About the Report
Base Year 2024Saudi Arabia Therapeutic Respiratory Devices Market Overview
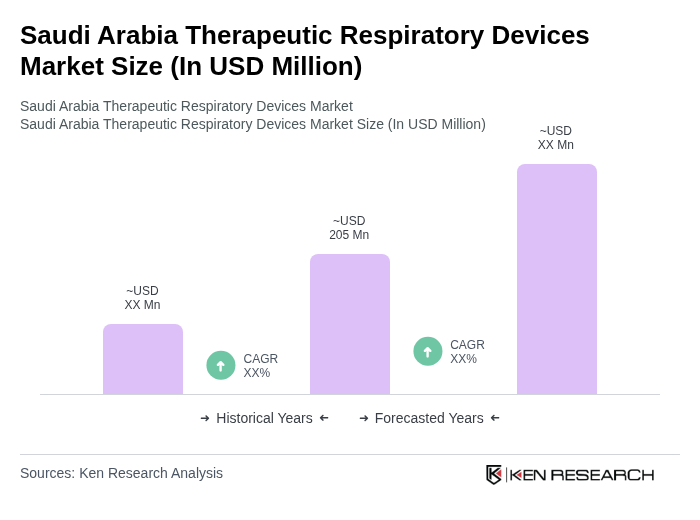
- The Saudi Arabia Therapeutic Respiratory Devices Market is valued at USD 205 million, based on a five-year historical analysis. Growth is primarily driven by the increasing prevalence of chronic respiratory diseases such as asthma, COPD, and sleep apnea, rising healthcare expenditure, and advancements in technology that enhance device efficiency and patient outcomes. The adoption of smart and connected respiratory devices, as well as portable solutions, is accelerating market expansion. Government initiatives under Vision 2030, which emphasize healthcare innovation and access, further stimulate demand for advanced respiratory care solutions .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their well-established healthcare infrastructure, high population density, and the presence of leading healthcare facilities. These cities are also focal points for medical tourism, further boosting the demand for therapeutic respiratory devices .
- In 2023, the Saudi Food and Drug Authority (SFDA) issued the "Medical Devices Interim Regulation" (SFDA.MD/RI 2023), mandating that all healthcare facilities must comply with updated safety and performance standards for respiratory devices. This regulation covers device registration, post-market surveillance, and strict adherence to safety protocols, ensuring patient safety and improving the quality of care, thereby driving the demand for compliant therapeutic devices in the market .
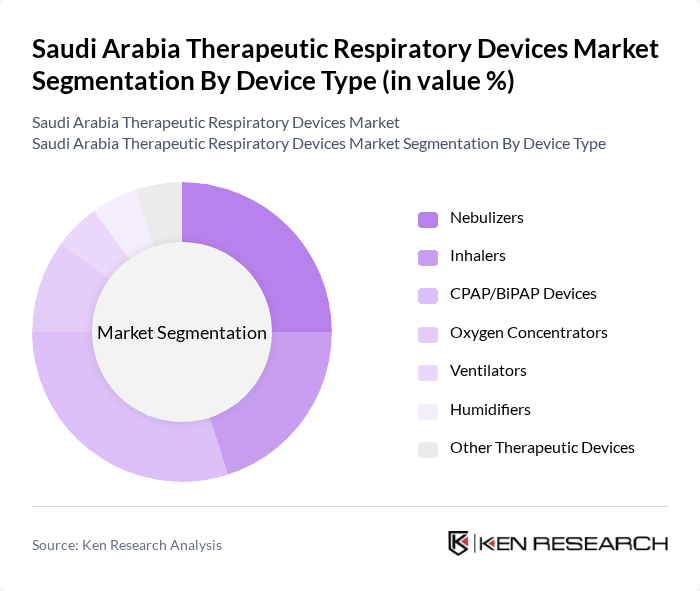
Saudi Arabia Therapeutic Respiratory Devices Market Segmentation
By Device Type:The market is segmented into various device types, including nebulizers, inhalers, CPAP/BiPAP devices, oxygen concentrators, ventilators, humidifiers, and other therapeutic devices. Among these, nebulizers and CPAP/BiPAP devices are particularly prominent due to their widespread use in treating chronic respiratory conditions. The increasing awareness of respiratory health, the growing elderly population, and the adoption of smart, portable devices contribute to the demand for these devices. The market is also witnessing a shift towards devices equipped with digital monitoring and cloud connectivity for improved patient compliance and outcomes .
