Region:Middle East
Author(s):Dev
Product Code:KRAB1812
Pages:99
Published On:January 2026
By Service Type:

The service type segmentation includes various subsegments such as Bioanalytical Testing Services, Method Development & Validation, Stability Testing & Storage, Raw Material & Finished Product Testing, Microbiological Testing & Environmental Monitoring, Physical & Chemical Characterization, and Others. Among these, Bioanalytical Testing Services is the leading subsegment, driven by the increasing need for drug development and regulatory compliance. The demand for accurate and reliable bioanalytical data is critical for pharmaceutical companies, making this segment a focal point in the market.
By Molecule Type:
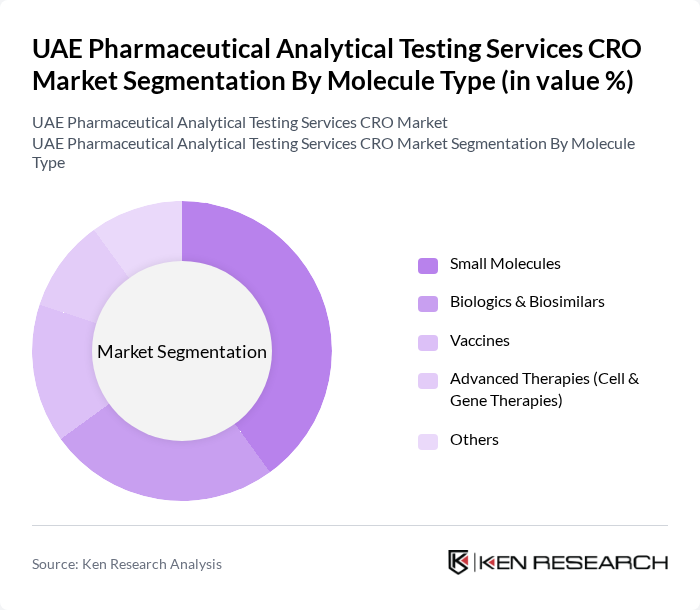
This segmentation includes Small Molecules, Biologics & Biosimilars, Vaccines, Advanced Therapies (Cell & Gene Therapies), and Others. The Small Molecules segment is currently the dominant category, primarily due to the extensive use of small molecule drugs in various therapeutic areas. The established market for these drugs, combined with ongoing research and development efforts, ensures that this segment remains a key driver of growth in the analytical testing services market.
The UAE Pharmaceutical Analytical Testing Services CRO Market is characterized by a dynamic mix of regional and international players. Leading participants such as **Eurofins Scientific**, **SGS SA**, **Intertek Group plc**, **Charles River Laboratories International, Inc.**, **Pace Analytical Services, LLC**, **Labcorp Drug Development (Laboratory Corporation of America Holdings)**, **Covance Inc.**, **Medpace Holdings, Inc.**, **WuXi AppTec Co., Ltd.**, **Bioanalytical Systems, Inc. (BASi)**, **KCR S.A.**, **PRA Health Sciences, Inc.**, **Clinipace Inc.**, **Syneos Health, Inc.**, **Celerion, Inc.** contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE pharmaceutical analytical testing services market appears promising, driven by technological advancements and increasing collaboration between industry stakeholders. The integration of AI and machine learning is expected to enhance testing accuracy and efficiency, while the expansion of healthcare infrastructure will support the growth of analytical services. As regulatory frameworks evolve, CROs will need to adapt to new compliance requirements, ensuring they remain competitive in a rapidly changing landscape.
| Segment | Sub-Segments |
|---|---|
| By Service Type | **Bioanalytical Testing Services** **Method Development & Validation** **Stability Testing & Storage** **Raw Material & Finished Product Testing** **Microbiological Testing & Environmental Monitoring** **Physical & Chemical Characterization** Others |
| By Molecule Type | **Small Molecules** **Biologics & Biosimilars** **Vaccines** **Advanced Therapies (Cell & Gene Therapies)** Others |
| By Testing Stage | **Early Development (Preclinical)** **Clinical Development (Phase I–III)** **Commercial & Post-Marketing (Phase IV)** **Lifecycle Management / Line Extensions** Others |
| By Product Type Tested | **Active Pharmaceutical Ingredients (APIs)** **Finished Dosage Forms** **Excipients & Raw Materials** **Packaging & Container-Closure Systems** Others |
| By End-User | **Pharmaceutical & Biopharmaceutical Companies** **Biotechnology Firms & Startups** **Clinical Research Organizations (CROs)** **Academic & Research Institutions** **Government & Regulatory Agencies** Others |
| By Geography (Within UAE) | **Abu Dhabi** **Dubai** **Sharjah** **Ajman & Northern Emirates** Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Quality Control | 90 | Quality Assurance Managers, Laboratory Directors |
| Regulatory Compliance Testing | 70 | Regulatory Affairs Specialists, Compliance Officers |
| Stability Testing Services | 60 | R&D Scientists, Product Development Managers |
| Method Validation Services | 50 | Analytical Chemists, Laboratory Technicians |
| Impurity Testing Services | 80 | Pharmaceutical Analysts, Quality Control Supervisors |
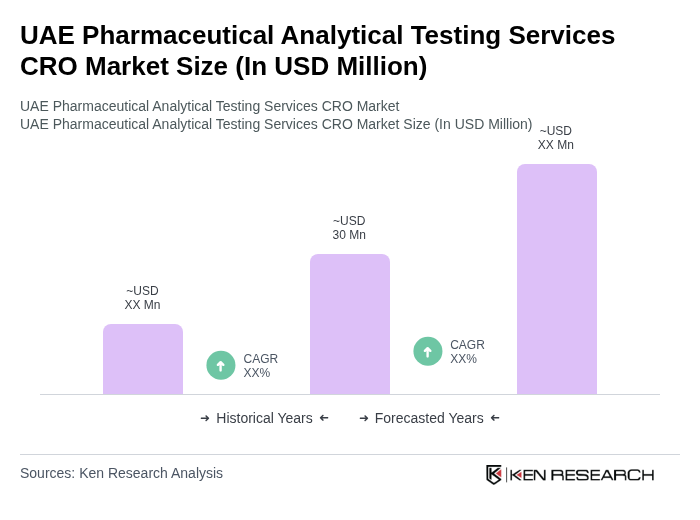
The UAE Pharmaceutical Analytical Testing Services CRO Market is valued at approximately USD 30 million, reflecting a five-year historical analysis. This growth is driven by the increasing demand for high-quality pharmaceutical products and stringent regulatory requirements.