About the Report
Base Year 2024Saudi Arabia South Korea Botulinum Toxin Type A Market Overview
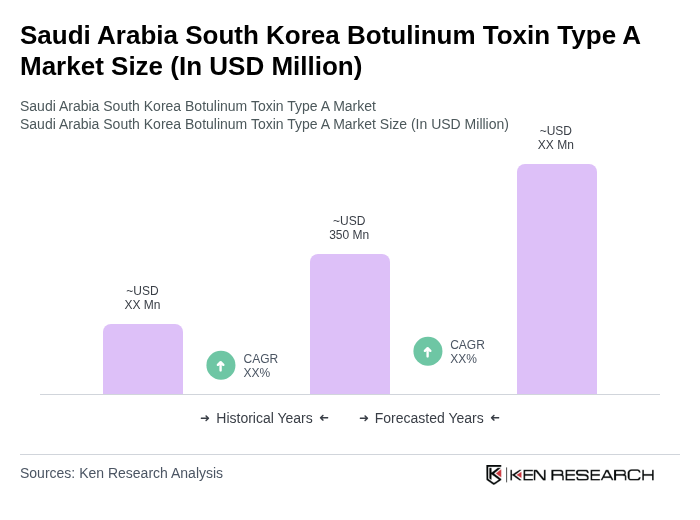
- The Saudi Arabia South Korea Botulinum Toxin Type A Market is valued at USD 350 million, based on a five-year historical analysis of the combined botulinum toxin markets in both countries. The South Korea botulinum toxin market generated around USD 305 million in revenue in a recent base year, while Saudi Arabia generated close to USD 50 million, with Botulinum Toxin Type A accounting for the dominant share in each market. This growth is primarily driven by increasing demand for aesthetic procedures, rising awareness about minimally invasive cosmetic treatments, growing medical tourism (particularly in South Korea), and expanding therapeutic applications of botulinum toxin in indications such as cervical dystonia, spasticity, chronic migraine, and hyperhidrosis.
- Key growth hubs in this market include major cities such as Riyadh in Saudi Arabia and Seoul in South Korea, which dominate due to their advanced healthcare infrastructure, concentration of specialized dermatology and aesthetic clinics, higher disposable incomes, and large populations seeking aesthetic enhancements. In Saudi Arabia, most facial injectable and aesthetic procedures are concentrated in Riyadh and Jeddah owing to well-developed healthcare facilities, while in South Korea, Seoul hosts hundreds of aesthetic clinics and is recognized as one of the leading global destinations for cosmetic procedures.
- In 2023, the Saudi Arabian government further strengthened the regulatory framework governing botulinum toxin products through requirements enforced by the Saudi Food and Drug Authority (SFDA), including marketing authorization for each toxin product, classification as prescription-only medicines, and controls on advertising and distribution. The SFDA’s Pharmaceutical Products Registration system and related guidance impose stringent approval processes for new botulinum toxin products, covering quality, safety, and efficacy data, while licensing requirements for healthcare facilities and practitioners ensure that only qualified, licensed physicians administer injectable neurotoxins in appropriate clinical settings. Complementing this, South Korea regulates botulinum toxin as a prescription pharmaceutical under the Pharmaceutical Affairs Act administered by the Ministry of Food and Drug Safety (MFDS), which mandates product approval, Good Manufacturing Practice compliance, and professional qualification standards for physicians performing aesthetic and therapeutic injections.
Saudi Arabia South Korea Botulinum Toxin Type A Market Segmentation
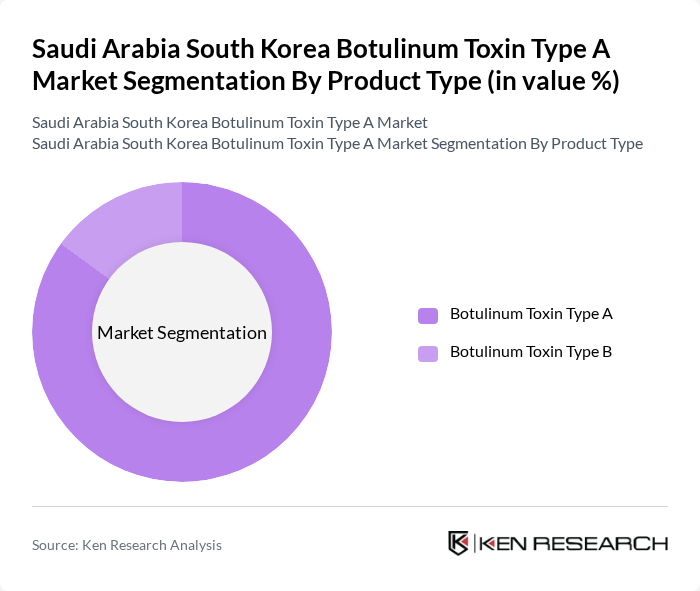
By Product Type:The product type segmentation includes Botulinum Toxin Type A and Botulinum Toxin Type B. Botulinum Toxin Type A is the leading subsegment, primarily due to its widespread use in aesthetic applications and therapeutic treatments, and accounts for the overwhelming majority of revenue in both Saudi Arabia and South Korea, where Type A represents close to the entire national botulinum toxin market. The demand for Botulinum Toxin Type B is comparatively lower, as it is mainly utilized for specific medical conditions such as cervical dystonia and is less commonly used for routine cosmetic procedures.
