Region:Middle East
Author(s):Shubham
Product Code:KRAD1919
Pages:99
Published On:December 2025
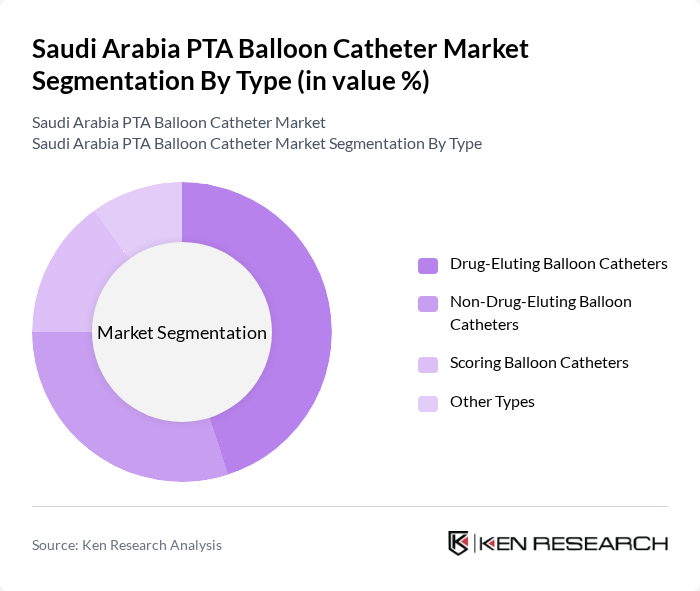
By Type:The market is segmented into various types of PTA balloon catheters, including Drug-Eluting Balloon Catheters, Non-Drug-Eluting Balloon Catheters, Scoring Balloon Catheters, and Other Types. Among these, Drug-Eluting Balloon Catheters are gaining traction due to their effectiveness in reducing restenosis rates and improving patient outcomes. The increasing preference for minimally invasive procedures is driving the demand for these advanced catheters, making them a dominant choice in interventional cardiology.
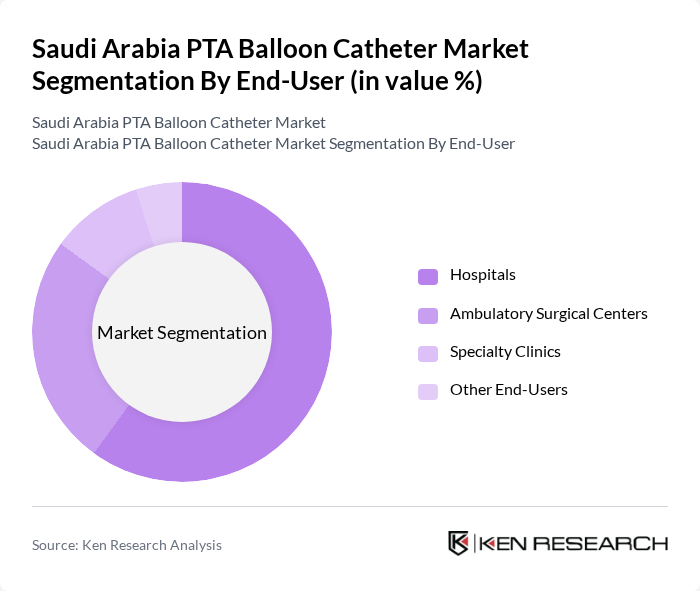
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, and Other End-Users. Hospitals are the primary end-users of PTA balloon catheters, driven by the high volume of cardiovascular procedures performed in these facilities. The increasing number of specialized cardiac centers within hospitals enhances the adoption of advanced catheter technologies, further solidifying their market leadership.
The Saudi Arabia PTA Balloon Catheter Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, B. Braun Melsungen AG, Terumo Corporation, Cordis Corporation, Cook Medical, Johnson & Johnson, Asahi Intecc Co., Ltd., Merit Medical Systems, Inc., AngioDynamics, Inc., Spectranetics Corporation, Philips Healthcare, Stryker Corporation, Biotronik SE & Co. KG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the PTA balloon catheter market in Saudi Arabia appears promising, driven by ongoing healthcare reforms and technological advancements. The government's commitment to enhancing healthcare infrastructure under Vision 2030 is expected to facilitate increased access to advanced medical devices. Additionally, the growing trend towards minimally invasive procedures will likely boost demand for PTA balloon catheters, as patients and providers seek effective, less invasive treatment options. Continued investment in medical technology will further support market expansion.
| Segment | Sub-Segments |
|---|---|
| By Type | Drug-Eluting Balloon Catheters Non-Drug-Eluting Balloon Catheters Scoring Balloon Catheters Other Types |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Other End-Users |
| By Application | Coronary Angioplasty Peripheral Angioplasty Other Applications |
| By Material | Polyurethane Polyethylene Other Materials |
| By Distribution Channel | Direct Sales Distributors Online Sales Other Channels |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Others | Custom Balloon Catheters Specialty Balloon Catheters |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 100 | Cardiologists, Interventional Radiologists |
| Medical Device Distributors | 80 | Sales Managers, Product Specialists |
| Healthcare Procurement Units | 70 | Procurement Officers, Supply Chain Managers |
| Clinical Research Organizations | 60 | Clinical Researchers, Regulatory Affairs Specialists |
| Patient Advocacy Groups | 50 | Patient Representatives, Healthcare Advocates |
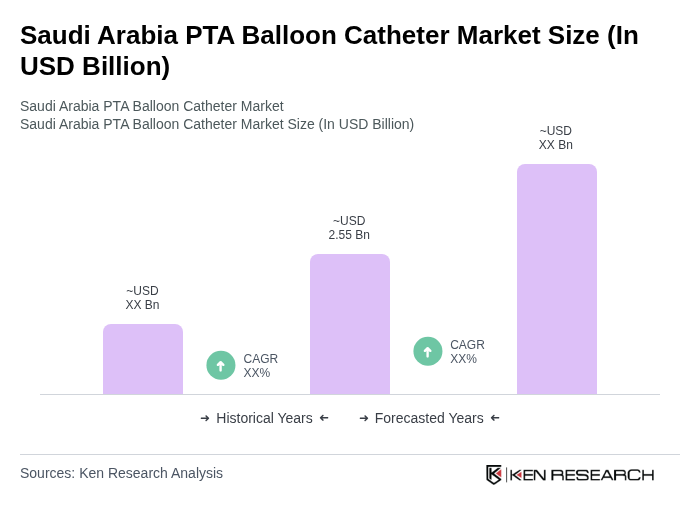
The Saudi Arabia PTA Balloon Catheter Market is valued at approximately USD 2.55 billion. This valuation reflects the increasing demand for minimally invasive interventions due to the rising prevalence of cardiovascular diseases and an aging population in the region.