Region:Middle East
Author(s):Rebecca
Product Code:KRAA4569
Pages:84
Published On:September 2025
 Market.png)
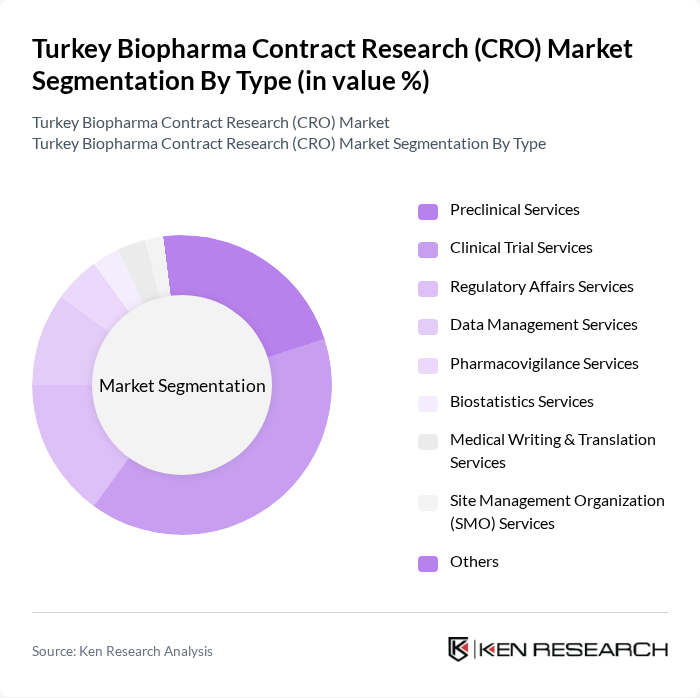
By Type:The Turkey Biopharma CRO Market is segmented into various types of services that address the diverse needs of biopharmaceutical companies. Clinical Trial Services is the leading segment, supported by the increasing volume of clinical trials and the demand for efficient trial management. Preclinical Services maintain a significant share as companies seek to validate drug candidates prior to clinical phases. Regulatory Affairs Services are essential for navigating Turkey's evolving regulatory landscape, while Data Management and Pharmacovigilance Services ensure data integrity and patient safety. The market is also witnessing increased demand for biostatistics, medical writing, and site management services as trials grow in complexity .
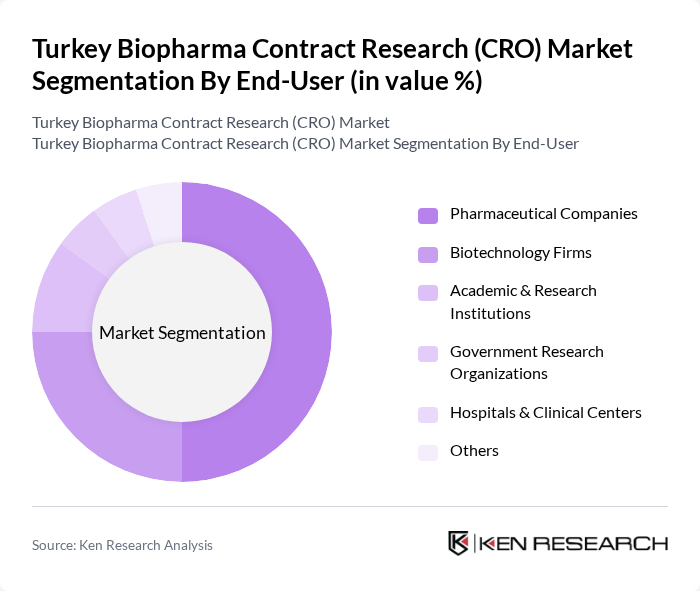
By End-User:The end-user segmentation of the Turkey Biopharma CRO Market includes Pharmaceutical Companies, Biotechnology Firms, Academic & Research Institutions, Government Research Organizations, and Hospitals & Clinical Centers. Pharmaceutical Companies lead due to their extensive clinical trial requirements and regulatory needs. Biotechnology Firms are increasingly prominent, focusing on innovative therapies and requiring specialized CRO support. Academic and government institutions contribute to research and early-phase studies, while hospitals and clinical centers play a key role in patient recruitment and trial execution .
The Turkey Biopharma Contract Research (CRO) Market is characterized by a dynamic mix of regional and international players. Leading participants such as ICON plc, IQVIA, Syneos Health, Parexel International Corporation, Labcorp Drug Development (formerly Covance), Medpace Holdings, Inc., KCR S.A., Ergomed plc, Pharmactive ?laç Sanayi ve Ticaret A.?., TRIAL Nation Clinical Research, Ac?badem Labmed Klinik Laboratuvarlar A.?., Anadolu Sa?l?k Merkezi (Johns Hopkins Medicine affiliate), SSS International Clinical Research, Dokuz Eylul Teknoloji Geli?tirme A.?. (DETTO), Worldwide Clinical Trials contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Turkey biopharma CRO market appears promising, driven by advancements in personalized medicine and the increasing adoption of digital health technologies. As the healthcare landscape evolves, CROs are expected to leverage data analytics and patient-centric approaches to enhance clinical trial efficiency. Furthermore, the collaboration between local and international CROs is likely to foster innovation, enabling Turkey to position itself as a competitive player in the global biopharma research arena.
| Segment | Sub-Segments |
|---|---|
| By Type | Preclinical Services Clinical Trial Services Regulatory Affairs Services Data Management Services Pharmacovigilance Services Biostatistics Services Medical Writing & Translation Services Site Management Organization (SMO) Services Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Government Research Organizations Hospitals & Clinical Centers Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Immunology & Autoimmune Diseases Rare Diseases/Orphan Drugs Others |
| By Service Model | Full-Service CROs Functional Service Providers (FSP) Hybrid Models |
| By Phase of Development | Phase I Phase II Phase III Phase IV |
| By Geographic Focus | Domestic Trials International Trials |
| By Pricing Model | Fixed Pricing Cost-Plus Pricing Performance-Based Pricing Risk-Sharing Models Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Management | 60 | Clinical Research Associates, Project Managers |
| Regulatory Affairs | 40 | Regulatory Affairs Managers, Compliance Officers |
| Biostatistics and Data Management | 45 | Biostatisticians, Data Managers |
| Pharmacovigilance Services | 40 | Pharmacovigilance Officers, Safety Managers |
| Market Access Strategies | 50 | Market Access Managers, Health Economists |
The Turkey Biopharma Contract Research (CRO) Market is valued at approximately USD 1.1 billion, reflecting a robust growth trajectory driven by increasing demand for biopharmaceuticals and a rise in clinical trials within the region.