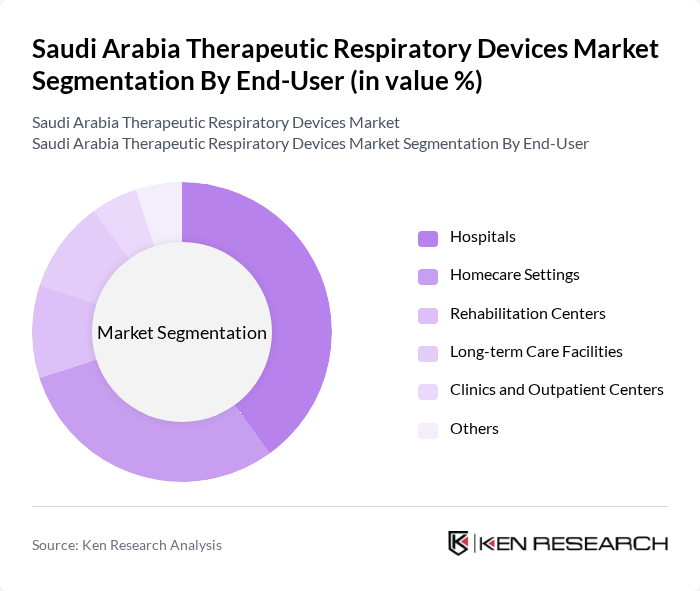
By End-User:The end-user segmentation includes hospitals, homecare settings, rehabilitation centers, long-term care facilities, clinics, and outpatient centers. Hospitals are the leading end-users due to their comprehensive respiratory care services and the high volume of patients requiring therapeutic devices. The trend towards home healthcare is gaining traction, driven by the increasing preference for at-home treatment options, the expansion of telemedicine, and the availability of portable respiratory devices. Rehabilitation centers and long-term care facilities are also increasing their adoption of advanced therapeutic devices to manage chronic respiratory conditions .
Saudi Arabia Therapeutic Respiratory Devices Market Competitive Landscape
The Saudi Arabia Therapeutic Respiratory Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Philips Healthcare, ResMed Inc., Medtronic plc, Vyaire Medical, Hill-Rom Holdings Inc., Invacare Corporation, Teleflex Incorporated, Smiths Medical, Nihon Kohden Corporation, GE Healthcare, Getinge AB, Dräger Medical contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Therapeutic Respiratory Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Respiratory Diseases:The prevalence of respiratory diseases in Saudi Arabia is on the rise, with over 2 million cases of asthma reported in the future. Chronic obstructive pulmonary disease (COPD) affects approximately 1.5 million individuals, contributing to a significant demand for therapeutic respiratory devices. The World Health Organization estimates that respiratory diseases account for 10% of total healthcare expenditures, highlighting the urgent need for effective treatment solutions in the region.
- Rising Geriatric Population:In the future, the geriatric population in Saudi Arabia is projected to reach 4.5 million, representing a 20% increase from 2020. This demographic shift is critical as older adults are more susceptible to respiratory conditions, necessitating advanced therapeutic devices. The Saudi government has recognized this trend, allocating approximately $1 billion to enhance healthcare services for the elderly, thereby driving demand for respiratory solutions tailored to this age group.
- Technological Advancements in Respiratory Devices:The Saudi Arabian market is witnessing rapid technological advancements, with investments exceeding $500 million in R&D for respiratory devices in the future. Innovations such as smart inhalers and portable oxygen concentrators are becoming increasingly prevalent. These advancements not only improve patient outcomes but also enhance the efficiency of healthcare delivery, aligning with the country's Vision 2030 goals to modernize healthcare infrastructure and services.
Market Challenges
- High Cost of Advanced Therapeutic Devices:The high cost of advanced therapeutic respiratory devices poses a significant challenge, with prices ranging from $1,000 to $5,000 per unit. This financial barrier limits accessibility for many patients, particularly in lower-income segments. The Saudi government is working to address this issue, but the current economic climate, with a projected GDP growth of only 2.5% in the future, complicates efforts to subsidize these costs effectively.
- Limited Awareness Among Healthcare Providers:Despite the growing need for therapeutic respiratory devices, there remains a significant knowledge gap among healthcare providers in Saudi Arabia. A recent study indicated that only 40% of healthcare professionals are adequately trained in the use of these devices. This lack of awareness can lead to underutilization and mismanagement of respiratory conditions, ultimately affecting patient outcomes and hindering market growth.
Saudi Arabia Therapeutic Respiratory Devices Market Future Outlook
The future of the therapeutic respiratory devices market in Saudi Arabia appears promising, driven by ongoing healthcare reforms and technological innovations. The government’s commitment to enhancing healthcare infrastructure is expected to facilitate better access to advanced respiratory solutions. Additionally, the integration of telemedicine and remote monitoring technologies will likely transform patient management, making it more efficient and patient-centered. As the market evolves, collaboration between manufacturers and healthcare providers will be crucial for addressing challenges and maximizing opportunities.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Saudi government is investing heavily in healthcare infrastructure, with plans to build 30 new hospitals by the future. This expansion will create significant opportunities for therapeutic respiratory device manufacturers to establish partnerships and increase market penetration, ultimately improving patient access to essential treatments.