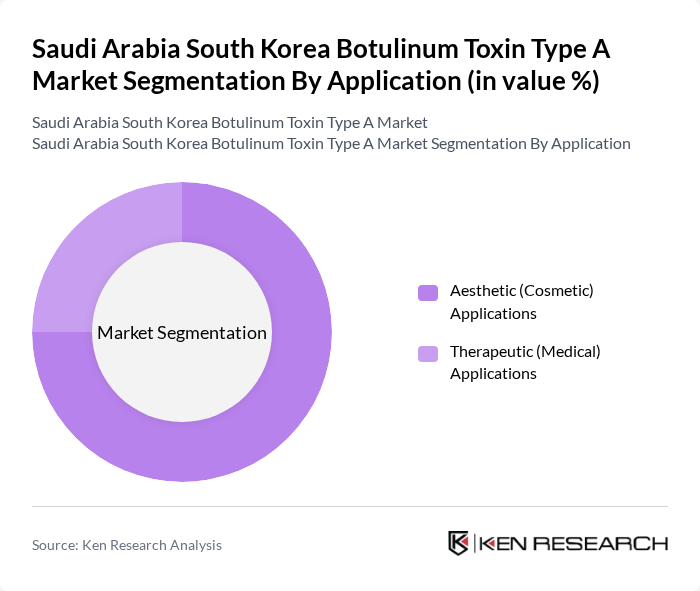
By Application:The application segmentation consists of Aesthetic (Cosmetic) Applications and Therapeutic (Medical) Applications. Aesthetic applications dominate the market, driven by increasing consumer interest in cosmetic enhancements, anti-aging treatments, and non-surgical procedures, along with strong medical tourism flows into South Korea for minimally invasive aesthetic solutions. Therapeutic applications, including treatment of neuromuscular and movement disorders, chronic migraine, and spasticity, are growing steadily as clinical indications expand and physician familiarity increases, but remain secondary in terms of overall market share.
Saudi Arabia South Korea Botulinum Toxin Type A Market Competitive Landscape
The Saudi Arabia South Korea Botulinum Toxin Type A Market is characterized by a dynamic mix of regional and international players. Leading participants such as AbbVie (Allergan Aesthetics), Ipsen, Merz Pharma, Galderma, Medytox, Hugel, Daewoong Pharmaceutical, Evolus, Lanzhou Institute of Biological Products, US WorldMeds, Metabiologics contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia South Korea Botulinum Toxin Type A Market Industry Analysis
Growth Drivers
- Increasing Demand for Aesthetic Procedures:The aesthetic procedures market in Saudi Arabia and South Korea is projected to reach approximately $1.7 billion, driven by a growing population seeking cosmetic enhancements. The rise in social media influence has led to a 35% increase in demand for non-surgical treatments, particularly botulinum toxin injections, as individuals prioritize appearance and self-esteem. This trend is supported by a cultural shift towards acceptance of cosmetic procedures, further fueling market growth.
- Rising Awareness about Non-Surgical Treatments:In future, it is estimated that around 70% of the population in both countries will be aware of non-surgical aesthetic treatments, a significant increase from previous years. This awareness is largely attributed to educational campaigns and increased media coverage, which have highlighted the benefits and safety of botulinum toxin procedures. As a result, more individuals are opting for these minimally invasive options, contributing to market expansion.
- Technological Advancements in Botulinum Toxin Formulations:The introduction of advanced botulinum toxin formulations, such as longer-lasting and more effective products, is expected to enhance treatment outcomes. In future, the market will see a 30% increase in the adoption of these innovative formulations, driven by ongoing research and development efforts. This technological progress not only improves patient satisfaction but also encourages more practitioners to offer these treatments, thereby boosting market growth.
