Region:Middle East
Author(s):Shubham
Product Code:KRAD1912
Pages:92
Published On:December 2025
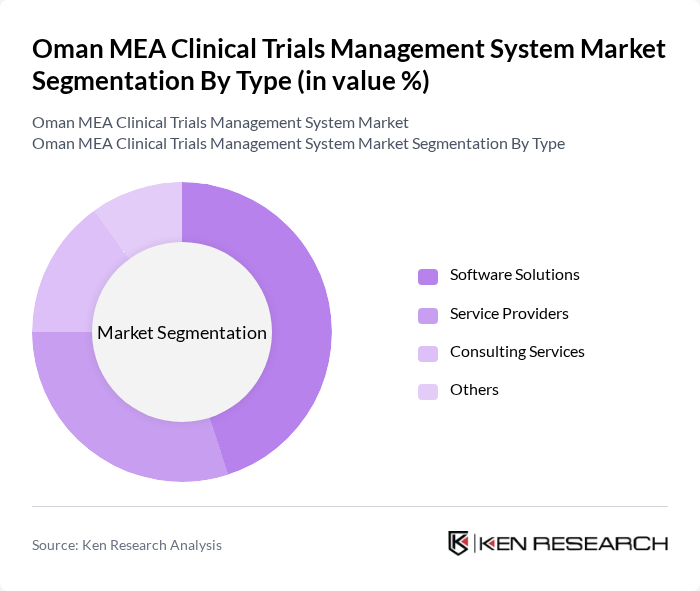
By Type:The market is segmented into software solutions, service providers, consulting services, and others. Among these, software solutions are leading due to the increasing demand for efficient data management and streamlined processes in clinical trials. The rise in digital transformation initiatives in healthcare is further propelling the adoption of software solutions, making them a critical component in the clinical trials management landscape.
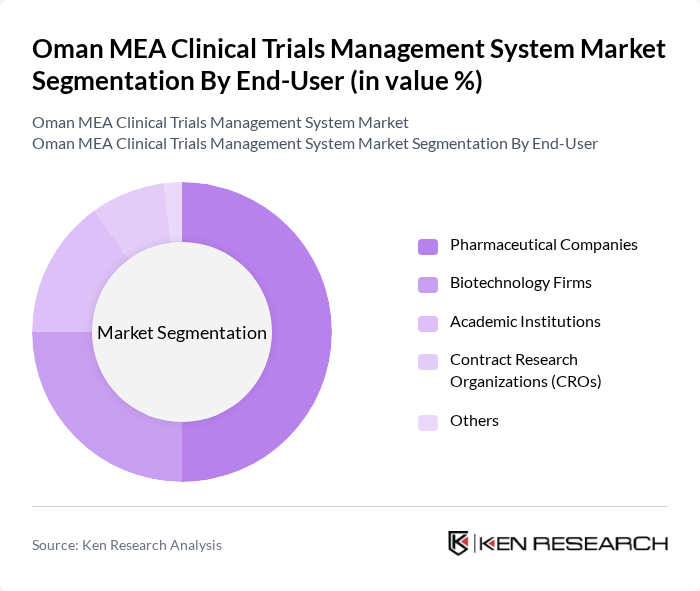
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, academic institutions, contract research organizations (CROs), and others. Pharmaceutical companies dominate this segment as they are the primary sponsors of clinical trials, driving the demand for comprehensive management systems to ensure compliance and efficiency in trial execution.
The Oman MEA Clinical Trials Management System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Oracle Corporation, Veeva Systems, Parexel International, Covance, PPD (Pharmaceutical Product Development), ICON plc, Syneos Health, CRF Health, ERT (eResearchTechnology), Bioclinica, Medpace, KCR, WCG Clinical, Clinipace contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman MEA Clinical Trials Management System market appears promising, driven by advancements in digital health and government support for infrastructure development. The integration of AI capabilities into clinical trial management systems is expected to enhance efficiency and patient engagement. Additionally, the establishment of a unified digital infrastructure through initiatives like the Oman Digital Triangle will facilitate the growth of cloud-based trial platforms, enabling more streamlined and effective clinical research processes across the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Software Solutions Service Providers Consulting Services Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Contract Research Organizations (CROs) Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Phase of Clinical Trials | Phase I Phase II Phase III Phase IV Others |
| By Geography | Muscat Salalah Sohar Nizwa Others |
| By Funding Source | Government Grants Private Investments Public-Private Partnerships Others |
| By Data Management Approach | Centralized Data Management Decentralized Data Management Hybrid Data Management Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 100 | Clinical Research Coordinators, Project Managers |
| Medical Device Trials | 80 | Regulatory Affairs Specialists, Clinical Trial Managers |
| Oncology Trials | 70 | Oncologists, Research Nurses |
| Cardiovascular Trials | 60 | Cardiologists, Clinical Research Associates |
| Neurology Trials | 50 | Neurologists, Data Managers |
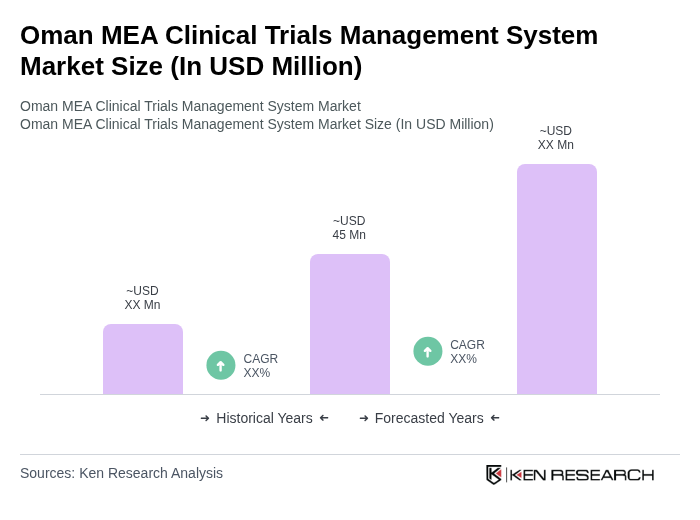
The Oman MEA Clinical Trials Management System Market is valued at approximately USD 45 million, driven by the increasing adoption of healthcare IT and a growing number of clinical trials in the region.