Region:Middle East
Author(s):Geetanshi
Product Code:KRAA6011
Pages:86
Published On:January 2026
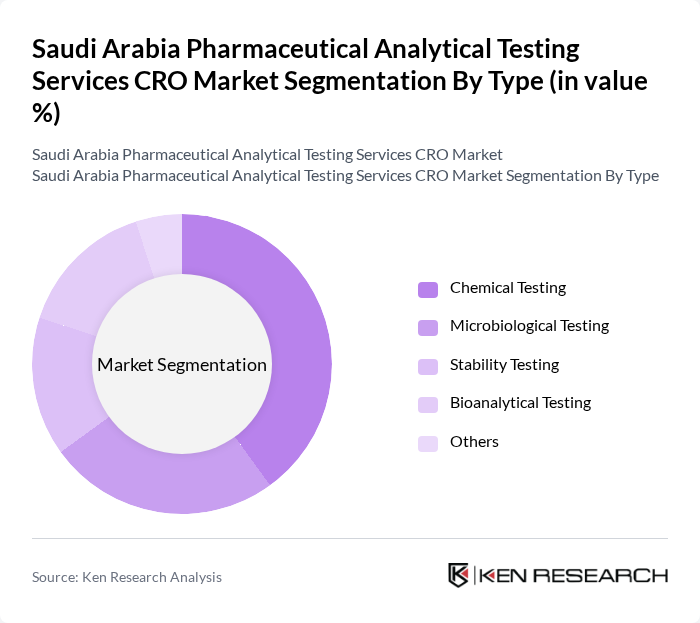
By Type:The market is segmented into various types of analytical testing services, including Chemical Testing, Microbiological Testing, Stability Testing, Bioanalytical Testing, and Others. Among these, Bioanalytical Testing is the leading segment, driven by the increasing need for drug testing and validation in clinical trials. The demand for precise and reliable bioanalytical data is critical for regulatory approvals, making this segment a focal point for CROs.
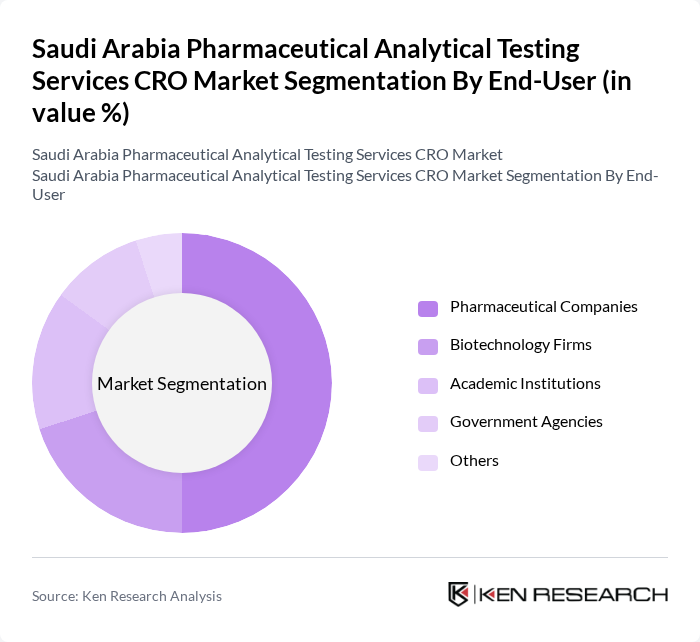
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, Government Agencies, and Others. Pharmaceutical Companies dominate this segment, as they are the primary clients for CROs, requiring extensive analytical testing services for drug development and regulatory compliance. The increasing number of pharmaceutical firms in Saudi Arabia is further propelling this segment's growth.
The Saudi Arabia Pharmaceutical Analytical Testing Services CRO Market is characterized by a dynamic mix of regional and international players. Leading participants such asEurofins Scientific,SGS S.A.,Charles River Laboratories,Pace Analytical Services,Intertek Group,Labcorp Drug Development,Medpace,Covance,WuXi AppTec,Bureau Veritas,KMR Group,Rho,Clinipace,PRA Health Sciences, andCelerioncontribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabian pharmaceutical analytical testing services market appears promising, driven by advancements in digital health and artificial intelligence. The digital health market is projected to grow from USD 2.5 billion in future to USD 16.94 billion by future, indicating a substantial increase in demand for analytical testing services. Additionally, the launch of regulatory sandboxes will facilitate innovation in testing methodologies, allowing CROs to explore new technologies and improve service delivery in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | **Chemical Testing** **Microbiological Testing** **Stability Testing** **Bioanalytical Testing** **Others** |
| By End-User | **Pharmaceutical Companies** **Biotechnology Firms** **Academic Institutions** **Government Agencies** **Others** |
| By Service Type | **Preclinical Services** **Clinical Services** **Post-Market Surveillance** **Others** |
| By Therapeutic Area | **Oncology** **Cardiovascular** **Neurology** **Others** |
| By Region | **Central Region** **Eastern Region** **Western Region** **Southern Region** |
| By Client Type | **Large Enterprises** **SMEs** **Startups** **Others** |
| By Testing Phase | **Phase I** **Phase II** **Phase III** **Others** |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Quality Control | 100 | Quality Control Managers, Laboratory Technicians |
| Regulatory Compliance Testing | 80 | Regulatory Affairs Managers, Compliance Officers |
| Stability Testing Services | 70 | Product Development Managers, R&D Scientists |
| Bioanalytical Testing Services | 60 | Clinical Research Coordinators, Pharmacokinetics Specialists |
| Microbiological Testing Services | 50 | Microbiologists, Quality Assurance Specialists |
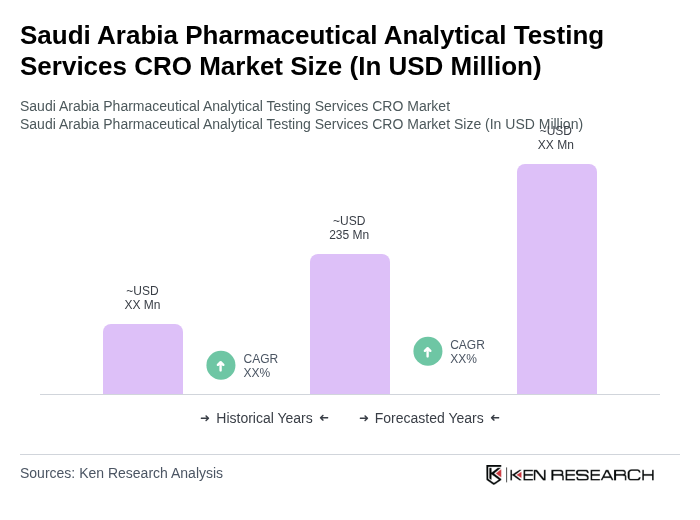
The Saudi Arabia Pharmaceutical Analytical Testing Services CRO Market is valued at approximately USD 45 million, reflecting a five-year historical analysis. This growth is driven by increasing demand for bioanalytical and method development services within pharmaceutical R&D.