Region:Middle East
Author(s):Rebecca
Product Code:KRAE0800
Pages:86
Published On:December 2025
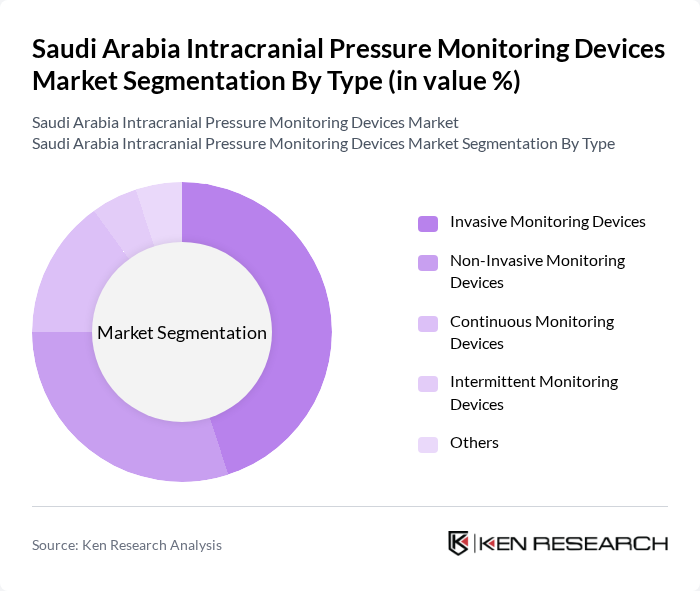
By Type:The market is segmented into various types of monitoring devices, including invasive and non-invasive options. Invasive monitoring devices are often preferred in critical care settings due to their accuracy and reliability in measuring intracranial pressure. Non-invasive devices are gaining traction due to their ease of use and reduced risk of complications. Continuous monitoring devices are particularly popular in hospitals, while intermittent monitoring devices are used in outpatient settings. The demand for these devices is influenced by advancements in technology and the growing need for effective patient management.
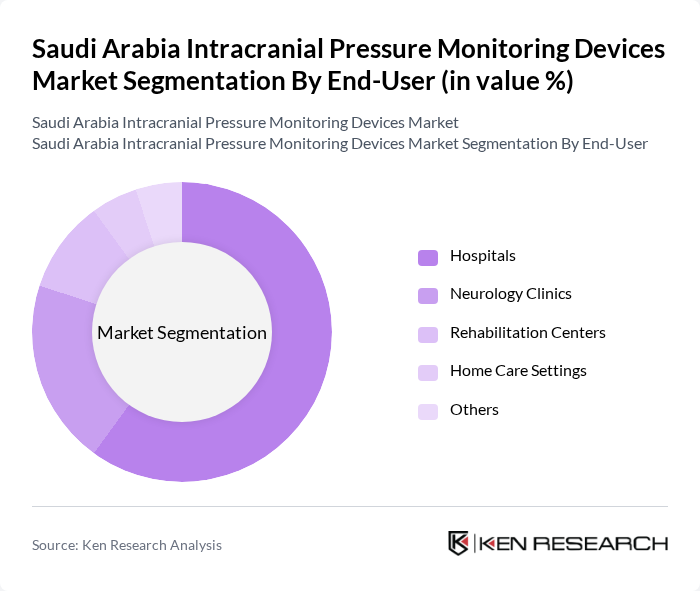
By End-User:The end-user segmentation includes hospitals, neurology clinics, rehabilitation centers, and home care settings. Hospitals are the primary end-users, accounting for a significant share of the market due to their need for advanced monitoring solutions in critical care units. Neurology clinics are also increasingly adopting these devices for outpatient management of patients with neurological disorders. Rehabilitation centers and home care settings are emerging as important segments as they focus on long-term patient monitoring and management.
The Saudi Arabia Intracranial Pressure Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Codman Neuro, Natus Medical Incorporated, Integra LifeSciences, Raumedic AG, NeuroWave Systems, ORA Medical, Sophysa, BrainScope Company, Inc., Cerenovus, Aesculap, B. Braun Melsungen AG, Stryker Corporation, Philips Healthcare, Siemens Healthineers contribute to innovation, geographic expansion, and service delivery in this space.
The future of the intracranial pressure monitoring devices market in Saudi Arabia appears promising, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence in monitoring devices is expected to enhance diagnostic accuracy and patient management. Additionally, the expansion of telemedicine services will facilitate remote monitoring, improving access to care. As healthcare infrastructure continues to develop, the market is poised for significant growth, addressing the rising demand for effective neurological care solutions.
| Segment | Sub-Segments |
|---|---|
| By Type | Invasive Monitoring Devices Non-Invasive Monitoring Devices Continuous Monitoring Devices Intermittent Monitoring Devices Others |
| By End-User | Hospitals Neurology Clinics Rehabilitation Centers Home Care Settings Others |
| By Application | Traumatic Brain Injury Stroke Management Hydrocephalus Treatment Brain Tumor Monitoring Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Retail Pharmacies Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Technology | Fiber Optic Technology Microchip Technology Wireless Technology Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 100 | Neurosurgeons, Department Heads |
| Critical Care Units | 80 | Intensivists, Critical Care Nurses |
| Medical Device Distributors | 60 | Sales Managers, Product Specialists |
| Healthcare Procurement | 70 | Procurement Officers, Supply Chain Managers |
| Regulatory Bodies | 50 | Regulatory Affairs Managers, Compliance Officers |
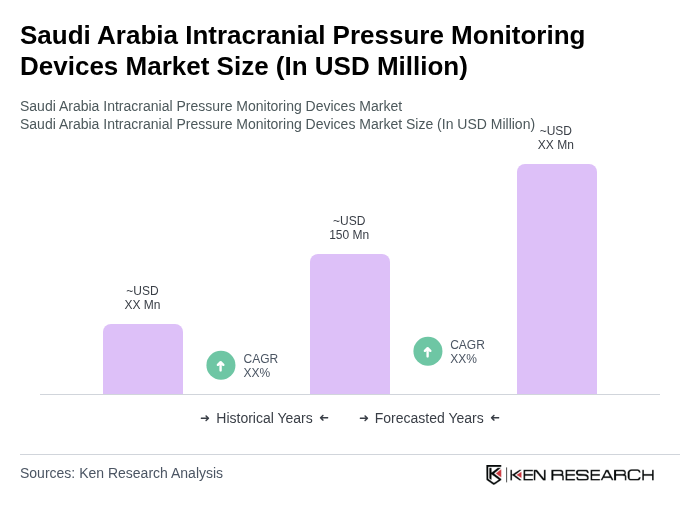
The Saudi Arabia Intracranial Pressure Monitoring Devices Market is valued at approximately USD 150 million, reflecting a significant growth driven by the increasing prevalence of neurological disorders and advancements in medical technology.