Market Challenges
- Stringent Regulatory Requirements:The botulinum toxin market faces significant challenges due to stringent regulatory frameworks in both Saudi Arabia and South Korea. In future, the approval process for new products may take up to 20 months, delaying market entry for innovative solutions. Compliance with these regulations requires substantial investment in clinical trials and documentation, which can deter smaller companies from entering the market, limiting competition and innovation.
- Risk of Adverse Effects and Patient Safety Concerns:Patient safety remains a critical concern, with reports indicating that approximately 6% of botulinum toxin procedures result in adverse effects. These risks can lead to negative publicity and decreased consumer confidence in aesthetic treatments. In future, heightened scrutiny from regulatory bodies may result in stricter guidelines, further complicating the landscape for practitioners and manufacturers, and potentially hindering market growth.
Saudi Arabia South Korea Botulinum Toxin Type A Market Future Outlook
The future of the botulinum toxin market in Saudi Arabia and South Korea appears promising, driven by increasing consumer demand for aesthetic enhancements and advancements in treatment technologies. As the healthcare infrastructure continues to expand, more individuals will have access to these procedures. Additionally, the integration of telemedicine and online consultations is expected to facilitate patient access, making aesthetic treatments more convenient and appealing, thus further propelling market growth in the coming years.
Market Opportunities
- Growth in Medical Tourism:The rise in medical tourism, particularly in Saudi Arabia, presents a significant opportunity for the botulinum toxin market. In future, the country is expected to attract over 1.2 million medical tourists, many seeking aesthetic procedures. This influx can drive demand for botulinum toxin treatments, benefiting local clinics and practitioners while enhancing the overall market landscape.
- Expansion of Product Applications Beyond Aesthetics:There is a growing trend towards utilizing botulinum toxin for therapeutic applications, such as treating migraines and excessive sweating. In future, the therapeutic segment is projected to account for 25% of the total market share, providing new revenue streams for manufacturers and practitioners. This diversification can significantly enhance market resilience and growth potential.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Botulinum Toxin Type A Botulinum Toxin Type B |
| By Application | Aesthetic (Cosmetic) Applications Therapeutic (Medical) Applications |
| By End User | Hospitals Specialty Clinics & Aesthetic Centers Dermatology Clinics Medical Spas Others |
| By Patient Demographics | By Age (Below 30 Years, 31–59 Years, 60+ Years) By Gender (Female, Male) |
| By Distribution Channel | Direct Sales to Healthcare Facilities Distributor / Wholesaler Channels Online / E-commerce Channels Others |
| By Country | Saudi Arabia South Korea |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health of South Korea)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Companies
Healthcare Providers and Clinics
Industry Associations (e.g., Korean Society of Aesthetic Plastic Surgery)
Financial Institutions
Players Mentioned in the Report:
AbbVie (Allergan Aesthetics)
Ipsen
Merz Pharma
Galderma
Medytox
Hugel
Daewoong Pharmaceutical
Evolus
Lanzhou Institute of Biological Products
US WorldMeds
Metabiologics
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia South Korea Botulinum Toxin Type A Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia South Korea Botulinum Toxin Type A Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia South Korea Botulinum Toxin Type A Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for aesthetic procedures
3.1.2 Rising awareness about non-surgical treatments
3.1.3 Expanding healthcare infrastructure
3.1.4 Technological advancements in botulinum toxin formulations
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition among key players
3.2.3 Risk of adverse effects and patient safety concerns
3.2.4 Limited reimbursement policies
3.3 Market Opportunities
3.3.1 Growth in medical tourism
3.3.2 Expansion of product applications beyond aesthetics
3.3.3 Increasing disposable income among consumers
3.3.4 Collaborations with dermatology clinics
3.4 Market Trends
3.4.1 Shift towards minimally invasive procedures
3.4.2 Rising popularity of combination therapies
3.4.3 Growth in online consultations and telemedicine
3.4.4 Increasing focus on personalized medicine
3.5 Government Regulation
3.5.1 Approval processes for new botulinum toxin products
3.5.2 Guidelines for clinical usage and safety
3.5.3 Regulations on advertising and marketing practices
