Region:Middle East
Author(s):Geetanshi
Product Code:KRAD1087
Pages:86
Published On:November 2025
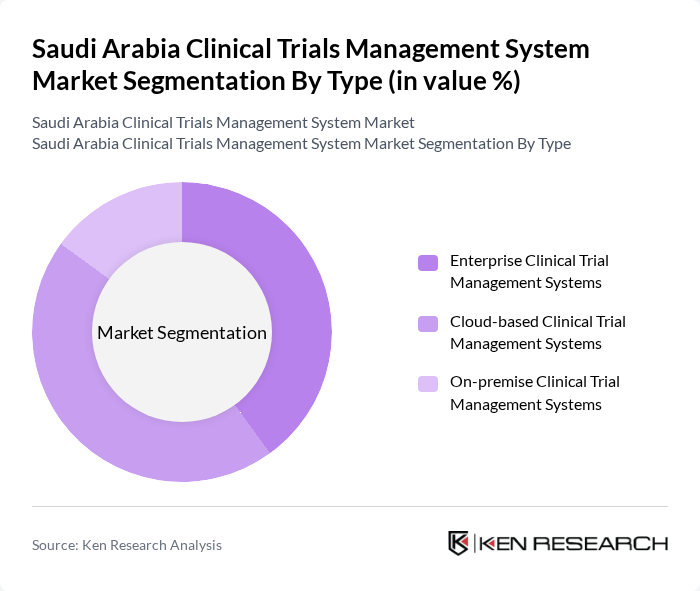
By Type:The market is segmented into three main types:Enterprise Clinical Trial Management Systems, Cloud-based Clinical Trial Management Systems, and On-premise Clinical Trial Management Systems. Cloud-based systems are increasingly preferred due to their scalability, real-time data access, and cost-effectiveness, supporting collaboration among stakeholders and enabling remote trial management. Enterprise and on-premise systems continue to serve organizations with specific security or integration requirements.
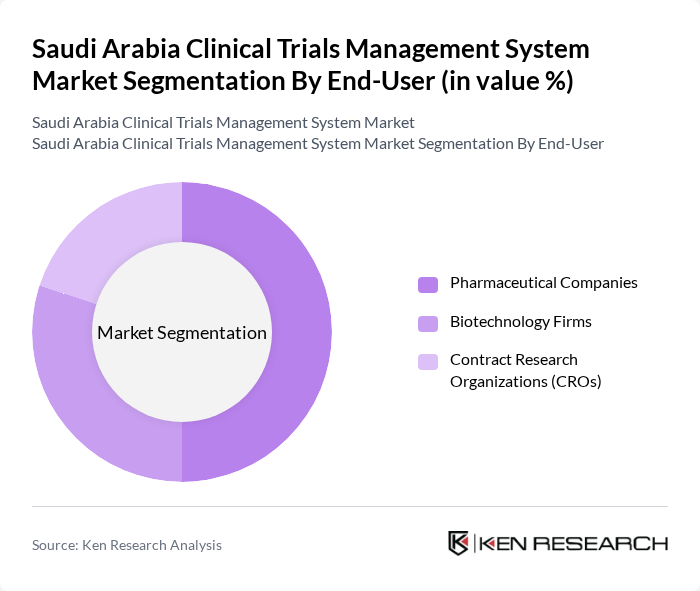
By End-User:The end-user segmentation includesPharmaceutical Companies, Biotechnology Firms, and Contract Research Organizations (CROs). Pharmaceutical companies dominate due to their role as primary sponsors of clinical trials and their need for robust data management and regulatory compliance. Biotechnology firms and CROs are also significant users, with CROs increasingly providing specialized trial management services.
The Saudi Arabia Clinical Trials Management System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Inc., Oracle Corporation, Veeva Systems Inc., Parexel International Corporation, Covance Inc., BioClinica, Inc., Medpace, Inc., CRF Health, ERT, Inc., PPD, Inc., Syneos Health, Inc., Clinipace Worldwide, WCG Clinical, ICON plc, and KCR contribute to innovation, geographic expansion, and service delivery in this space.
The future of the clinical trials management system market in Saudi Arabia appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on innovative treatment solutions. As the prevalence of chronic diseases continues to rise, the demand for efficient clinical trials will increase. Furthermore, advancements in technology, such as AI integration and decentralized trial models, are expected to enhance operational efficiency and patient engagement, ultimately transforming the clinical research landscape in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Enterprise Clinical Trial Management Systems Cloud-based Clinical Trial Management Systems On-premise Clinical Trial Management Systems |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) |
| By Application | Site Management Patient Recruitment Data Management |
| By Deployment Mode | Cloud Deployment On-Premise Deployment |
| By Geography | Central Region Eastern Region Western Region |
| By Regulatory Compliance | FDA Compliance EMA Compliance |
| By Solution Type | Niche Solutions Customizable Platforms |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 100 | Clinical Research Associates, Project Managers |
| Medical Device Trials | 60 | Regulatory Affairs Managers, Clinical Trial Coordinators |
| Oncology Clinical Studies | 50 | Oncologists, Research Nurses |
| Cardiovascular Trials | 40 | Cardiologists, Clinical Research Coordinators |
| Rare Disease Research | 40 | Patient Advocacy Representatives, Clinical Investigators |
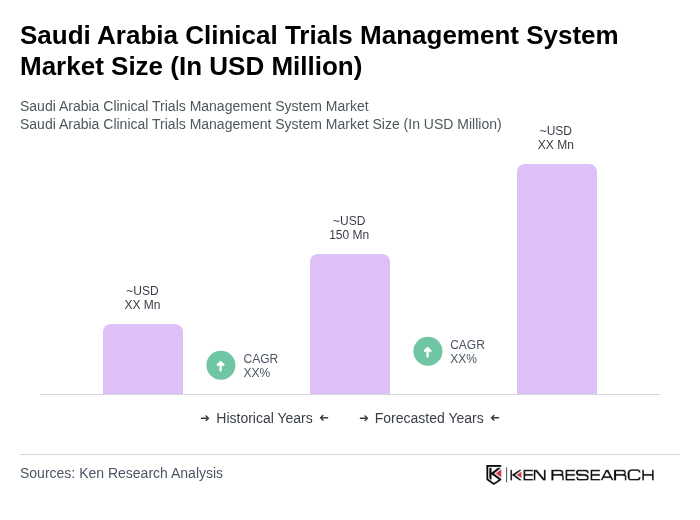
The Saudi Arabia Clinical Trials Management System market is valued at approximately USD 150 million, driven by an increase in clinical trials, investments in research and development, and the adoption of advanced digital platforms for efficient clinical data management.