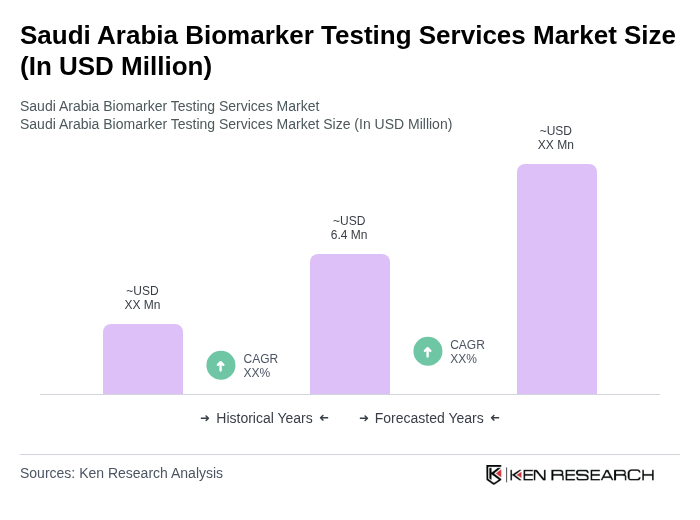
The Saudi Arabia Biomarker Testing Services Market is valued at approximately USD 6.4 million, reflecting growth driven by the increasing prevalence of chronic diseases and advancements in personalized medicine.