3.5.4 Monitoring and reporting of adverse events
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia South Korea Botulinum Toxin Type A Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia South Korea Botulinum Toxin Type A Market Segmentation
8.1 By Product Type
8.1.1 Botulinum Toxin Type A
8.1.2 Botulinum Toxin Type B
8.2 By Application
8.2.1 Aesthetic (Cosmetic) Applications
8.2.2 Therapeutic (Medical) Applications
8.3 By End User
8.3.1 Hospitals
8.3.2 Specialty Clinics & Aesthetic Centers
8.3.3 Dermatology Clinics
8.3.4 Medical Spas
8.3.5 Others
8.4 By Patient Demographics
8.4.1 By Age (Below 30 Years, 31–59 Years, 60+ Years)
8.4.2 By Gender (Female, Male)
8.5 By Distribution Channel
8.5.1 Direct Sales to Healthcare Facilities
8.5.2 Distributor / Wholesaler Channels
8.5.3 Online / E-commerce Channels
8.5.4 Others
8.6 By Country
8.6.1 Saudi Arabia
8.6.2 South Korea
9. Saudi Arabia South Korea Botulinum Toxin Type A Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Revenue (USD Million) from Botulinum Toxin Type A
9.2.3 3-year Revenue CAGR (%) – Botulinum Toxin Portfolio
9.2.4 Market Share (%) in Target Geographies
9.2.5 Average Selling Price Positioning (Premium / Mid / Value)
9.2.6 EBITDA Margin (%) – Aesthetic / Neurotoxin Segment
9.2.7 R&D Intensity (% of Sales Invested in R&D)
9.2.8 Number of Approved Botulinum Toxin Type A SKUs
9.2.9 Geographic Footprint (Number of Countries Served)
9.2.10 Distribution Reach (Number of Clinics / Hospitals Covered)
9.2.11 Patient Volume Treated Annually (Estimated)
9.2.12 Brand Awareness / Preference Score (Survey-based)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 AbbVie (Allergan Aesthetics)
9.5.2 Ipsen
9.5.3 Merz Pharma
9.5.4 Galderma
9.5.5 Medytox
9.5.6 Hugel
9.5.7 Daewoong Pharmaceutical
9.5.8 Evolus
9.5.9 Lanzhou Institute of Biological Products
9.5.10 US WorldMeds
9.5.11 Metabiologics
10. Saudi Arabia South Korea Botulinum Toxin Type A Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Key Projects
10.2.3 Funding Sources
10.2.4 Future Projections
10.3 Pain Point Analysis by End-User Category
10.3.1 Aesthetic Clinics
10.3.2 Hospitals
10.3.3 Medical Spas
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Technology Acceptance
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Case Studies
10.5.3 Future Use Cases
10.5.4 Others
11. Saudi Arabia South Korea Botulinum Toxin Type A Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health and beauty industry associations in Saudi Arabia and South Korea
- Review of published articles and journals focusing on botulinum toxin applications and market trends
- Examination of regulatory frameworks and guidelines from health authorities in both countries
Primary Research
- Interviews with dermatologists and cosmetic surgeons specializing in botulinum toxin treatments
- Surveys conducted with distributors and suppliers of botulinum toxin products
- Focus groups with consumers who have undergone botulinum toxin procedures
Validation & Triangulation
- Cross-validation of findings through multiple data sources including sales data and expert opinions
- Triangulation of market insights from healthcare professionals, distributors, and consumer feedback
- Sanity checks through expert panel discussions to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and cosmetic surgery trends
- Segmentation of the market by application areas such as aesthetic and therapeutic uses
- Incorporation of demographic data to assess potential market growth in different age groups
Bottom-up Modeling
- Collection of sales data from leading botulinum toxin manufacturers and distributors
- Estimation of average treatment costs and frequency of procedures across different demographics
- Volume estimates based on the number of procedures performed annually in both countries
Forecasting & Scenario Analysis
- Multi-variable forecasting using factors such as economic growth, consumer spending, and healthcare advancements
- Scenario analysis based on potential regulatory changes and shifts in consumer preferences
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Aesthetic Procedures Market | 150 | Dermatologists, Cosmetic Surgeons |
| Therapeutic Applications | 100 | Neurologists, Pain Management Specialists |
| Consumer Insights on Botulinum Toxin | 150 | Patients, Beauty Enthusiasts |
| Distribution Channels Analysis | 80 | Pharmaceutical Distributors, Medical Supply Managers |
| Market Trends and Innovations | 70 | Industry Experts, Market Analysts |
Frequently Asked Questions
What is the current value of the Botulinum Toxin Type A market in Saudi Arabia and South Korea?
The combined Botulinum Toxin Type A market in Saudi Arabia and South Korea is valued at approximately USD 350 million, with South Korea contributing around USD 305 million and Saudi Arabia about USD 50 million, reflecting significant market activity in both regions.