Region:Middle East
Author(s):Rebecca
Product Code:KRAC2631
Pages:89
Published On:October 2025
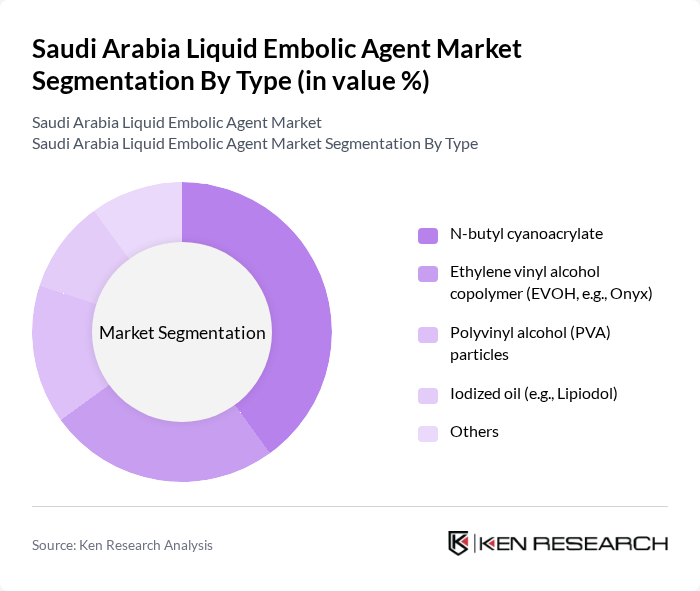
By Type:The liquid embolic agent market is segmented into N-butyl cyanoacrylate, ethylene vinyl alcohol copolymer (EVOH), polyvinyl alcohol (PVA) particles, iodized oil, and others. N-butyl cyanoacrylate remains the leading subsegment due to its established effectiveness in a wide range of embolization procedures and its rapid polymerization properties, which enable precise vessel occlusion. The growing preference for minimally invasive techniques and the introduction of new formulations have further strengthened its market position .
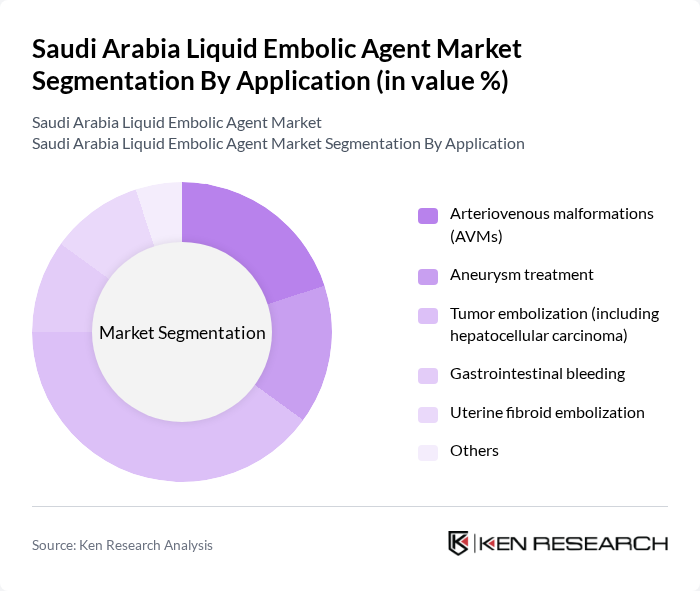
By Application:Applications of liquid embolic agents include arteriovenous malformations (AVMs), aneurysm treatment, tumor embolization, gastrointestinal bleeding, uterine fibroid embolization, and others. Tumor embolization, particularly for hepatocellular carcinoma, is the most significant application due to the rising incidence of liver cancer and the proven efficacy of liquid embolic agents in targeted tumor therapy. The segment's growth is further supported by increasing adoption of embolotherapy for oncological and neurovascular indications .
The Saudi Arabia Liquid Embolic Agent Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Boston Scientific Corporation, Johnson & Johnson (including Cerenovus), Terumo Corporation, Stryker Corporation, Cook Medical, Merit Medical Systems, AngioDynamics, Penumbra, Inc., Asahi Intecc Co., Ltd., Balt, B. Braun SE, GEM Srl, BlackSwan Vascular, Inc., INVAMED contribute to innovation, geographic expansion, and service delivery in this space.
The future of the liquid embolic agent market in Saudi Arabia appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the government continues to enhance healthcare infrastructure, the integration of advanced technologies, including artificial intelligence, will likely improve treatment planning and patient outcomes. Additionally, the rise in telemedicine services will facilitate better access to specialized care, further supporting market growth and innovation in treatment options.
| Segment | Sub-Segments |
|---|---|
| By Type | N-butyl cyanoacrylate Ethylene vinyl alcohol copolymer (EVOH, e.g., Onyx) Polyvinyl alcohol (PVA) particles Iodized oil (e.g., Lipiodol) Others |
| By Application | Arteriovenous malformations (AVMs) Aneurysm treatment Tumor embolization (including hepatocellular carcinoma) Gastrointestinal bleeding Uterine fibroid embolization Others |
| By End-User | Hospitals Ambulatory surgical centers Specialty clinics Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Price Range | Low price range Mid price range High price range |
| By Policy Support | Subsidies for healthcare providers Tax incentives for manufacturers Grants for research and development Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Interventional Radiology Practices | 60 | Interventional Radiologists, Medical Directors |
| Hospital Procurement Departments | 50 | Procurement Managers, Supply Chain Coordinators |
| Clinical Research Institutions | 40 | Clinical Researchers, Medical Device Evaluators |
| Medical Device Distributors | 45 | Sales Managers, Product Specialists |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
The Saudi Arabia Liquid Embolic Agent Market is valued at approximately USD 8 million, driven by the increasing prevalence of vascular diseases and advancements in medical technology, alongside a growing emphasis on minimally invasive procedures.