- Growing Demand for Home Healthcare Solutions:The demand for home healthcare solutions is surging, with an estimated 1.2 million patients in Saudi Arabia requiring at-home respiratory care in the future. This trend presents a lucrative opportunity for companies to develop and market portable respiratory devices, catering to the needs of patients seeking convenience and effective management of their conditions at home.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Device Type | Nebulizers Inhalers CPAP/BiPAP Devices Oxygen Concentrators Ventilators Humidifiers Other Therapeutic Devices |
| By End-User | Hospitals Homecare Settings Rehabilitation Centers Long-term Care Facilities Clinics and Outpatient Centers Others |
| By Distribution Channel | Direct Sales to Healthcare Facilities Online Retail Platforms Medical Supply Stores Pharmacies Authorized Distributors Others |
| By Application | Chronic Obstructive Pulmonary Disease (COPD) Asthma Sleep Apnea Acute Respiratory Infections Other Respiratory Conditions |
| By Technology | Manual Devices Electronic Devices Smart Connected Devices Cloud-Based Monitoring Systems Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Region | Central Region (Riyadh) Eastern Region Western Region (Jeddah) Northern Region Southern Region |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Insurance Companies and Payers
Industry Associations and Organizations
Pharmaceutical Companies
Players Mentioned in the Report:
Philips Healthcare
ResMed Inc.
Medtronic plc
Vyaire Medical
Hill-Rom Holdings Inc.
Invacare Corporation
Teleflex Incorporated
Smiths Medical
Nihon Kohden Corporation
GE Healthcare
Getinge AB
Drager Medical
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Therapeutic Respiratory Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Therapeutic Respiratory Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Therapeutic Respiratory Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of respiratory diseases
3.1.2 Rising geriatric population
3.1.3 Technological advancements in respiratory devices
3.1.4 Government initiatives for healthcare improvement
3.2 Market Challenges
3.2.1 High cost of advanced therapeutic devices
3.2.2 Limited awareness among healthcare providers
3.2.3 Regulatory hurdles in device approval
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in R&D
3.3.3 Growing demand for home healthcare solutions
3.3.4 Partnerships with local distributors
3.4 Market Trends
3.4.1 Shift towards portable and user-friendly devices
3.4.2 Integration of IoT in respiratory devices
3.4.3 Focus on preventive healthcare
3.4.4 Rise in telemedicine and remote monitoring
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced approval processes for new devices
3.5.3 Incentives for local manufacturing
3.5.4 Compliance with international standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Therapeutic Respiratory Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Therapeutic Respiratory Devices Market Segmentation
8.1 By Device Type
8.1.1 Nebulizers
8.1.2 Inhalers
8.1.3 CPAP/BiPAP Devices
8.1.4 Oxygen Concentrators
8.1.5 Ventilators
8.1.6 Humidifiers
8.1.7 Other Therapeutic Devices
8.2 By End-User
8.2.1 Hospitals
8.2.2 Homecare Settings
8.2.3 Rehabilitation Centers
8.2.4 Long-term Care Facilities
8.2.5 Clinics and Outpatient Centers
8.2.6 Others
8.3 By Distribution Channel
8.3.1 Direct Sales to Healthcare Facilities
8.3.2 Online Retail Platforms
8.3.3 Medical Supply Stores
8.3.4 Pharmacies
8.3.5 Authorized Distributors
8.3.6 Others
8.4 By Application
8.4.1 Chronic Obstructive Pulmonary Disease (COPD)
8.4.2 Asthma
8.4.3 Sleep Apnea
8.4.4 Acute Respiratory Infections
8.4.5 Other Respiratory Conditions
8.5 By Technology
8.5.1 Manual Devices
8.5.2 Electronic Devices
8.5.3 Smart Connected Devices
8.5.4 Cloud-Based Monitoring Systems
8.5.5 Others
8.6 By Patient Demographics
8.6.1 Pediatric Patients
8.6.2 Adult Patients
8.6.3 Geriatric Patients
8.6.4 Others
8.7 By Region
8.7.1 Central Region (Riyadh)
8.7.2 Eastern Region
8.7.3 Western Region (Jeddah)
8.7.4 Northern Region
8.7.5 Southern Region
9. Saudi Arabia Therapeutic Respiratory Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, or Small)
9.2.3 Year-over-Year Revenue Growth Rate (%)
9.2.4 Market Penetration Rate in Saudi Arabia (%)
9.2.5 Healthcare Facility Adoption Rate (%)
9.2.6 Product Innovation Index (New Products Launched Annually)
9.2.7 Distribution Network Coverage (Number of Authorized Distributors)
9.2.8 Average Device Pricing Strategy (Premium/Mid-Range/Budget)
9.2.9 Brand Recognition Score (1-10 Scale)
9.2.10 Customer Satisfaction Rating (Based on Healthcare Provider Feedback)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Philips Healthcare
