Region:Middle East
Author(s):Geetanshi
Product Code:KRAC9572
Pages:81
Published On:November 2025
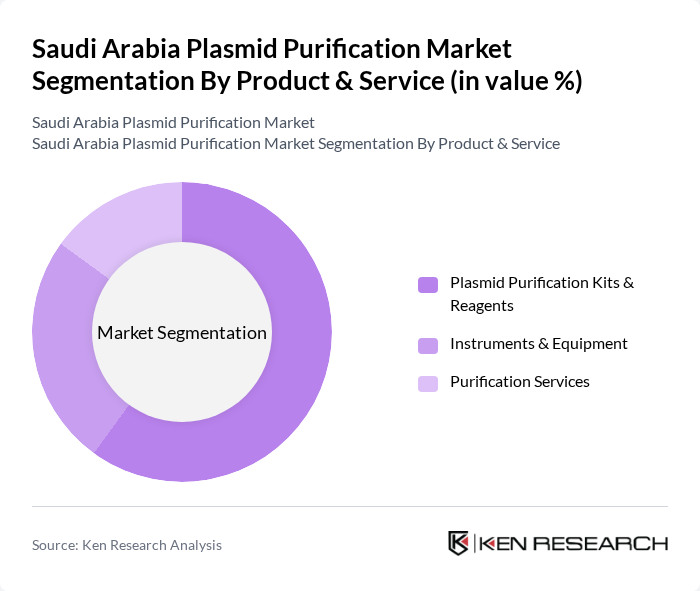
By Product & Service:The product and service segmentation includes Plasmid Purification Kits & Reagents, Instruments & Equipment, and Purification Services. Among these, Plasmid Purification Kits & Reagents dominate the market due to their essential role in various applications, including gene therapy and vaccine development. The increasing focus on personalized medicine, the adoption of automated purification systems, and the growing number of research projects in biotechnology are driving the demand for these kits. Instruments & Equipment and Purification Services also contribute significantly, but the kits and reagents are preferred for their convenience, efficiency, and ability to deliver high-purity plasmid DNA for downstream applications .
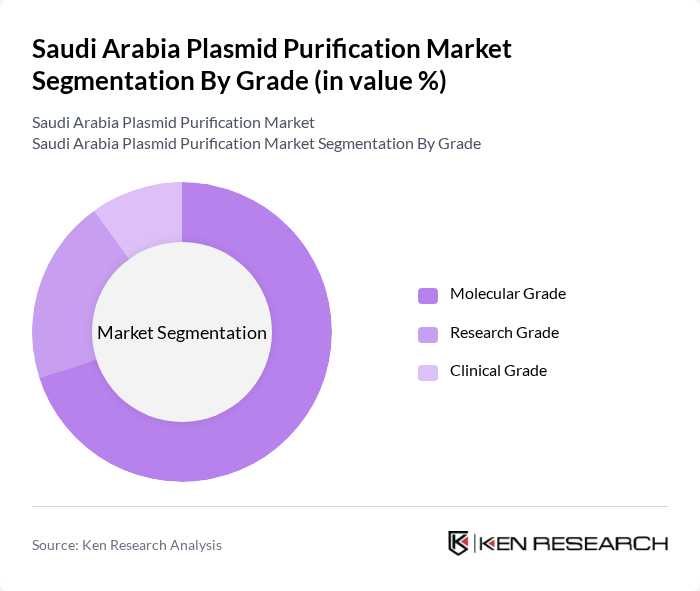
By Grade:The grade segmentation encompasses Molecular Grade, Research Grade, and Clinical Grade. The Molecular Grade segment is leading the market, primarily due to its widespread use in research and development activities. As the demand for high-quality plasmid DNA increases, particularly in gene therapy and vaccine production, the Molecular Grade products are preferred for their purity and reliability. Research Grade and Clinical Grade products also play vital roles, but the Molecular Grade is favored for its versatility across various applications .
The Saudi Arabia Plasmid Purification Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific, QIAGEN, Merck KGaA (MilliporeSigma), Bio-Rad Laboratories, Promega Corporation, Agilent Technologies, Takara Bio Inc., New England Biolabs, GenScript Biotech Corporation, SeraCare Life Sciences (now part of LGC Biosearch Technologies), Lonza Group AG, Sartorius AG, SaudiVax Ltd., Charles River Laboratories, Integrated DNA Technologies (IDT) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia plasmid purification market appears promising, driven by increasing investments in biotechnology and a growing emphasis on personalized medicine. As the government continues to support research and development initiatives, the market is likely to witness significant advancements in purification technologies. Additionally, the integration of automation and artificial intelligence in purification processes is expected to enhance efficiency and reduce costs, making plasmid purification more accessible to a broader range of stakeholders in the healthcare sector.
| Segment | Sub-Segments |
|---|---|
| By Product & Service | Plasmid Purification Kits & Reagents Instruments & Equipment Purification Services |
| By Grade | Molecular Grade Research Grade Clinical Grade |
| By Application | Gene Therapy Vaccine Development Research and Development Diagnostics |
| By End-User | Pharmaceutical & Biopharmaceutical Companies Academic & Research Institutions Contract Development & Manufacturing Organizations (CDMOs) Others |
| By Technology | Column Chromatography Precipitation Methods Filtration Techniques Affinity Chromatography Others |
| By Scale of Operation | Small Scale Medium Scale Large Scale |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotechnology Firms | 60 | R&D Managers, Lab Directors |
| Pharmaceutical Companies | 50 | Procurement Managers, Quality Control Officers |
| Academic Research Institutions | 40 | Principal Investigators, Research Scientists |
| Contract Research Organizations (CROs) | 45 | Operations Managers, Project Leads |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
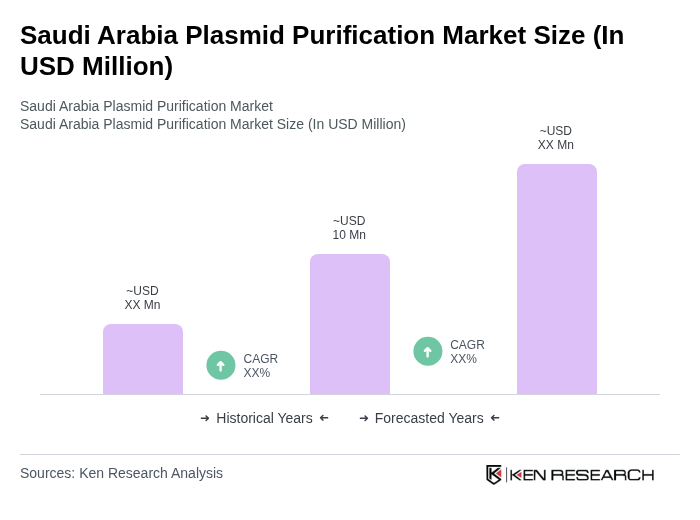
The Saudi Arabia Plasmid Purification Market is valued at approximately USD 10 million, driven by the increasing demand for plasmid DNA in gene therapy, vaccine development, and research applications.