9.5.2 ResMed Inc.
9.5.3 Medtronic plc
9.5.4 Vyaire Medical
9.5.5 Hill-Rom Holdings Inc.
9.5.6 Invacare Corporation
9.5.7 Teleflex Incorporated
9.5.8 Smiths Medical
9.5.9 Nihon Kohden Corporation
9.5.10 GE Healthcare
9.5.11 Getinge AB
9.5.12 Dräger Medical
10. Saudi Arabia Therapeutic Respiratory Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Healthcare Infrastructure
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Funding Sources
10.2.4 Project Timelines
10.3 Pain Point Analysis by End-User Category
10.3.1 Device Availability and Supply Chain Issues
10.3.2 Training and Technical Support Needs
10.3.3 Maintenance and Service Challenges
10.3.4 Cost and Budget Constraints
10.4 User Readiness for Adoption
10.4.1 Awareness Levels of Advanced Devices
10.4.2 Training Requirements
10.4.3 Technology Acceptance
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Satisfaction
10.5.3 Expansion Opportunities
10.5.4 Long-term Benefits
11. Saudi Arabia Therapeutic Respiratory Devices Market Future Size, 2025-2033
11.1 By Value (USD Million)
11.2 By Volume (Units)
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Assessment
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Competitive Advantage Analysis
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Metrics
3. Distribution Plan
3.1 Urban Healthcare Facility Strategies
3.2 Regional Hospital Network Tie-ups
3.3 E-commerce and Online Pharmacy Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
3.6 Inventory Management
3.7 Performance Tracking
4. Channel & Pricing Gaps
4.1 Underserved Healthcare Segments
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Healthcare Provider Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Volume Discount Strategies
4.7 Pricing Adjustments
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Healthcare Provider Segments Analysis
5.3 Product Development Opportunities
5.4 Market Entry Strategies
5.5 Healthcare Provider Feedback Utilization
5.6 Innovation Opportunities
5.7 Market Trends Alignment
6. Customer Relationship
6.1 Loyalty Programs for Healthcare Facilities
6.2 After-sales Service and Support
6.3 Technical Support Strategies
6.4 Feedback Mechanisms
6.5 Relationship Management Tools
6.6 Customer Retention Strategies
6.7 Performance Metrics
7. Value Proposition
7.1 Sustainability and Infection Control Initiatives
7.2 Integrated Supply Chains
7.3 Healthcare Provider-Centric Approaches
7.4 Competitive Differentiation
7.5 Value-Added Services
7.6 Market Positioning
7.7 Performance Metrics
8. Key Activities
8.1 Regulatory Compliance and SFDA Approval
8.2 Branding Initiatives
8.3 Distribution Setup
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare associations and government health ministries
- Review of published articles and white papers on respiratory device innovations and trends
- Examination of trade statistics and import/export data specific to therapeutic respiratory devices
Primary Research
- Interviews with healthcare professionals, including pulmonologists and respiratory therapists
- Surveys conducted with hospital procurement managers and medical device distributors
- Focus group discussions with patients using therapeutic respiratory devices
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including industry reports and expert opinions
- Triangulation of market data with sales figures from leading manufacturers
- Sanity checks through expert panel reviews comprising industry veterans and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and respiratory disease prevalence
- Segmentation of the market by device type, including nebulizers, inhalers, and CPAP machines
- Incorporation of government health initiatives aimed at improving respiratory health
Bottom-up Modeling
- Collection of sales data from major distributors and manufacturers of therapeutic respiratory devices
- Estimation of device usage rates based on patient demographics and disease incidence
- Calculation of market size using volume x average selling price for each device category
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population growth and healthcare access
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement for Respiratory Devices | 100 | Procurement Managers, Medical Equipment Buyers |
| Patient Experience with Inhalation Therapy | 70 | Chronic Respiratory Disease Patients, Caregivers |
| Market Insights from Respiratory Therapists | 50 | Respiratory Therapists, Clinical Specialists |
| Sales Insights from Device Manufacturers | 60 | Sales Managers, Product Development Leads |
| Regulatory Perspectives on Device Approvals | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Therapeutic Respiratory Devices Market?
The Saudi Arabia Therapeutic Respiratory Devices Market is valued at approximately USD 205 million, driven by the rising prevalence of chronic respiratory diseases and advancements in technology that enhance device efficiency and patient outcomes.